 |
 |
 |
| |
Big Gaps in HCV/HIV Care Continuum Across Europe at Start of 2015
|
| |
| |
16th European AIDS Conference, October 25-27, 2017. Milan
Mark Mascolini
In January 2015 more than 20% of HIV-infected people positive for anti-HCV antibody had no documented HIV RNA test, according to a large EuroSIDA analysis [1]. Among HCV RNA-positive people, fewer than half started HCV therapy.
The World Health Organization estimates that 2.3 million people worldwide are infected with both HCV and HIV. To identify gaps in testing, treatment, and cure of HIV-positive people in Europe, a EuroSIDA team mapped a continuum of care and checked records to determine proportions of people who completed each step from testing anti-HCV antibody positive through achieving sustained virologic suppression (SVR).
EuroSIDA is an ongoing observational cohort study following more than 22,000 people with HIV infection. Cohort members included in the HCV continuum analysis had to be in follow-up on January 1, 2015, anti-HCV positive before that date, and more than 16 years old. EuroSIDA divides its member countries into South, Central West, Central East, East, and North. The continuum of care has 8 steps (which did not include an estimate of undiagnosed people with HCV). The researchers calculated continuum falloff in two groups of stages, 1 to 3 and 4 to 8:
1. Anti-HCV positive
2. Ever HCV RNA tested
3. Currently HCV RNA positive
4. Ever HCV RNA positive
5. Ever received treatment for HCV
6. Treatment completed
7. Follow-up HCV RNA available
8. SVR12 (interferon-free regimens) or SVR24 (interferon-containing regimens)
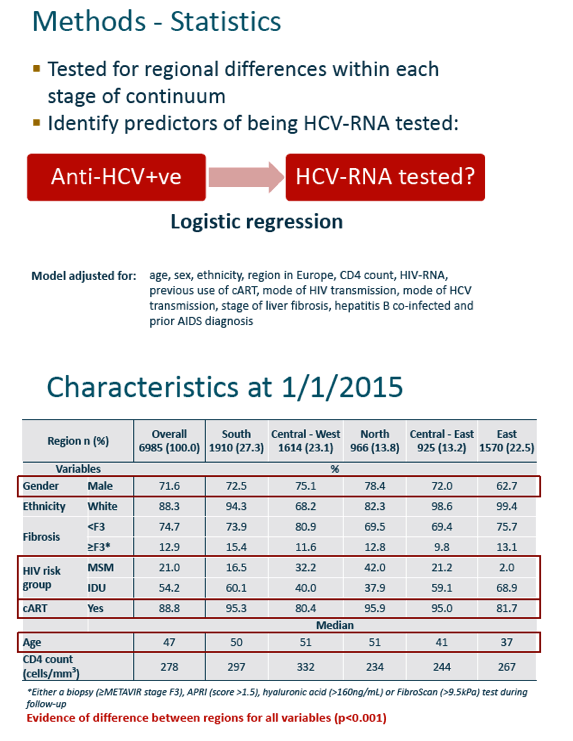
The investigators tested for regional differences within each stage of the continuum and used logistic regression to identify predictors of being tested for HCV RNA after testing anti-HCV-positive. The regression analysis adjusted for age, sex, ethnicity, region of Europe, CD4 count, HIV RNA, prior AIDS, and several liver-related factors.
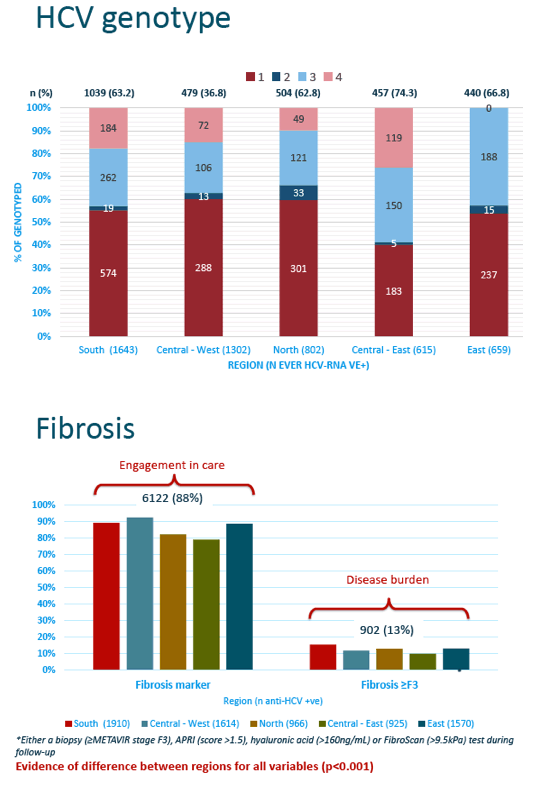
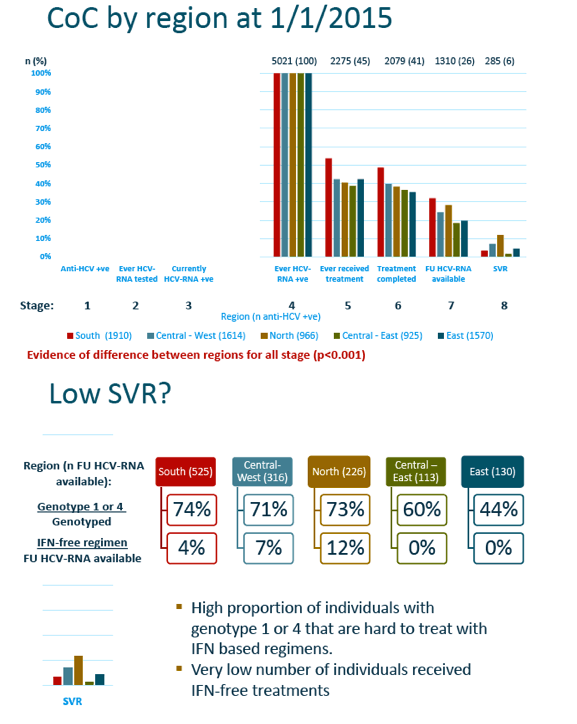
The analysis included 6985 EuroSIDA members, 72% of them men, 54% drug injectors, and 89% taking antiretroviral therapy. Median age stood at 47 and median CD4 count at 278. HCV genotype prevalence varied from region to region, with about half overall genotype 1, about one quarter genotype 3, and about 15% genotype 4. The researchers determined that 88% of participants across the 5 regions had a fibrosis marker recorded--evidence of engagement in care--but rates varied significantly from one region to the next (P < 0.001).
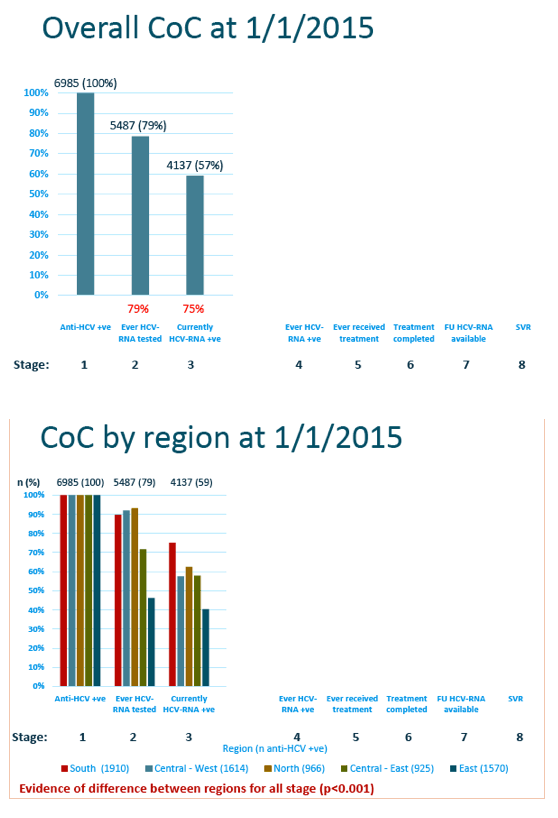
The EuroSIDA team charted substantial falloff across the first three stages of the continuum: Of 6985 people included as anti-HCV-positive, 5487 (79%) got tested for HCV RNA and 4137 (59%) currently tested positive for HCV RNA. Discontinuity between steps 1 and 2 and between 2 and 3 varied significantly between EuroSIDA regions (P < 0.001), with the worst continuity at each step recorded in the East (Russia, Belarus, Georgia, Ukraine, Baltic countries).
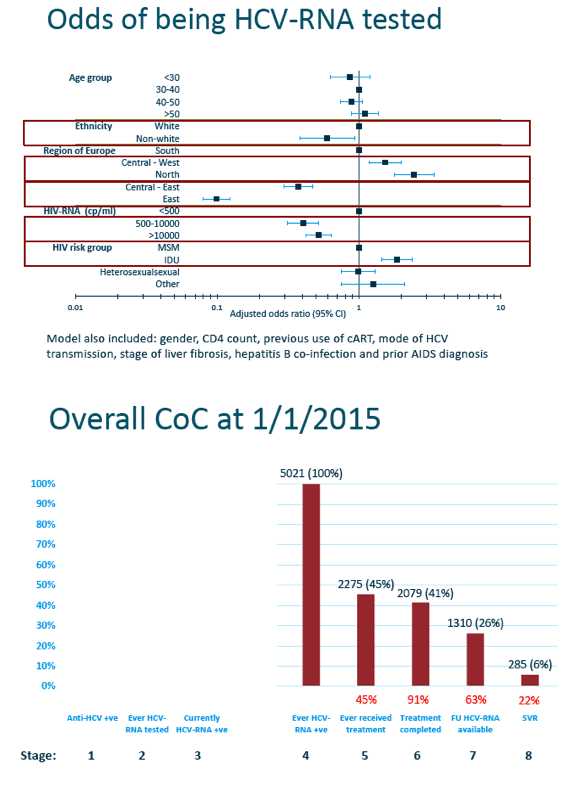
Factors independently associated with lower odds of getting an HCV RNA test were nonwhite ethnicity, Central East or East region (versus South), and HIV RNA 500 to 10,000 copies or over 10,000 copies (versus under 500). Drug injection as a transmission route (versus sex between men) independently raised odds of getting tested for HCV RNA. Age did not independently affect chances of HCV RNA testing.
Among 5021 people ever tested for HCV RNA, only 2275 (45%) ever received anti-HCV therapy, 2079 (41%) completed treatment, 1310 (26%) had a follow-up HCV RNA available, and 285 (6%) attained SVR. Again falloff at each step differed significantly between EuroSIDA regions, with the South having the best follow-through at each stage except the final step, SVR, where the North had the best result.
The low SVR rate reflects predominant use of interferon-based regimens at the study date, January 1, 2015. Interferon/ribavirin therapy lacked potency against HCV genotypes 1 and 4, which predominate in the EuroSIDA population. The researchers calculated that about 83% of regimens consisted of interferon plus ribavirin, about 8% interferon with a direct-acting antiviral (DAA), and about 9% DAAs without interferon.
The EuroSIDA team concluded that a "significant proportion of individuals [are] lost at all stage of our proposed HCV continuum." They suggested that the big gap between testing and starting therapy probably reflects several factors, including treatment access, lack of DAAs, and patient traits. The low SVR rate partly reflects study definitions, but also the low proportion who began and finished DAA therapy at the study date.
Reference
1. Amele S, Peters L, Lundgren JD, et al. The hepatitis C continuum of care among HIV infected Individuals in EuroSIDA. 16th European AIDS Conference. October 25-27, 2017. Milan. Abstract PS9/1.




|
| |
|
 |
 |
|
|