 |
 |
 |
| |
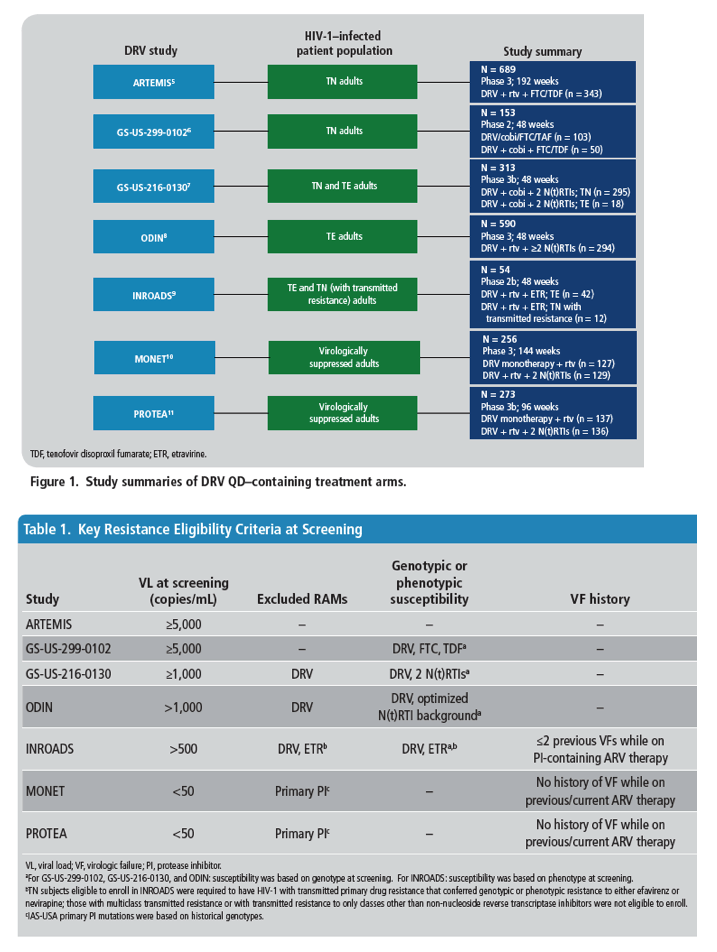
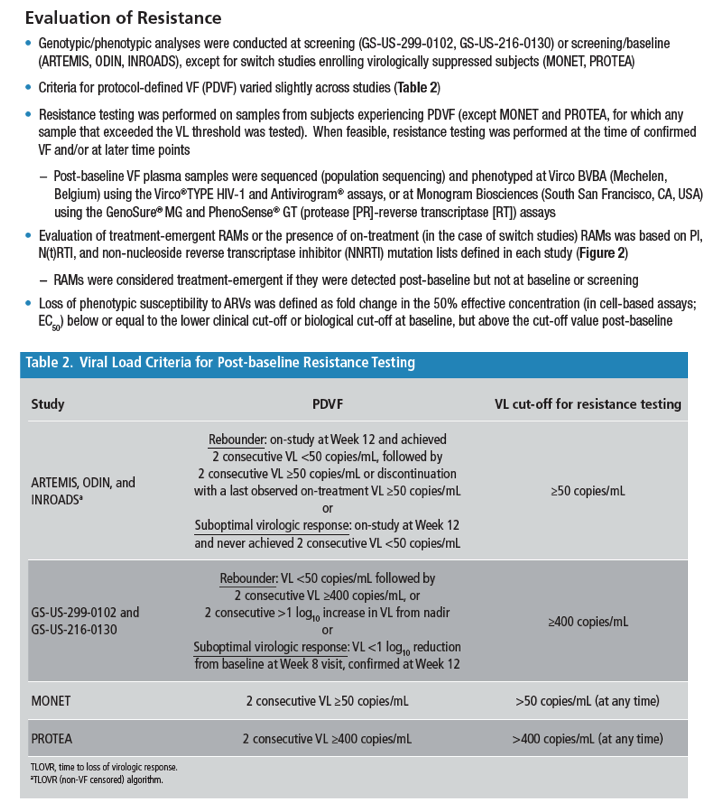
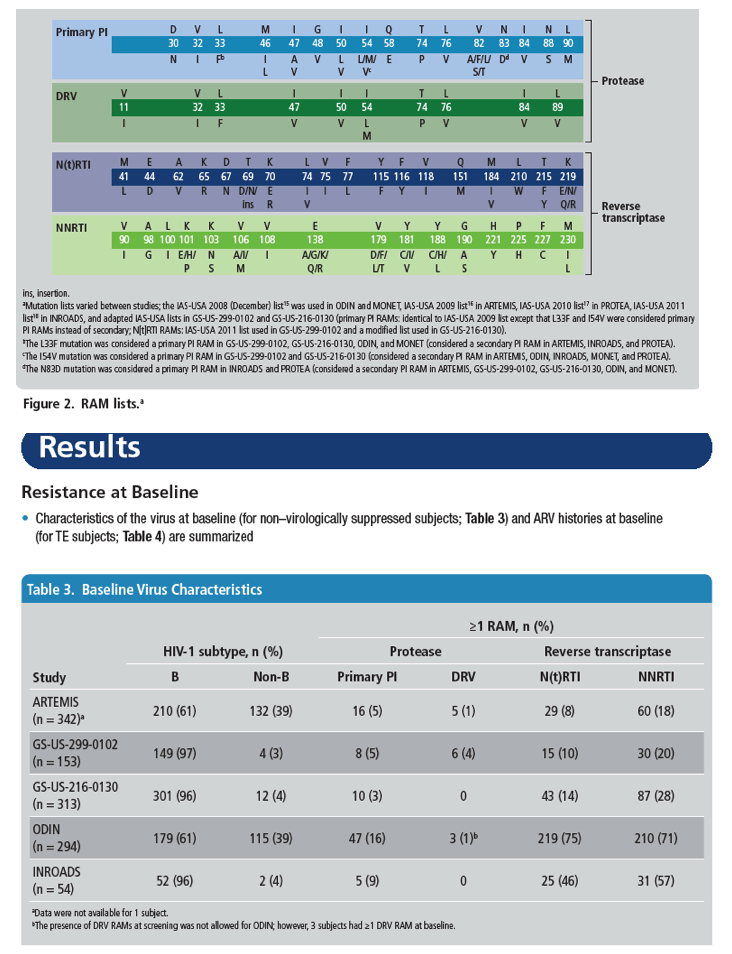
Low HIV-1 Resistance in Subjects Using Darunavir Once-daily Regimens Across Studies
|
| |
| |
Download the PDF here
HIV-Dart: Development of a protease inhibitor-based single-tablet complete HIV-1 regimen of darunavir/cobicistat/emtricitabine/tenofovir alafenamide (DCFTAF) - (12/15/16)
Reported by Jules Levin
CROI 2017 Feb 14-16 Seattle WA
Erkki Lathouwers,1,* Eric Y. Wong,2 Donghan Luo,2 Sareh Seyedkazemi,2 Sandra De Meyer,1 Kimberley Brown2
1Janssen Infectious Diseases BVBA, Beerse, Belgium; 2Janssen Scientific Affairs, LLC, Titusville, NJ, USA.

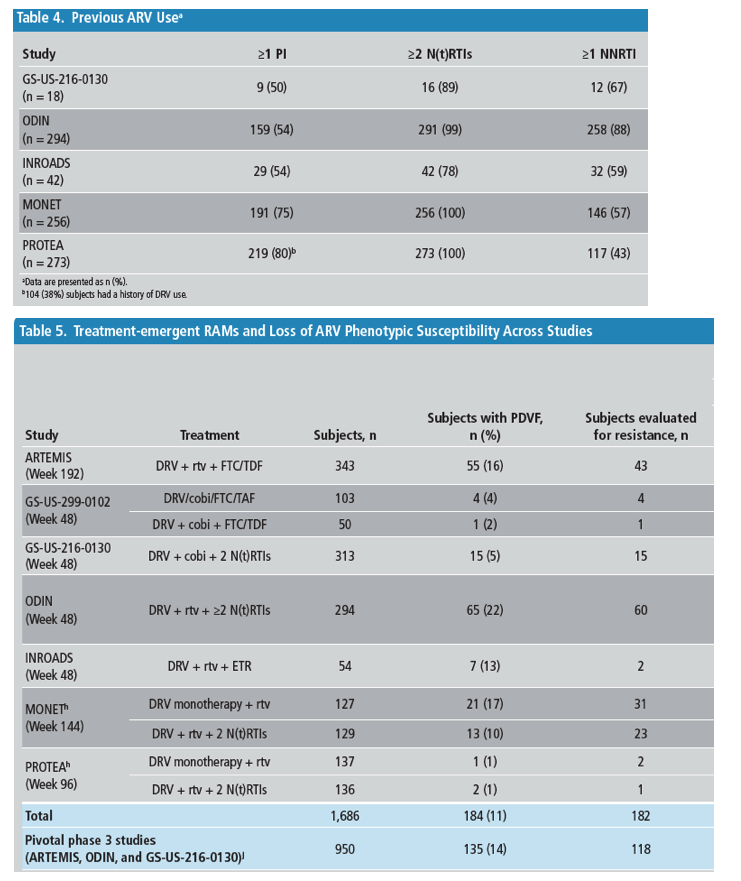
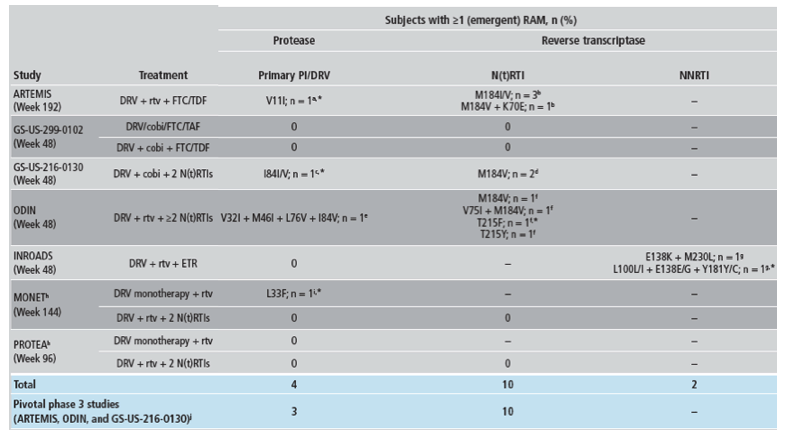
*Indicates no loss of phenotypic susceptibility for the corresponding ARVs.
aSubject developed V11I, a secondary PI RAM, after treatment stop (discontinuation due to noncompliance), with no loss of DRV or PI phenotypic susceptibility observed.
bFor all 4 subjects, there was a loss of FTC susceptibility.
cOne TE subject developed I84I/V, which was both a primary PI RAM and a DRV RAM; DRV and PI phenotypic susceptibility were not lost.
dBoth subjects (1 TN and 1 TE) used an FTC and TDF N(t)RTI backbone and lost susceptibility to FTC.
eOne subject (DRV QD arm) developed 4 primary PI RAMs, 3 of which were also DRV RAMs (V32I, L76V, and I84V); DRV, amprenavir, atazanavir, indinavir, and nelfinavir phenotypic susceptibility were lost, possibly related to previous VF with lopinavir + rtv treatment.
fFor 3 of these subjects, development of N(t)RTI RAMs was associated with loss of susceptibility to an N(t)RTI in the optimized background regimen; 2 subjects (1 who developed M184V and 1 who developed V75I + M184V) each lost susceptibility to abacavir and lamivudine, and 1 subject (who developed T215Y) lost susceptibility to zidovudine.
gThe subject who developed E138K + M230L lost susceptibility to ETR. The subject who developed L100L/I + E138E/G + Y181Y/C became partially susceptible to ETR.
hOn-treatment RAMs are reported for the 2 switch studies (MONET and PROTEA). One MONET subject (DRV + rtv +2 N[t]RTIs arm) who had detectable primary PI and N(t)RTI RAMs was excluded because these mutations were already present at the start of the study as a result of a previous VF.
iOne subject had L33F, both a primary PI RAM and a DRV RAM, detected at Week 12 with HIV-1 RNA of 63 copies/mL, but was resuppressed at subsequent visits until Week 144.
jCombined data from the 3 pivotal phase 3 studies of DRV QD.

|
| |
|
 |
 |
|
|