 |
 |
 |
| |
Efficacy and Safety of Direct Antiviral Agents in Cirrhotic HCV-HIV Coinfected Patients
|
| |
| |
Reported by Jules Levin
CROI 2017 Feb 14-16 Seattle, WA
J. Navarro1,2, M. Laguno3, HH. Vilchez4, J. Guardiola5, J.A. Carrion6, L. Force7, M. Cairo8, C. Cifuentes9 and M. Crespo10 on Behalf of Catalano-Balear Study Grup
1Hospital Universitari de la Vall d'Hebron. Universitat Autonoma de Barcelona. Barcelona, Espana. 2Institut de Recerca Hospital Vall d'Hebron. Barcelona, Espana. 3Hospital Clinic de Barcelona. Barcelona, Espana. 4Hospital Universitari de Son Espases. Palma de Mallorca, Espana. 5Hospital de la Santa Creu i Sant Pau. Barcelona, Espana. 6Hospital del Mar, Institut Hospital del Mar d'Investigacions Mèdiques (IMIM). Barcelona, Espana. Universitat Autonoma de Barcelona (UAB). Barcelona, Espana. 7Hospital de Mataro. Mataro, Espana.8Hospital Universitari Mútua de Terrassa. Terrassa, Espana. 9Hospital de Son Llàtzer. Palma de Mallorca, Espana. 10Complexo Hospitalario Universitario de Vigo, IIS Galicia Sur, Espana.

Abstract Body:
There is scarce data regarding efficacy and safety of new direct antiviral agents (DAA) in cirrhotic HCV-HIV coinfected patients.
Multicenter prospective cohort analysis carried-out in 13 Spanish hospitals between January and December 2015. Inclusion criteria: 1) Cirrhosis diagnosed by transient elastography (TE>14.6KPa) or by sonographic, endoscopic and/or clinical data. 2) HCV-treatment based on DAA according to drug availability and physician discretion. 3) HCV-HIV coinfection with stable ART and controlled HIV infection. Primary endpoint: Overall efficacy, defined as the percentage of patients with undetectable HCV-RNA at week 12 after treatment (SVRS12). Secondary endpoints: Safety (percentage of withdrawal due to toxicity and/or hepatic decompensation) and efficacy according to regimen used and HCV genotype. Change in TE value after HCV treatment was also evaluated.
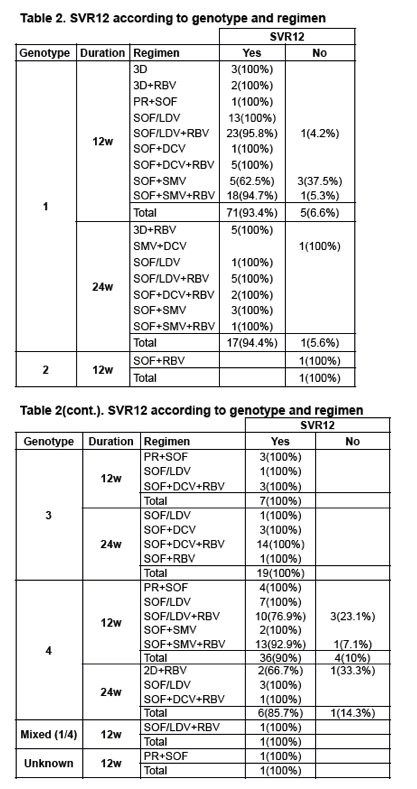
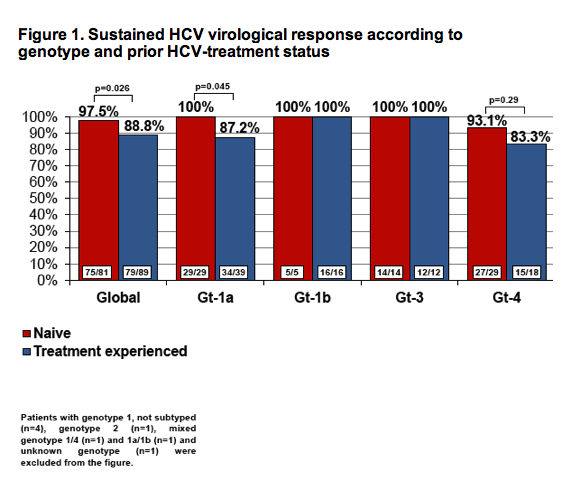
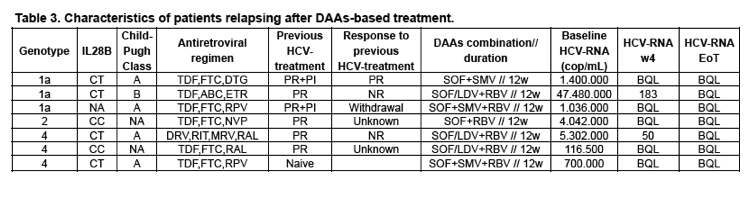
A total of 201 patients started treatment. Mostly, male (n=150; 74.3%), Caucasian (n=198; 98%) and ex-IDUs (n=165; 81.7%). Genotypes: Gt-1a, 75 (37.1%); Gt-1b, 27 (13.4%); Gt-4, 51 (25.2%) and Gt-3, 38 (18.8%). Baseline median TE was 20.7KPa (IQR 16.1-33) and HCV-RNA log10 6.1UI/mL (IQR 5.7-6.6). Most patients had Child-Pugh A class score (n=153; 75.7%) and 36 (17.8%) of them suffered prior hepatic decompensation. There were 104 (51.5%) pretreated patients, of whom 40.4% (n=42) with null response. SVR12 data was available in 170 (84.6%) patients. More commonly used regimens: SOF/LDV+RBV, 43 (25.3%) patients; SOF+SMV+RBV, 34 (20%); SOF/LDV, 26 (15.3%) and SOF+DCV+RBV, 25 (14.7%). Overall, 92.9% (158/170) of patients achieved SVR12, without differences between genotypes (Table 1). A significant lower SVR12 was observed in pretreated as compared to naive patients (88.8% vs. 97.5%; p=0.026). Causes of treatment failure were: 7 (4.1%) relapses, 2 (1.2%) lost to follow-up, 1 (0.6%) toxicity-related discontinuation, 1 (0.6%) hepatic decompensation and 1 (0.6%) viral breakthrough. RBV dose modification was needed in 20 (16.3%) cases, mainly due to anemia (n=17). On-treatment hepatic decompensation was seen in 4 (2.4%) patients (encephalopathy and ascites, 2 each). At week 12 after treatment, TE decreased a mean of 5.6 KPa (95%CI 1.8-9.2; p=0.004) as compared with baseline.
In our cohort, 92.9% of cirrhotic HCV-HIV coinfected patients achieved SVR12, which was associated with a significant decrease in TE. Only 2 (1.2%) patients had to stop treatment due to adverse events

|
| |
|
 |
 |
|
|