 |
 |
 |
| |
No Evidence for Ongoing HIV-1 Replication
in Lymph Nodes During Suppressive ART
|
| |
| |
- No Sign of Ongoing HIV Replication in Lymph Nodes During ART
25th Conference on Retroviruses and Opportunistic Infections (CROI), March 4-7, 2018, Boston
Mark Mascolini
Exhaustive analysis of paired lymph node and blood cell samples in 5 people on suppressive antiretroviral therapy (ART) turned up no evidence of ongoing HIV replication in lymph nodes when viral load remains undetectable in plasma [1]. National Institutes of Health researchers and collaborators believe their findings suggest that "HIV infection is likely maintained by the proliferation of cells infected prior to ART, not by ongoing cycles of viral replication in either the peripheral blood or the lymph nodes."
Work over the past few years yielded conflicting findings on whether HIV continues to replicate in sanctuary sites when viral load remains below 50 copies in blood during ART [2,3]. Sometimes analysis of the same data produced divergent conclusions [3,4].
Addressing this controversy, Mary Kearney and collaborators conducted a new study to determine whether HIV continues to replicate in lymph nodes during suppressive ART.
The investigation had three parts: (1) analysis of HIV proviral genetics before ART and after 2 to 13 years of suppressive ART in paired lymph node mononuclear cell (LNMC) and peripheral blood mononuclear cells (PBMC) samples, (2) analysis of HIV integration sites in paired lymph node and blood samples, and (3) analysis of HIV RNA expression levels in single cells in paired samples. The investigators characterized proviral populations and expression by cell-associated RNA- and DNA- single-genome sequencing of p6-PR-RT. They compared proviral sequences phylogenetically and by testing for panmixia. They identified infected cell clones by integration site assay in PBMCs and LNMCs from one donor.
The analysis involved LNMC and PBMC samples from 5 people with continuous viral suppression during ART ranging from 1.8 to 12.9 years. All 5 participants come from the SCOPE cohort, "a prospective study of HIV-1 infected volunteers designed to provide a specimen bank of samples with carefully characterized clinical data" [5]. HIV DNA could be detected in all PBMC samples at levels ranging from 14 to 3226 copies per million cells. One person had fewer than 8 HIV DNA copies per million cells in LNMC, while LNMC levels ranged from 423 to 3637 copies per million cells in the other 4.
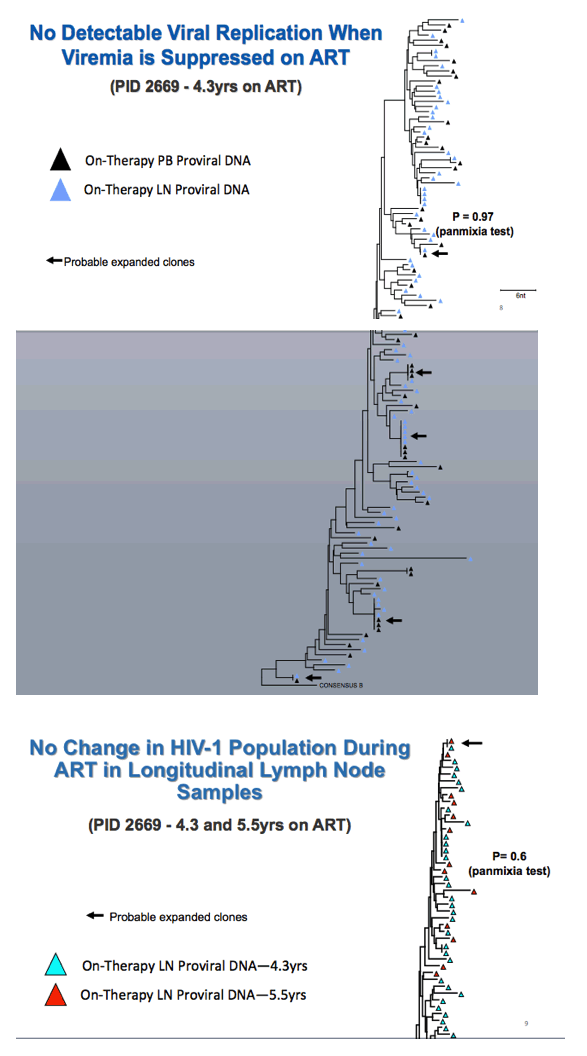
Phylogenetic analysis of pre-ART proviral DNA in PBMCs and on-treatment proviral DNA in PBMCs and LNMCs showed no detectable HIV replication when viral load is undetectable at those sites. Comparison of on-therapy proviral DNA in PBMCs and LNMC disclosed (1) no compartmentalization between peripheral blood and lymph nodes and (2) no detectable viral replication during HIV suppression in peripheral blood.
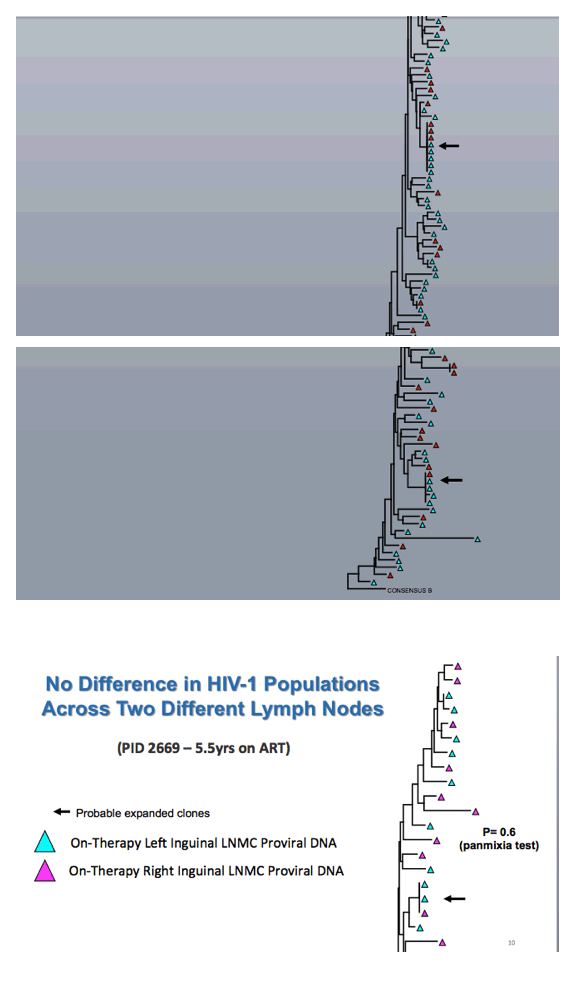
Comparing on-therapy proviral DNA in LNMCs after 4.3 and 5.5 years of ART in one person, the researchers detected no change in the HIV population over time. Comparing on-therapy proviral DNA in LNMCs from the left and right inguinal lymph nodes of a single person revealed no difference in HIV populations at these different sites.
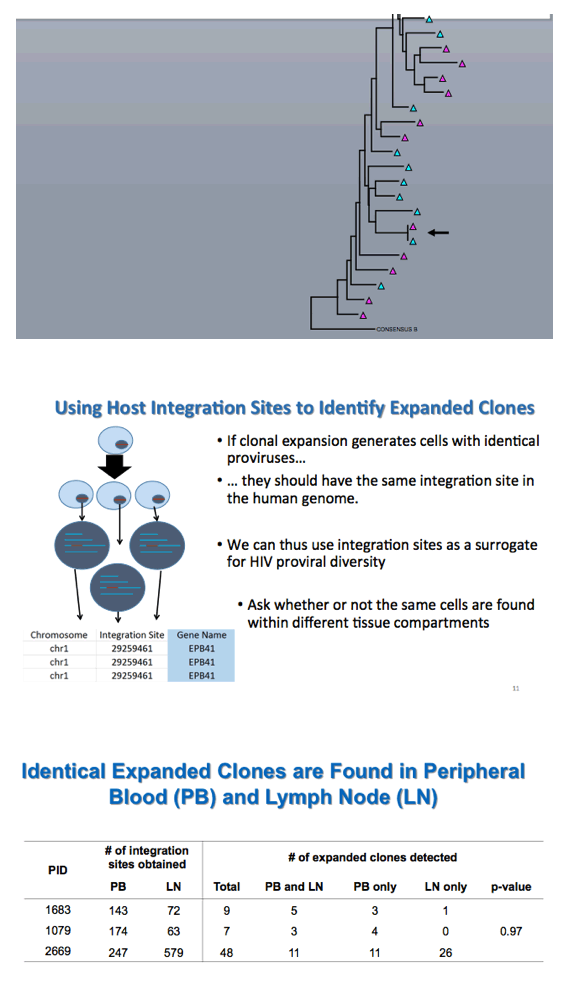
Kearney explained that "if clonal expansion generates cells with identical proviruses, they should have the same integration site in the human genome." Thus integration site analysis offers a surrogate for HIV proviral diversity. This experiment confirmed identical expanded viral clones in peripheral blood and lymph nodes. Further analysis determined that the same fraction of infected cells had unspliced HIV RNA in lymph nodes and peripheral blood.
The investigators concluded that no evidence indicates ongoing HIV replication in lymph nodes when ART controls HIV replication in peripheral blood. They determined that infected cells are well mixed between blood and lymph nodes, a finding that argues against compartmentalization. The collaborators proposed that eradicating HIV will depend on eliminating cells infected before ART begins, not on developing antiretrovirals with better lymph node penetration.
References
1. McManus WR, Bale MJ, Spindler J, et al. No evidence for ongoing HIV replication in lymph nodes during suppressive ART. 25th Conference on Retroviruses and Opportunistic Infections (CROI). March 4-7, 2018. Boston. Abstract 70.
2. Kearney MF, Spindler J, Shao W, et al. Lack of detectable HIV-1 molecular evolution during suppressive antiretroviral therapy. PLoS Pathog. 2014;10:e1004010.
3. Lorenzo-Redondo R, Fryer HR, Bedford T, et al. Persistent HIV-1 replication maintains the tissue reservoir during therapy. Nature. 2016;530:51-56.
4. Rosenbloom DIS, Hill AL, Laskey SB, Siliciano RF. Re-evaluating evolution in the HIV reservoir. Nature. 2017;551:E6-E9.
5. ClinicalTrials.gov. SCOPE: Observational Study of the Consequences of the Protease Inhibitor Era. ClinicalTrials.gov identifier NCT00187512.
WEBCAST: http://www.croiwebcasts.org/console/player/37166?mediaType=slideVideo&&crd_fl=1&ssmsrq=1521305553234&ctms=5000&csmsrq=904
CROI-2018: Integrase Inhibitor Penetration Generally Worse in Lymphoid Tissue Than Blood Cells- (03/06/18)
Persistent HIV-1 replication maintains
the tissue reservoir during therapy......http://www.natap.org/2016/HIV/nature16933.pdf
Ramon Lorenzo-Redondo1*, Helen R. Fryer2*, Trevor Bedford3, Eun-Young Kim1, John Archer4, Sergei L. Kosakovsky Pond5, Yoon-Seok Chung6, Sudhir Penugonda1, Jeffrey G. Chipman7, Courtney V. Fletcher8, Timothy W. Schacker9, Michael H. Malim10, Andrew Rambaut11, Ashley T. Haase12, Angela R. McLean2 & Steven M. Wolinsky1 - HIV-1 DNA in cells from blood and inguinal lymph nodes collected from three subjects at three separate times (at day 0, and after
3 and 6 months of treatment), described elsewhere13. Previous work established that viral sequences contemporaneously sampled from lymphoid tissue in different locations are genetically homogeneous17, consistent with CD4+ T cell homing and trafficking18. Consequently, detailed assessment of a portion of a solitary lymph node is no more susceptible to bias than wider anatomical sampling. We also sequenced viral RNA in the plasma (day 0) from these three study subjects. Two subjects (1727 and 1679) had well-suppressed infections (<48 viral genome copies per ml of plasma); and the third subject (1774) continued to have measureable amounts of viral RNA in plasma after 3, but not 6, months of treatment (Extended Data Fig. 1). Subjects 1727 and 1679 were each infected with HIV-1 for approximately 3 to 4 months and were antiretroviral-drug-naive before the study. Subject 1774 was infected with HIV-1 for approximately 17 years and was antiretroviral therapy-experienced, but had not received any treatment for at least
1 year before the study.
- Lymphoid tissue is a key reservoir established by HIV-1 during acute infection. It is a site associated with viral production, storage of viral particles in immune complexes, and viral persistence. Although combinations of antiretroviral drugs usually suppress viral replication and reduce viral RNA to undetectable levels in blood, it is unclear whether treatment fully suppresses viral replication in lymphoid tissue reservoirs. Here we show that virus evolution and trafficking between tissue compartments continues in patients with undetectable levels of virus in their bloodstream. We present a spatial and dynamic model of persistent viral replication and spread that indicates why the development of drug resistance is not a foregone conclusion under conditions in which drug concentrations are insufficient to completely block virus replication. These data provide new insights into the evolutionary and infection dynamics of the virus population within the host, revealing that HIV-1 can continue to replicate and replenish the viral reservoir despite potent antiretroviral therapy.
Persistent HIV-1 replication is associated with lower
antiretroviral drug concentrations in lymphatic tissues.....http://www.natap.org/2014/HIV/PNAS-2014-Fletcher-1318249111 copy.pdf
Courtney V. Fletchera, Kathryn Staskusb,1, Stephen W. Wietgrefeb, Meghan Rothenbergerc, Cavan Reillyd, Jeffrey G. Chipmane, Greg J. Beilmane, Alexander Khorutsc, Ann Thorkelsonc, Thomas E. Schmidtc, Jodi Andersonc, Katherine Perkeyb, Mario Stevensonf, Alan S. Perelsong, Daniel C. Douekh, Ashley T. Haaseb, and Timothy W. Schackerc,2 - We prospectively treated 12 subjects with ARVs and performed multiple samplings of LN, ileum and rectum, and PB after initiating ART to determine intracellular (IC) concentrations of the ARVs in these tissues and to assess the impact of treatment on virus production, measured by reduced numbers of productively infected HIV-1–RNA+ cells and HIV-1 RNA in virions associated with the FDCn. Ten of the subjects were naïve to ART, and two subjects had been previously treated but had been off therapy for >1 y. In all subjects, commercial genotyping assays confirmed that the virus isolated from their plasma was sensitive to the planned ART. Subjects received tenofovir disoproxil fumarate (TDF)/emtricitabine (FTC) in combination with efavirenz (EFV; n = 6), atazanavir (ATV)/ritonavir (RTV) (n = 4), and darunavir (DRV)/RTV (n = 2). Subjects were followed for 6 mo with LN, ileum, and rectal biopsies obtained just before initiation of ART (month 0; M0), and again at months 1, 3, and 6 (M1, M3, and M6). PB was obtained at monthly intervals and a 24-h pharmacokinetic study in PB was done at M3.
- Antiretroviral therapy can reduce HIV-1 to undetectable levels in peripheral blood, but the effectiveness of treatment in suppressing replication in lymphoid tissue reservoirs has not been determined. Here we show in lymph node samples obtained before and during 6 mo of treatment that the tissue concentrations of five
of the most frequently used antiretroviral drugs are much lower than in peripheral blood. These lower concentrations correlated with continued virus replication measured by the slower decay or increases in the follicular dendritic cell network pool of visions and with detection of viral RNA in productively infected cells. The evidence of persistent replication associated with apparently suboptimal drug concentrations argues for development and evaluation of novel therapeutic strategies that will fully suppress viral replication in lymphatic tissues. These strategies could avert the long-term clinical consequences of chronic immune activation driven directly or indirectly by low-level viral replication to thereby improve immune reconstitution.




|
| |
|
 |
 |
|
|