 |
 |
 |
| |
Switching tenofovir disoproxil fumarate to tenofovir alafenamide - not only bone and kidney
|
| |
| |
Reported by Jules Levin
20th International Workshop on Comorbidities and Adverse Drug Reactions in HIV Oct 13-14 2018 NYC
Florian Berger 1, Ana Milinkovic 2, Alejandro Arenas-Pinto3, 4, Stefan Mauss 1
1 Center for HIV and Hepatogastroenterology, Dusseldorf, Germany; 2 Chelsea and Westminster Hospital, London, United Kingdom; 3 Institute for Global Health, University College London, UK; 4 MRC Clinical Trials Unit at UCL, London, UK
program abstract
Switching tenofovir disoproxil fumarate to
tenofovir alafenamide not only bone and kidney
F Berger1, A Milinkovic2, A Arenas-Pinto3,4, S Mauss1
1Center for HIV and Hepatogastroenterology, Dusseldorf, Germany; 2Chelsea and Westminster Hospital, London, United Kingdom; 3Institute for Global Health, University College London, United Kingdom; 4MRC Clinical Trials Unit at UCL, London, United Kingdom
Introduction: Switching from tenofovir disoproxil fumarate (TDF) to tenofovir alafenamide (TAF) has been recommended as a strategy to reduce the risk of
renal and bone toxicities. However, its effect on lipid profile has not been a focus of interest.
Methods: This analysis consists of a retrospective data collection on effectively suppressed HIV-positive patients who were switched from TDF to TAF-based antiretroviral treatment (ARVT) due to medical reasons (bone, kidney disease) or as a result of optimization of therapy, in a single site (Center for HIV and Hepatogastroenterology, Dusseldorf). All components of ARVT for all patients were maintained with the single substitution of TDF to TAF. Only patients on stable lipid lowering therapy were included. Lipid profile was measured before switch and at 12 week intervals after initiation of TAF.
Results: A total of 385 virologically suppressed patients on stable TDF-containing regimens were included. Median age was 50 (IQR 42-56) years, 90% were male, 93% Caucasian, with a median BMI of 23.52 sqm/kg (IQR 21.26-25.61) at switching. Statin use was reported in 11% of patients and diabetes in 5%.
At switching, mean triglycerides were 126 mg/dl (IQR 87-191) and triglycerides >200 mg/dl were reported in 21% of patients, mean total cholesterol (TC) was 186 mg/dl (IQR 162-210) and 7% of patients had TC >240 mg/dl, mean LDL-cholesterol was 71 mg/dl (IQR 95-139).
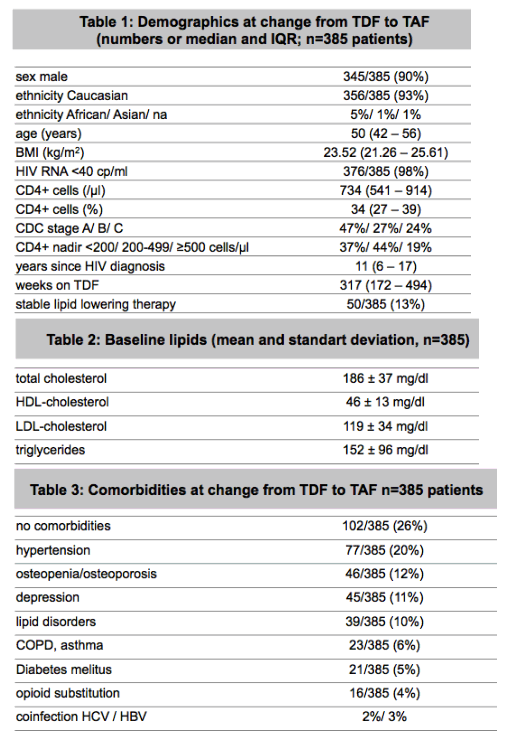
At 12 and 24 weeks, mean triglycerides had increased by +25.75 mg/dl (sd 100.54) and +13.09 mg/dl (sd 44.26), and TC had increased by +20.33 mg/dl (sd 26.38) and +13.09 mg/dl (sd 44.26) after switch to TAF, respectively, and differences in mean changes were significant (P<0.0001) for both parameters, mean ratio of TC/HDL-cholesterol showed a significant decrease 3 months after switching -2.31 (sd 13.35; P<0.01).
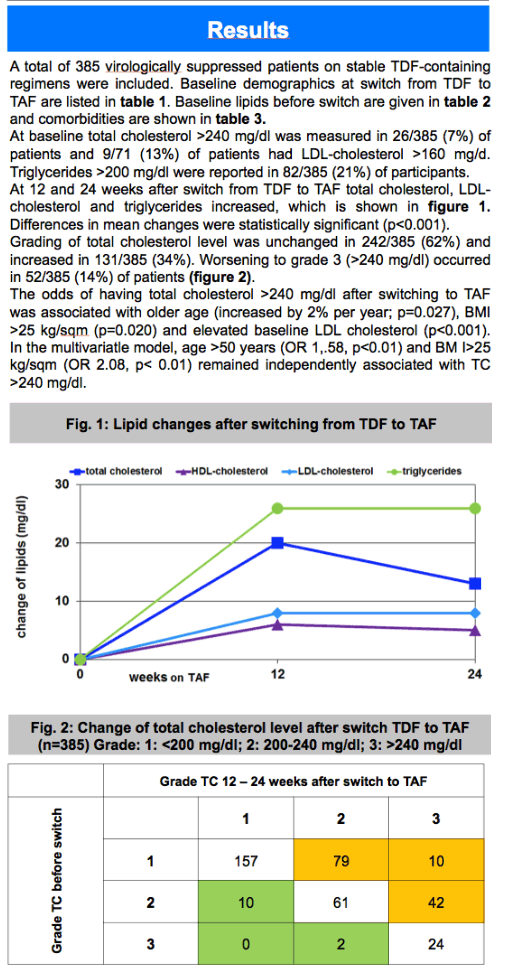
Regardless of baseline TC, switching to TAF increases mean TC (P<0.0001), the odds of having TC>240 mg/dl after switching was associated with older age (increased by 2% per year; P=0.027), BMI>25 kg/sqm (P=0.020) and elevated baseline LDL-cholesterol (P<0.001). In the multivariable model, age >50 and BMI>25 kg/sqm remained independently associated with TC>240 mg/dl (OR 1.58 and 2.08, respectively).
Conclusions: Results of our study show that patients with pre-existing higher CV risk are more likely to show increases in lipids after switching from TDF, which may worsen the cardiovascular risk profile. In these patients risk and benefit of switching TDF to TAF should be carefully accessed, taking lipids into consideration.


|
| |
|
 |
 |
|
|