 |
 |
 |
| |
Insurer Denial of DAA Prescription Requests Rising Across United States
|
| |
| |
25th Conference on Retroviruses and Opportunistic Infections (CROI), March 4-7, 2018, Boston
Mark Mascolini
Contrary to the perception that insurers have loosened the reins on approving prescriptions for anti-HCV direct-acting antivirals (DAAs), a nationwide US study traced an increase in absolute prescription denials from January 2016 through August 2017 [1]. Overall, insurers denied more than one third of all DAA prescription requests, and the rate was higher for commercial insurers than for Medicaid or Medicare.
High cost of DAAs initially led US insurers to restrict access to the curative drugs based on liver disease severity, specialist consultation, or substance use abstinence. Researchers in Ohio, Michigan, and Pennsylvania noted that prior studies of insurance acceptance of DAA prescriptions did not represent a national sample and were done early in the DAA era. They conducted this countrywide analysis to see if insurer acceptance of DAA prescriptions has changed, given the availability of more DAAs, a perceived relaxation of prescription OKs, and a public health focus on HCV elimination through DAA therapy.
This prospective analysis used data from Diplomat Pharmacy, a national specialty pharmacy, examining DAA prescription submissions from January 2016 through April 2017. The researchers assessed prescription acceptance or absolute denial through August 2017. Absolute denial meant an insurer denied any DAA prescription; insurer requests for alternate regimens because of formulary restrictions did not count as denials.
The analysis included 9025 DAA prescriptions, 4702 (52%) covered by Medicaid, 1821 (20%) by Medicare, and 2502 (28%) by commercial insurers. The overall absolute denial rate stood at 36% for the whole study period. Absolute denials proved more frequent with commercial insurers (52.4%) than with Medicaid (34.5%) or Medicare (14.7%).
In the six calendar quarters covered by the study, the overall absolute prescription denial rate began at 36% in the first quarter, fell to 28% in the second and third quarters, then rose to 36%, 42%, and 44% in the last three quarters. Commercial insurers had the highest proportion of denials across all six quarters: 52%, 35%, 38%, 59%, 59%, and 64%. Medicare made the fewest denials: 15%, 10%, 8%, 14%, 20%, and 23%. Denial rates for Medicaid were 35%, 31%, 31%, 32%, 39%, and 41%.
The researchers call for concerned parties to address this lack of DAA access if they hope to eliminate HCV. They believe the impact of liver fibrosis stage, substance use, and type of prescriber on DAA prescription denial needs more study.
Reference
1. Gowda C, Lott S, Grigorian M, et al. Increasing incidence of denial of DAA therapy for chronic HCV by insurance type. 25th Conference on Retroviruses and Opportunistic Infections (CROI). March 4-7, 2018. Boston. Abstract 600.
-----------------------------------------------
INCREASING INCIDENCE OF DENIAL OF DAA THERAPY FOR CHRONIC HCV BY INSURANCE TYPE
C. Gowda1,2; S. Lott3, M. Grigorian3, D.M. Carbonari4; M.E. Saine4; S. Trooskin5,6; J.A. Roy4; J.R. Kostman5,6; P. Ulrick3; V. Lo Re III2,4,6
1Department of Pediatrics, The Ohio State College of Medicine, Columbus, OH, USA; 2Department of Medicine, Perelman School of Medicine, University of Pennsylvania, Philadelphia, PA, USA; 3EnvoyHealth, Flint, MI, USA; 4Department of Biostatistics, Epidemiology, and Informatics, Center for Clinical Epidemiology and Biostatistics, Center for Pharmacoepidemiology Research and Training, Perelman School of Medicine, University of Pennsylvania, Philadelphia, PA, USA; 5Philadelphia Field Initiating Group for HIV Trials, Philadelphia, PA, USA; 6Penn Center for AIDS Research, Perelman School of Medicine, University of Pennsylvania, Philadelphia, PA, USA


On January 1, 2018, PA state Medicaid removed all restrictions to DAA reimbursement based on liver fibrosis stage, due largely to advocacy by Stacey Trooskin. However, prior to this date, PA Medicaid had requirement of stage 2 and then stage 1 during the period studied.
Since January 2018, in Philadelphia, we have seen much fewer denials. However, one of the strengths of this study is that it provides a national sample (45 states).
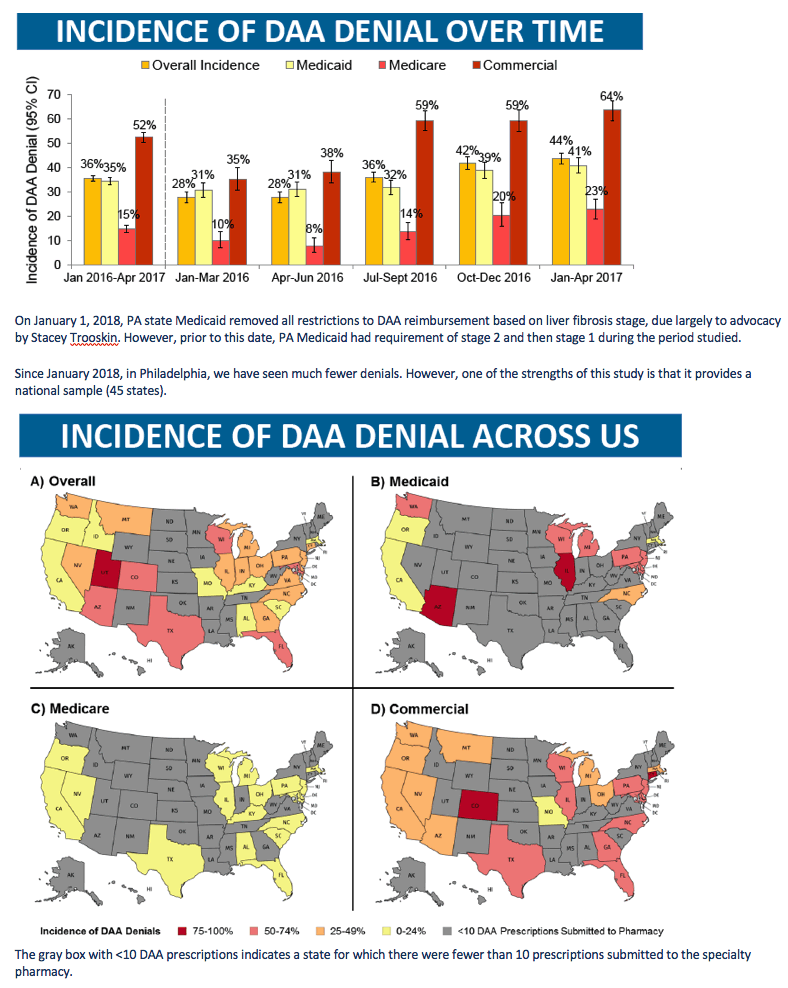
The gray box with <10 DAA prescriptions indicates a state for which there were fewer than 10 prescriptions submitted to the specialty pharmacy.
|
| |
|
 |
 |
|
|