 |
 |
 |
| |
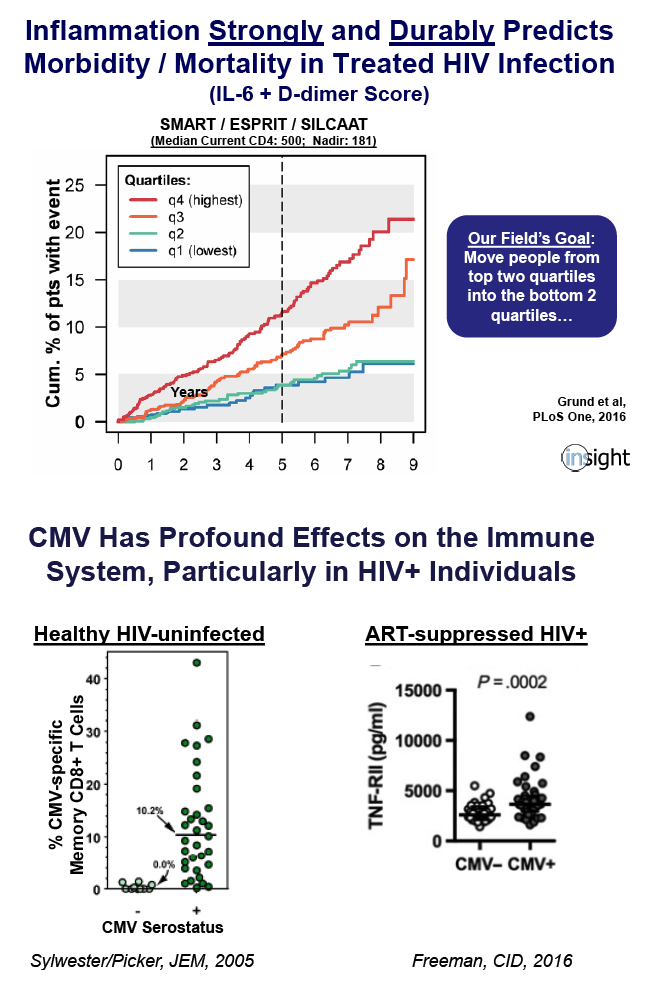
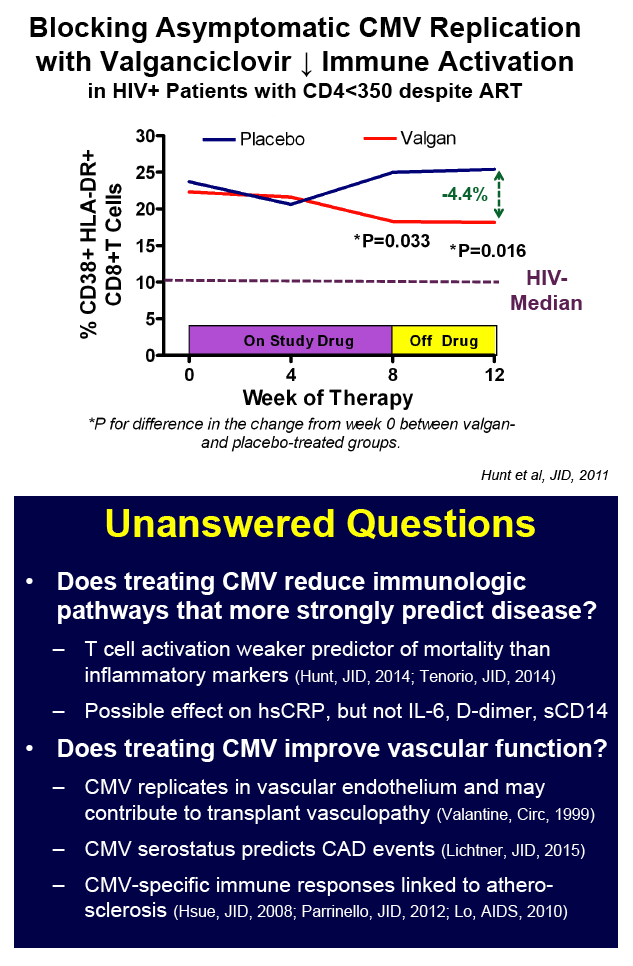
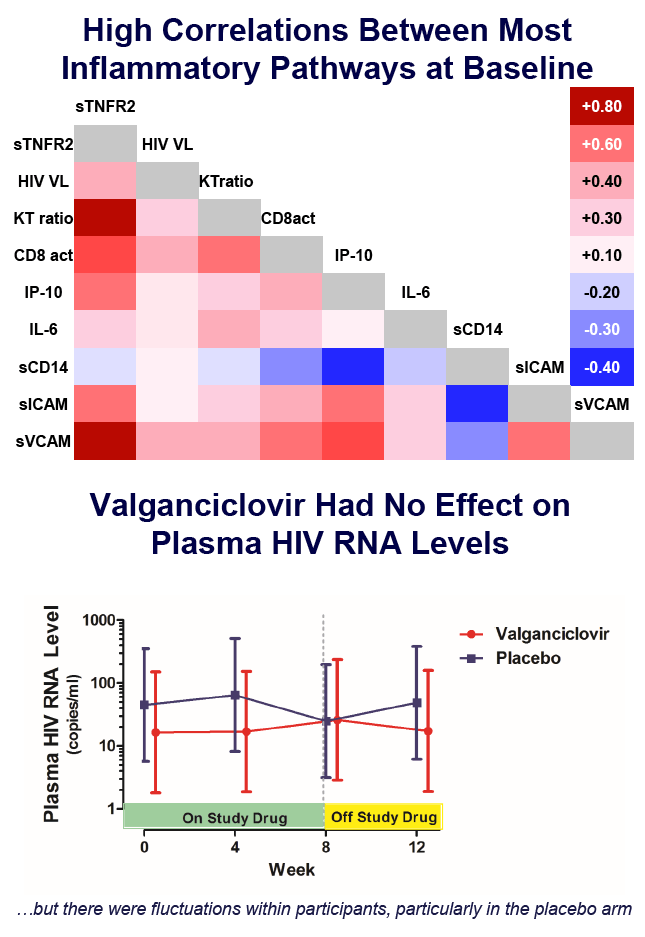
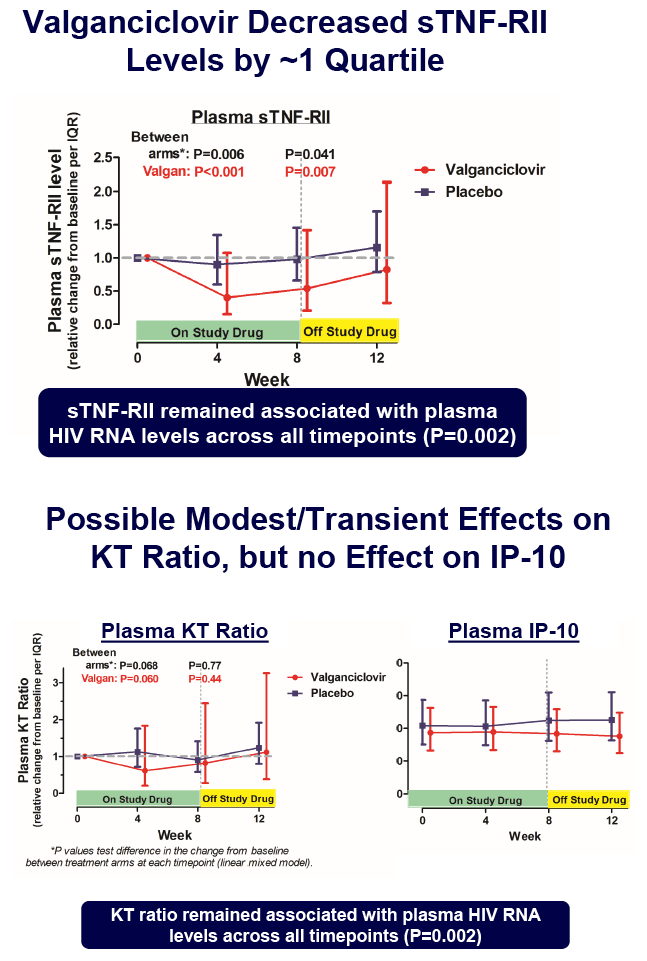
Valganciclovir Reduces an Immune Activation Pathway that Strongly Predicts Morbidity and Mortality in Treated HIV Infection
|
| |
| |
Gabriele Beck-Engeser, BS
Peter W. Hunt, MD
20th International Workshop on Comorbidities and Adverse Drug Reactions in HIV
Reported by Jules Levin
from Jules: its my understanding there is new CMV treatment called Letermovir that is safer than current CMV medications and thus justify its consideration as an anti-inflammatory as an anti CMV drug to reduced immune activation & inflammation as an anti-aging treatment in HIV, thus this study and future research......
https://en.wikipedia.org/wiki/Letermovir
Letermovir (INN; trade name Prevymis) is an antiviral drug for the treatment of cytomegalovirus (CMV) infections. It has been tested in CMV infected patients with allogeneic stem cell transplants and may also be useful for other patients with a compromised immune system such as those with organ transplants or HIV infections.[1] The drug was developed by Merck & Co., Inc as investigative compound MK-8228.[2]
The drug was granted fast track status by the US Food and Drug Administration (FDA) and orphan drug status by the European Medicines Agency.[1] In the United States, it is approved for prophylaxis of CMV infection and disease in adult CMV-seropositive recipients of an allogeneic hematopoietic stem cell transplant.[3]




|
| |
|
 |
 |
|
|