 |
 |
 |
| |
Good In-Practice SVR12 With Sofosbuvir Regimens After HCV PI Failure in HIV/HCV Coinfected
|
| |
| |
IDWeek2017/IDSA, October 4-8, 2017, San Diego
Mark Mascolini
HCV/HIV-coinfected patients in whom an HCV protease inhibitor (PI) had failed had a 93% 12-week sustained virologic response (SVR12) rate with a sofosbuvir-containing regimen in a small single-center analysis [1]. The SVR12 rate in this study is similar to that attained with ledipasvir/sofosbuvir in the ION-4 trial in treatment-experienced HCV/HIV patients [2].
Despite high SVR rates with DAA regimens in HCV/HIV-coinfected people, a fraction of DAA regimens fail and pose a retreatment challenge. Clinical researchers from Albany Medical Center conducted this retrospective review to determine clinical traits and SVR12 rates in HCV/HIV-coinfected people retreated with a sofosbuvir-containing regimen after failure of an HCV NS3/4a PI. Sofosbuvir is a nucleotide analog inhibitor of NS5B polymerase available as a single agent (Sovaldi) or coformulated with the HCV NS5A inhibitor ledipasvir (Harvoni).
The study involved patients with HCV genotype 1 and HIV whose last regimen included an HCV PI and failed. All got retreated with a sofosbuvir-containing combination between January 2014 and December 2015. Everyone had an undetectable HIV load when HCV retreatment began, and all received care from a primary provider who could refer patients to a hepatologist and to a pharmacist for medication review.
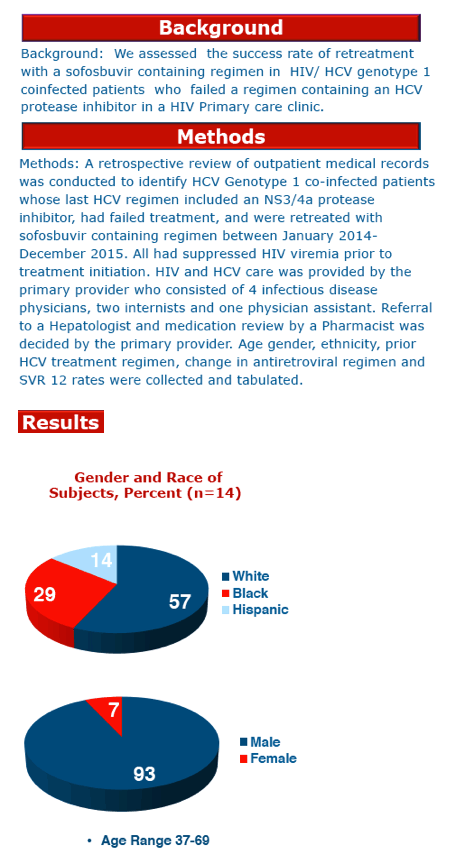
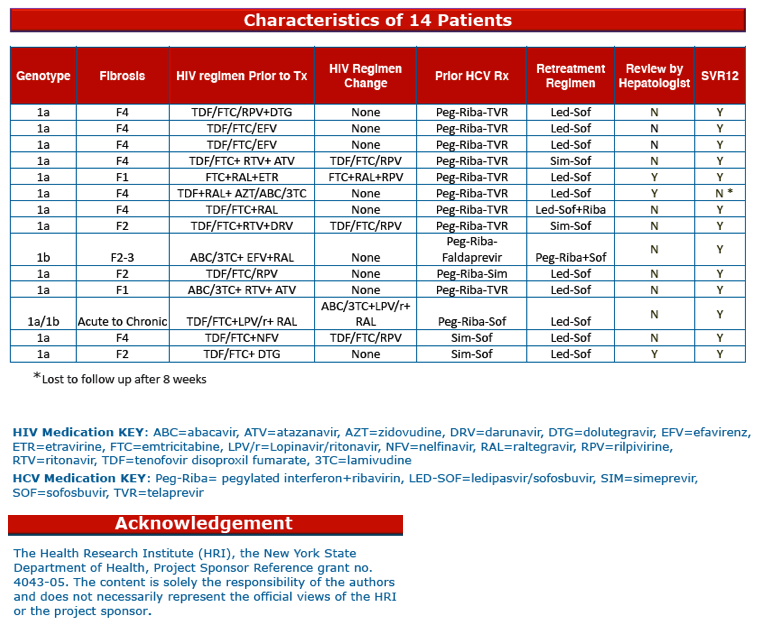
The analysis involved 14 people, 13 of them men, with ages ranging from 37 to 69. Eight people were white, 4 black, and 2 Hispanic. The failing regimen was telaprevir plus pegylated interferon (Peg-IFN)/ribavirin in 9, simeprevir/sofosbuvir in 2, and 1 each taking PegIFN/ribavirin/faldaprevir, PegIFN/ribavirin/simeprevir, or PegIFN/ribavirin/sofosbuvir. Seven of 14 people had F4 fibrosis, 1 was F2-F3, 3 were F2, 2 were F1, and 1 had an unknown fibrosis status.
Retreatment consisted of ledipasvir/sofosbuvir in 10, simeprevir/sofosbuvir in 2, ledipasvir/sofosbuvir/ribavirin in 1, and PegIFN/ribavirin/sofosbuvir in 1. Five people had to change their antiretrovirals when switching to the sofosbuvir regimen because of potential drug-drug interactions.
Thirteen people (93%) achieved SVR12 with retreatment and 1 was lost to follow-up after taking ledipasvir/sofosbuvir for 8 weeks. This response rate compares well with the SVR12 attained with ledipasvir/sofosbuvir in 185 previously treated HCV/HIV patients in the ION-4 trial, 96.8% [2]. Among 53 ION-4 participants who had already taken an HCV PI plus PegIFN/ribavirin, SVR12 with ledipasvir/sofosbuvir was 98.1%.
The Albany Medical Center researchers believe their findings "demonstrate that successful management of HIV/HCV coinfected patients can be done in a busy primary care HIV clinic using a multidisciplinary approach."
References
1. Gilroy SA, Choudhary M, Faragon J, Ells P, Kingsley Z. Treatment of HCV NS3/4a protease inhibitor experienced patients with sofosbuvir containing regimens in an outpatient clinic. IDWeek2017/IDSA. October 4-8, 2017. San Diego. Abstract 2228. https://idsa.confex.com/idsa/2017/webprogram/Handout/id5794/POSTER245_2228.pdf
2. Naggie S, Cooper C, Saag M, et al. Ledipasvir and sofosbuvir for HCV in patients coinfected with HIV-1. N Engl J Med. 2015;373:705-713. www.natap.org/2015/IAS/nejmoa1501315(1).pdf
--------------------------------
Treatment of HCV NS3/4a protease inhibitor experienced patients with sofosbuvir containing regimens in an Outpatient Clinic
Shelley A. Gilroy MD, Madhuchhanda Choudhary MD, John Faragon PharmD, Peter Ells MD, Zoe Kingsley RN, Joseph Cirabisi



|
| |
|
 |
 |
|
|