 |
 |
 |
| |
Phase 3 randomized controlled trial of switching to emtricitabine/tenofovir alafenamide (F/TAF) from abacavir/lamivudine (ABC/3TC) in virologically suppressed adults: week 48 results
|
| |
| |
Switching to TAF/FTC Noninferior to Staying With ABC/3TC in Double-Blind Trial
16th European AIDS Conference, October 25-27, 2017. Milan
Mark Mascolini
Switching to tenofovir alafenamide/emtricitabine (TAF/FTC) proved virologically noninferior to staying with abacavir/lamivudine (ABC/3TC), while keeping the same third agent, in a 48-week 556-person double-blind trial [1]. Kidney and bone markers were similar with the two regimens.
TAF/FTC is replacing tenofovir disoproxil fumarate/FTC in single-tablet coformulations and in prescriptions written. But the international team who conducted this study observed that no trial has directly compared TAF with the nucleoside ABC. To make that comparison, they conducted a phase 3 randomized, double-blind, double-dummy trial in the European Union and the United States.
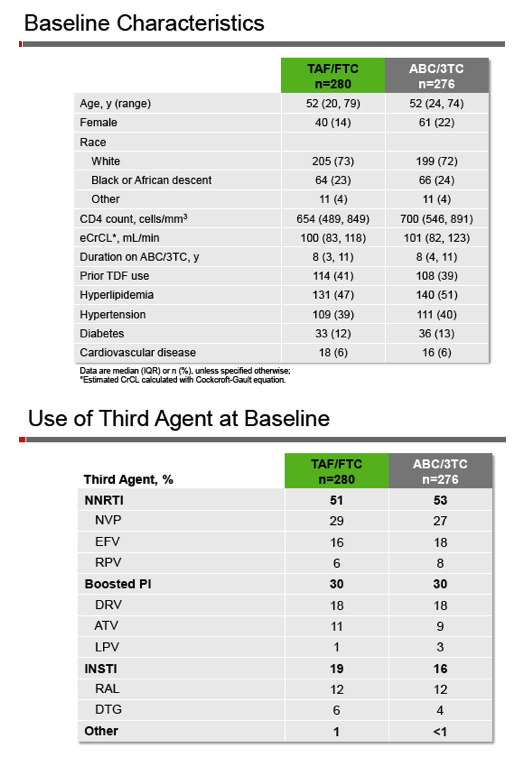
All participants had a viral load below 50 copies for at least 6 months on an ABC/3TC-containing regimen (excluding single-tablet regimens), and everyone had estimated creatinine clearance at or above 50 mL/min. Researchers randomized 276 people to continue ABC/3TC and the third antiretroviral and 280 to swap ABC/3TC for TAF/FTC while keeping the same third antiretroviral. The primary endpoint was proportion with a viral load below 50 copies at 48 weeks by snapshot analysis, with a 10% noninferiority margin.
Both groups had a median age of 52 years, about three quarters were white, and 14% assigned to TAF versus 22% assigned to TAF were women. Respective median CD4 counts were 654 and 700, and 41% and 39% had used TDF. Both groups had taken ABC/3TC for a median of 8 years. About half of both groups were taking a nonnucleoside with ABC/3TC, 30% a boosted protease inhibitor, and about 18% an integrase inhibitor.
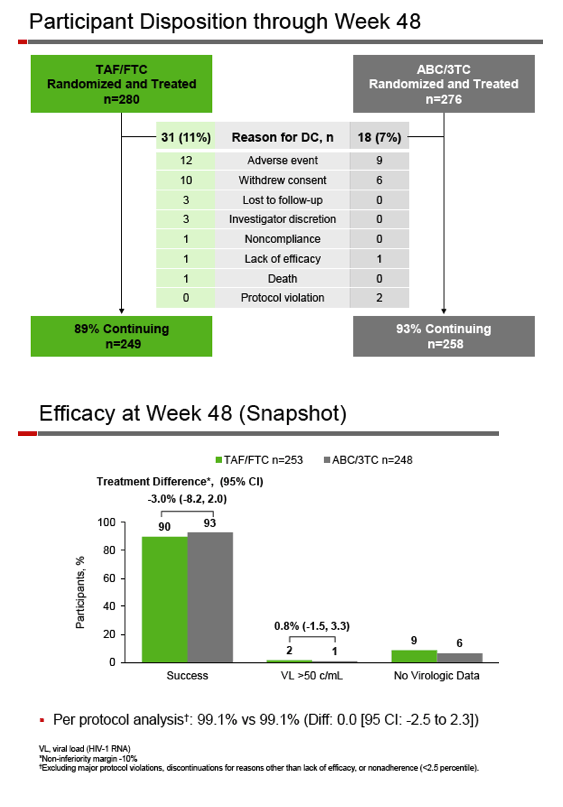
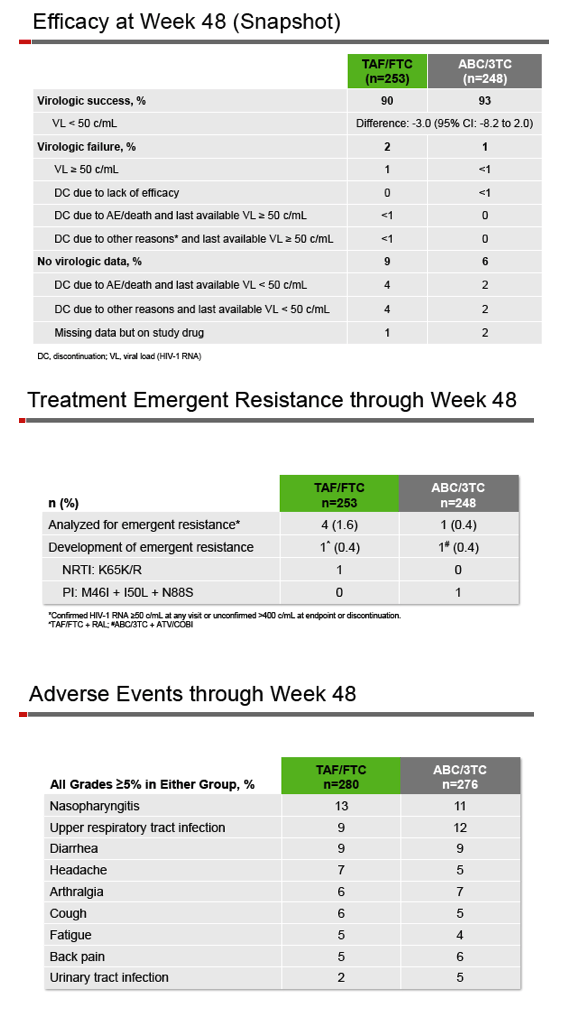
Among 280 people randomized to TAF, 249 (89%) continued treatment for 48 weeks, while 258 of 276 in the ABC arm (93%) made it to 48 weeks. At that point 90% in the TAF group and 93% in the ABC group maintained a viral load below 50 copies. The calculated difference and 95% confidence interval (-3.0%, -8.2 to 2.0) established the noninferiority of switching to TAF/FTC in a virally suppressed population. In a per-protocol analysis, 99% in both treatment arms kept their viral load below 50 copies. Virologic failure rates were 2% in the TAF group and 1% in the ABC group. A resistance analysis focused on 4 people with virologic failure in the TAF arm and 1 in the ABC arm. Resistant virus emerged with failure in 1 person in each group (K65K/R with the TAF regimen, protease inhibitor mutations with the ABC regimen).
Twelve people (4%) randomized to TAF and 9 (3%) randomized to ABC had adverse events leading them to stop study drugs. Drug-related serious adverse events arose in 0.7% taking TAF and 0.4% taking ABC. Grade 3 or 4 lab abnormalities appeared in 22% assigned to TAF and 18% assigned to continue ABC.
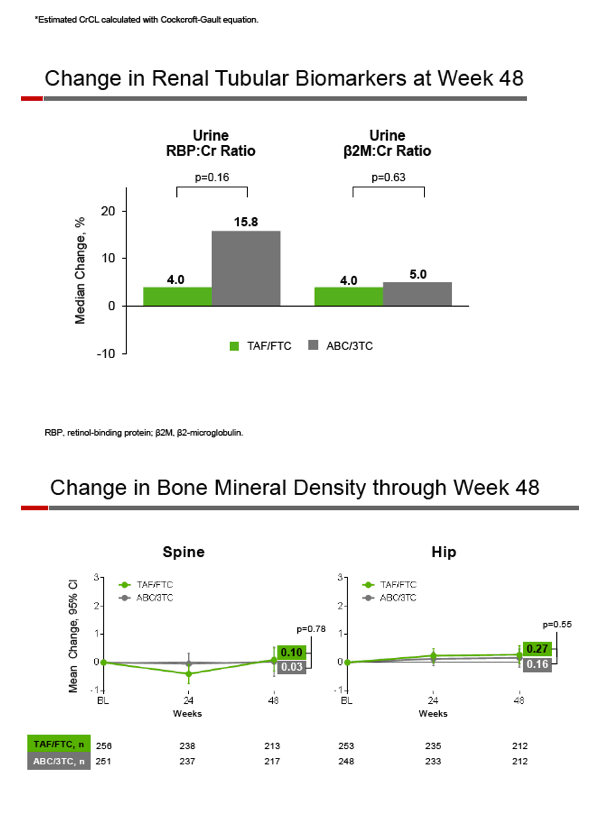
Through 48 weeks estimated creatinine clearance rose by a median of 1.3 mL/min with TAF/FTC while falling 1.1 mL/min with ABC/3TC (P = 0.05). Changes in renal tubular markers over 48 weeks did not differ significantly between the two groups. No cases of proximal renal tubulopathy or Fanconi syndrome developed in either group.
Spine and hip bone mineral density changed hardly at all in either group through 48 weeks. The only significant 48-week lipid change involved "good" high-density lipoprotein (HDL) cholesterol, which fell 2 mg/dL with TAF and rose 2 mg/dL with continued ABC (P < 0.001). Total-to-HDL cholesterol ratio did not change in either group, and similar proportions started lipid-lowering drugs (9% on TAF and 7% on ABC, P = 0.43).
The researchers proposed that "TAF/FTC provides an alternative backbone to ABC/3TC with similar effects on kidney and bone."
Reference
1. Winston A, Post FA, DeJesus E, et al. Phase 3 randomized controlled trial of switching to emtricitabine/tenofovir alafenamide (F/TAF) from abacavir/lamivudine (ABC/3TC) in virologically suppressed adults: week 48 results. 16th European AIDS Conference. October 25-27, 2017. Milan. Abstract PS8/4.




|
| |
|
 |
 |
|
|