 |
 |
 |
| |
Glycemic Control & HbA1C Change After SVR
|
| |
| |
No HbA1c Change After SVR With DAAs, Regardless of HIV or Diabetes Status
Improvement in Glycemic Control of Type 2 Diabetes After Successful Treatment of Hepatitis C Virus - (09/13/17).....We identified 2,435 patients with diabetes who underwent interferon-free and ribavirin-free DAA-based antiviral treatment for HCV in the national Veterans Affairs health care system. Changes in average hemoglobin A1c (HbA1c) level and use of antidiabetic medications 1 year before and after antiviral treatment were compared between patients who achieved sustained virologic response (SVR) and those who did not.
IDWeek2017/IDSA, October 4-8, 2017, San Diego
Mark Mascolini
Sustained virologic response (SVR) with direct-acting antivirals (DAAs) for HCV infection had no impact on hemoglobin A1c--the diabetes marker--in a 251-person study, regardless of whether patients had HIV infection or diabetes [1]. HIV blunted improvements in transaminase levels after SVR.
HCV infection boosts the risk of diabetes, but research linked HCV clearance with interferon-based therapy to lower fasting glucose and improved insulin resistance and diabetes. Some prior work found decreased glucose and HbA1c with DAA therapy. With colleagues at other institutions, researchers at the National Institute of Allergy and Infectious Diseases conducted this study to see whether SVR with DAAs affects HbA1c and other values in people with or without HIV or diabetes.
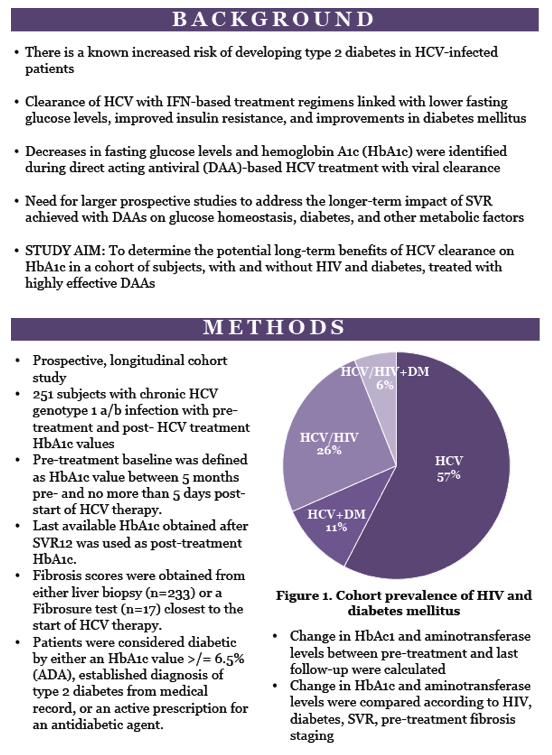
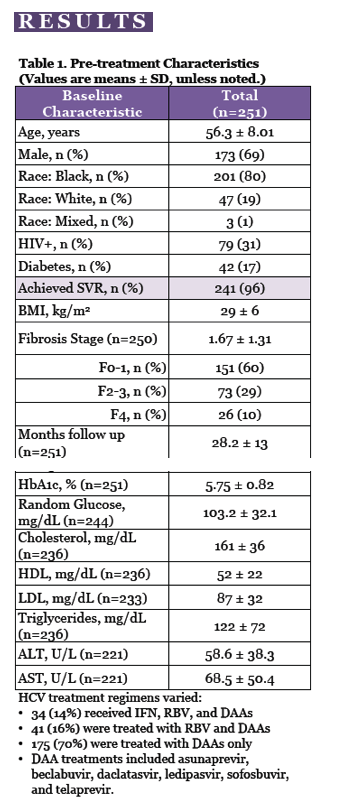
This longitudinal cohort study involved 251 patients with chronic HCV infection (genotype 1a/b) who had HbA1c measured within 5 months before DAA therapy and 5 days after treatment began. The researchers used the last HbA1c available after SVR12 as the posttreatment value. They considered a person diabetic if HbA1c stood at or above 6.5% or if a clinical diagnosis or active antidiabetic therapy could be established.
Of the 251 study participants, 57% had only HCV infection, 26% had HCV and HIV, 11% had HCV and diabetes, and 6% had HCV, HIV, and diabetes. Study group age averaged 56.3 years, 69% were men, 80% black, and 19% white. Most people, 70%, received only DAAs for their HCV, while 16% took DAAs plus ribavirin, and 14% took DAAs, ribavirin, and interferon. A large majority of these people--241 (96%)--achieved SVR.
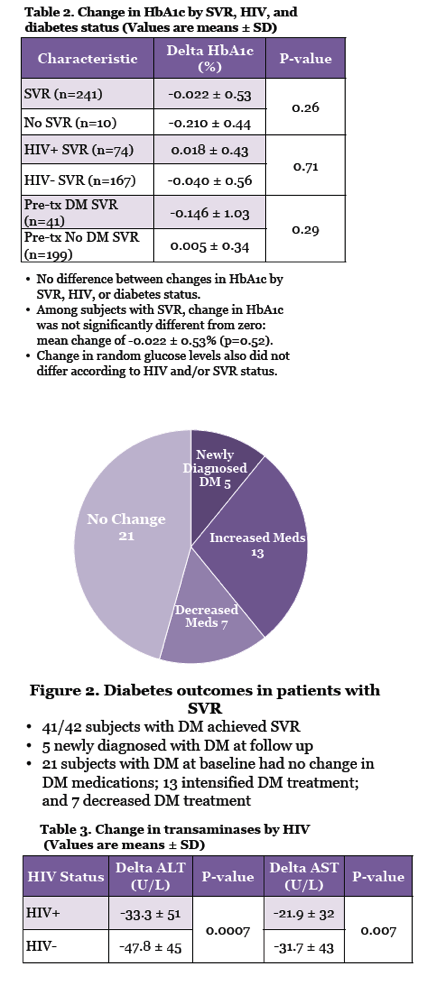
Baseline HbA1c averaged 5.75%. Through an average follow-up of 28.2 months, change in HbA1c did not did not differ significantly (1) with versus without SVR, (2) with SVR and with or without HIV, or (3) with SVR and with or without pretreatment diabetes. Among people with SVR, HbA1c change did not differ significantly from 0 (-0.022 +/- 0.53%, P = 0.52). Forty-one of 42 people with diabetes attained SVR, while 5 people got a new diabetes diagnosis during follow-up.
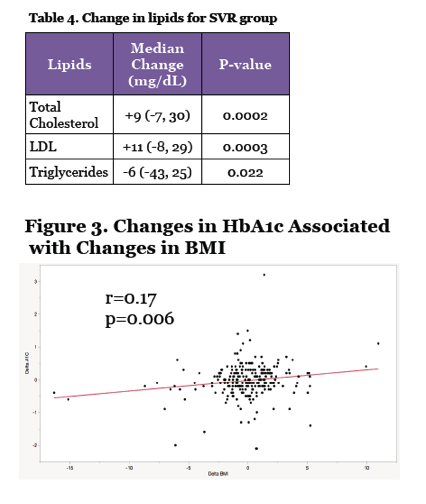
The liver enzyme alanine aminotransferase (ALT) fell 47.8 U/L during DAA therapy in people without HIV and 33.3 U/L in those with HIV, a significant difference (P = 0.0007). Aspartate aminotransferase also fell more during treatment in people without versus with HIV (31.7 versus 21.9 U/L, P = 0.007). People with SVR had significant jumps in median total cholesterol (+9 mg/dL, P = 0.0002) and "bad" low-density lipoprotein (LDL) cholesterol (+11 mg/dL, P = 0.0003), while their triglycerides dropped significantly (-6 mg/dL, P = 0.022).
The researchers noted that insulin resistance and glucose metabolism may improve over the short term during DAA therapy, "but our data suggest these effects are likely not durable or clinically significant." They called for larger, longer studies of DAA-treated populations with and without diabetes and HIV to get a better grasp on "long-term implications and health outcomes of HCV clearance."
Reference
1. Chaudhury C, Sheehan J, Chairez C, et al. Long-term evaluation of hemoglobin A1c following hepatitis C therapy in patients with and without HIV co-infection. IDWeek2017/IDSA. October 4-8, 2017. San Diego. Abstract 2224.
------------------------------------------------

Long-term Evaluation of Hemoglobin A1c following Hepatitis C Therapy in Patients with and without HIV Co-infection
Chloe Chaudhury1, Julia Sheehan1, Cheryl Chairez1, Elizabeth Akoth2, Chloe Gross2, Sarah Kattakuzhy2, Elana Rosenthal2, Shyam Kottilil2, Henry Masur3 and Colleen Hadigan1
1National Institute of Allergy and Infectious Diseases, Bethesda MD, 2Institute of Human Virology, Division of Infectious Diseases, University of Maryland, Baltimore MD, 3Critical Care Medicine Department, NIH Clinical Center, Bethesda, MD
|
| |
|
 |
 |
|
|