 |
 |
 |
| |
WEIGHT GAIN AFTER SWITCH FROM EFAVIRENZ-BASED TO INTEGRASE INHIBITOR-BASED REGIMENS - Atripla-to-Integrase Inhibitor Switch Tied to Weight Gain Over 18 Months
|
| |
| |
Atripla-to-Integrase Inhibitor Switch Tied to Weight Gain Over 18 Months
IDWeek2017/IDSA, October 4-8, 2017, San Diego
Mark Mascolini
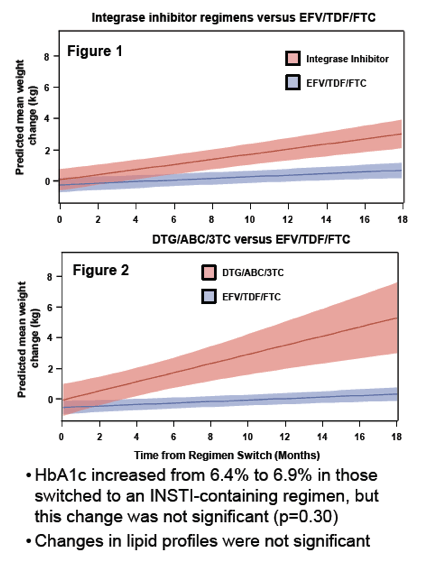
People who switched from single-tablet efavirenz/tenofovir/emtricitabine (Atripla) to an integrase inhibitor gained more weight through 18 months than people who stayed with Atripla or switched to a protease inhibitor (PI) [1]. Patients switching to single-tablet dolutegravir/abacavir/lamivudine (Triumeq) had the greatest weight gains.
Researchers at Nashville's Vanderbilt University conducted this study when local HIV providers noted weight gains in people swapping Atripla for Triumeq. The analysis involved three adult groups--those who switched to an integrase inhibitor (dolutegravir, raltegravir, or elvitegravir), those who switched to a PI, and those who stayed with Atripla. Everyone took Atripla for at least 2 years, always with a viral load below 1000 copies, and had at least 18 months of follow-up after the switch (or after a "sham switch" from Atripla). The researchers used linear mixed-effects models to assess average weight differences over time, after adjustment for baseline age, sex, race, CD4 count, and weight.
Study groups included 136 people who switched to an integrase inhibitor, 34 who switched to a PI, and 325 who continued Atripla. Respective proportions of women were 14%, 29%, and 14% and median weights stood at 82.5 kg, 75.2 kg, and 80.3 kg. Median age was about 39 across the three groups, and about 40% of the study group were nonwhite. All three groups had a median CD4 count above 500 at the switch.
People who switched to an integrase inhibitor gained more weight through 18 months than those who stayed with Atripla (average 2.9 versus 0.9 kg, P < 0.01 for integrase change, P = 0.003 for continued Atripla). Participants switching to a PI gained an average 0.7 kg (P = 0.81). People who switched to Triumeq gained more weight than those who switched to raltegravir or elvitegravir (5.3 versus 2.8 kg), but this difference lacked statistical significance (P = 0.19). Hemoglobin A1c, a diabetes marker, rose from 6.4% to 6.9% in people who traded Atripla for an integrase inhibitor. This change was nonsignificant (P = 0.30) but pushed the group across the 6.5% threshold indicating diabetes. Lipid changes were not significant.
The researchers concluded that swapping Atripla for an integrase inhibitor is associated with weight gain. They noted that studies of bigger cohorts are needed to figure whether cardiometabolic risk factors change with a switch to an integrase inhibitor. Notably, a recent study of 839 women and 1759 men with HIV in Italy linked integrase inhibitors and tenofovir disoproxil fumarate to greater lean mass loss [2].
References
1. Norwood J, Turner M, Bofill C, et al. Weight gain after switch from efavirenz-based to integrase inhibitor-based regimens. IDWeek2017/IDSA. October 4-8, 2017. San Diego. Abstract 1398.
2. Jordan Lake J, Erlandson K, Sim M, et al. Lean mass declines consistently over 10 years in HIV-infected adults on antiretroviral therapy, with patterns differing by sex. 8th International Workshop on HIV and Aging. October 2-3, 2017. New York. Abstract 10.
http://www.natap.org/2017/AGE/AGE_03.htm
Antiretroviral initiation is associated with increased skeletal muscle area and fat content - (08/14/17)


|
| |
|
 |
 |
|
|