 |
 |
 |
| |
How many HIV mono-infected or HBV or HCV co-infected patients with undetectable viremia
should be monitored for liver disease severity in the presence of suspect NAFLD?
|
| |
| |
....."13.8%....This study suggests the need for dedicated diagnostic hepatological monitoring of HIV mono and co-infected patients with undetectable viremia."
Reported by Jules Levin
Presented at EACS 2017 Oct 20-24, Milan and at EASL NASH Summit in Rome Nov 11 2017-12-07
Guaraldi G 1, Franconi I 1, Menozzi M 1, Malagoli A 1, Santoro A 1, Borghi V 1, Besutti G2, Schepis F 3, Lonardo A 4, Mussini C
1Modena HIV Metabolic Clinic (MHMC), University of Modena and Reggio Emilia, Modena, Italy 2.Department of diagnostic medicine. University of Modena and Reggio Emilia 3. Gastroenterology Department, University of Modena and Reggio Emilia, Modena, Italy 4. Metabolic Internal Medicine, University of Modena and Reggio Emilia, Modena, Italy
Program abstract
Introduction
HIV infection is associated with fatty liver as a result of host factors, HIV per se, or (HCV or HBV) co-infections, the so called "virus-associated fatty liver disease" (VAFLD) even in the absence of detectable HIV, HBV or HCV viremia.
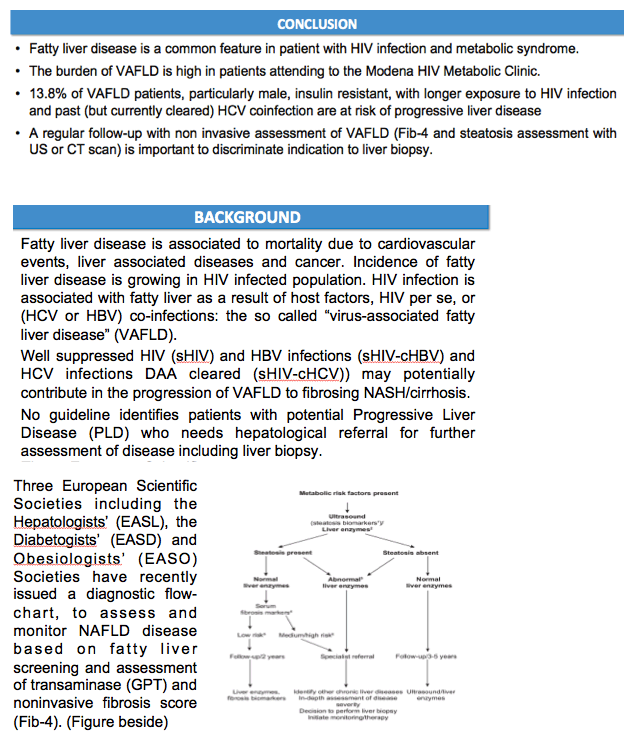
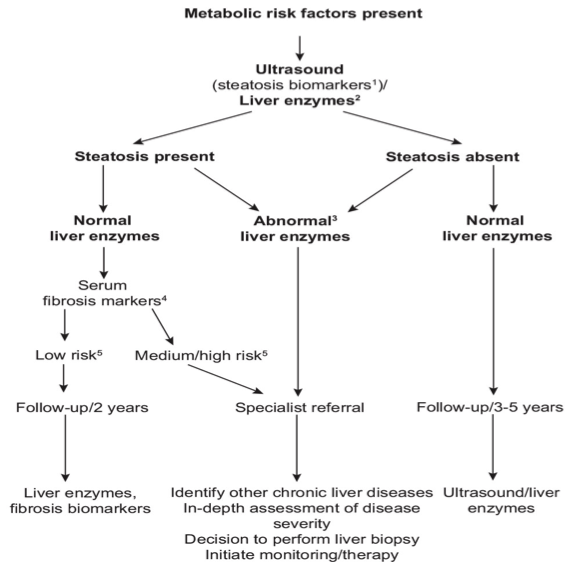
EASL have recently issued a diagnostic flow-chart, to assess and monitor NAFLD disease in virus-free patients based on fatty liver screening and assessment of transaminase (GPT) and non-invasive fibrosis score (Fib-4).
Aims
The objective of this study was to estimate and characterize HIV patients at risk for VAFLD using EASL- Clinical Practice Guidelines.
Methods
This cross-sectional study was conducted on patients attending the Modena HIV Metabolic Clinic.
Inclusion criteria were: patients with no high alcohol introduction (<2 alcohol unit/day) and HIV mono-infected or HBV/HIV or HCV/HIV co-infected (all with undetectable viremia). Exclusion criteria were any HIV-RNA, HBV-DNA or HCV-RNA detectability.
Fatty liver screening was offered per standard of care using abdominal CT (radiological criteria Liver/Spleen<1.1). Liver enzymes (GPT) cut-off for women was 19 IU, for men was 30 IU and Fib-4 cut-off was >2.64.
Results
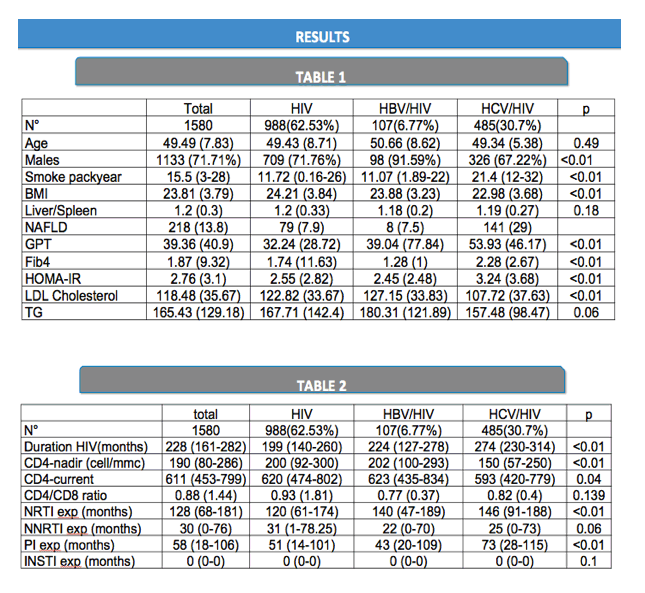
The whole population included 1580 patients (50 ±8 years, 28% females). Of them, 988 (62.5%) were HIV-monoinfected, 107 (6.8%) were HBV/HIV coinfected, and 485 (30.7%) were HCV/HIV coinfected.
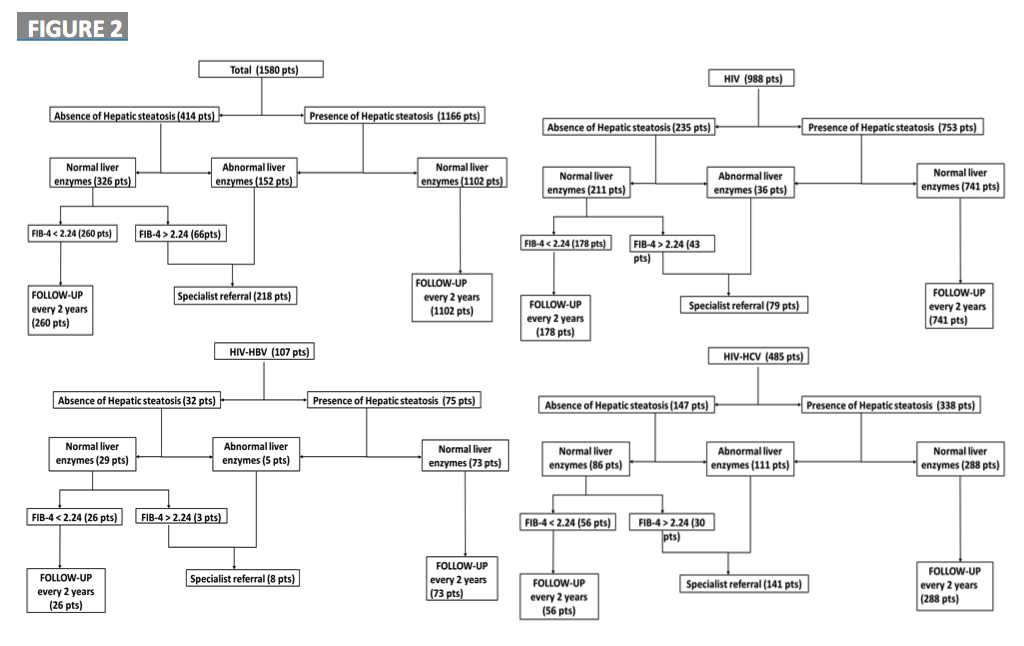
Figure 1 depicts EASL flowchart applied to all included patients (A), HIV monoinfected patients (B), HBV/HIV patients (C), and HCV/HIV patients (D). 13,8% of this HIV cohort (including 7% of HIV mono infected, 7.5% of HBV/HIV and 29% of HCV/HIV patients) are at risk of progressive liver disease and should be referred to hepatology consultation.
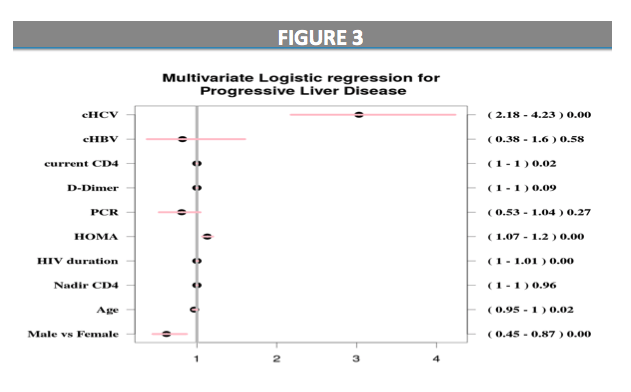
At logistic regression analyses, independent predictors for VAFLD were: male gender (OR=0.62, CI95%:0.46-0.83), duration of HIV infection per month (OR=1.01, CI95%:1.01-1.01, p<0.01), HOMA-IR (OR=1.13, CI95%1.07-1.2, p<0.01), and sHIV-cHCV group (OR=2.16 CI95%: 1.60-2.92 p<0.01), after correction for age, gender, CD4 nadir, current CD4, PCR, D-Dimer.
Conclusion
This study suggests the need for dedicated diagnostic hepatological monitoring of HIV mono and co-infected patients with undetectable viremia.

|
| |
|
 |
 |
|
|