 |
 |
 |
| |
Low-Dose Oral MK-8591 Protects Monkeys From Rectal SHIV Infection
|
| |
| |
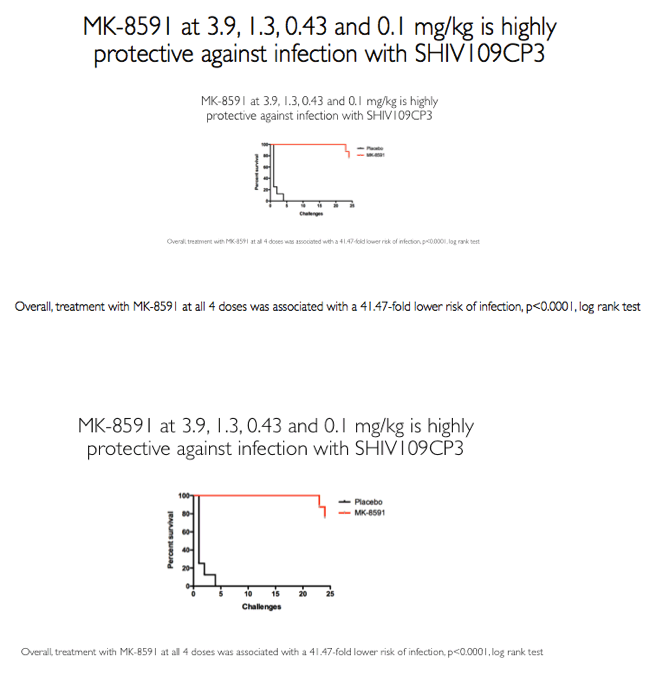
"The researchers concluded that MK-8591 remains completely protective against rectal SHIV at doses of 1.3 and 0.43 mg/kg weekly..."suggesting MK-8591 utility in extended-duration prophylaxis against HIV infection.....strong antiviral activity with weekly oral dosing"
25th Conference on Retroviruses and Opportunistic Infections (CROI), March 4-7, 2018, Boston
Mark Mascolini
At an oral dose of 1.30 or 0.43 mg/kg weekly, the nucleoside reverse transcriptase inhibitor (NRTI) MK-8591 protected 8 of 8 macaques from simian HIV (SHIV) infection after rectal exposure [1]. MK-8591 levels reached in this monkey study should be attainable in humans at doses under 250 ug weekly.
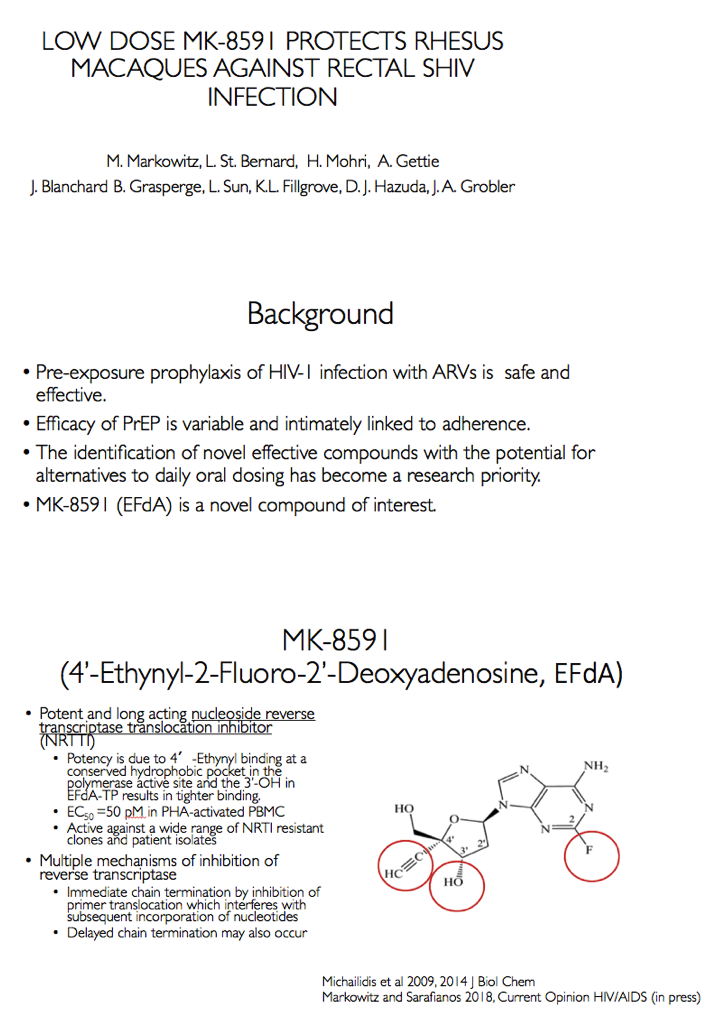
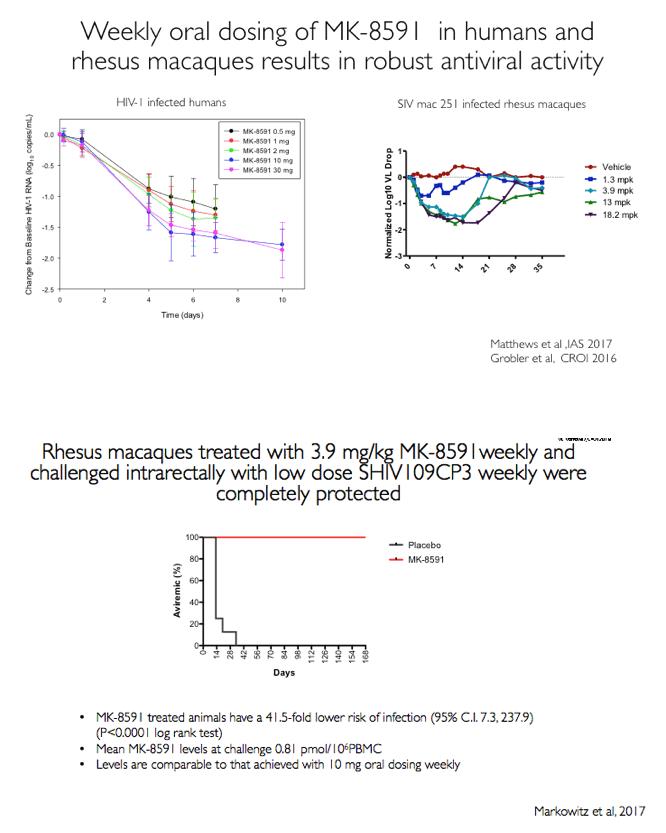
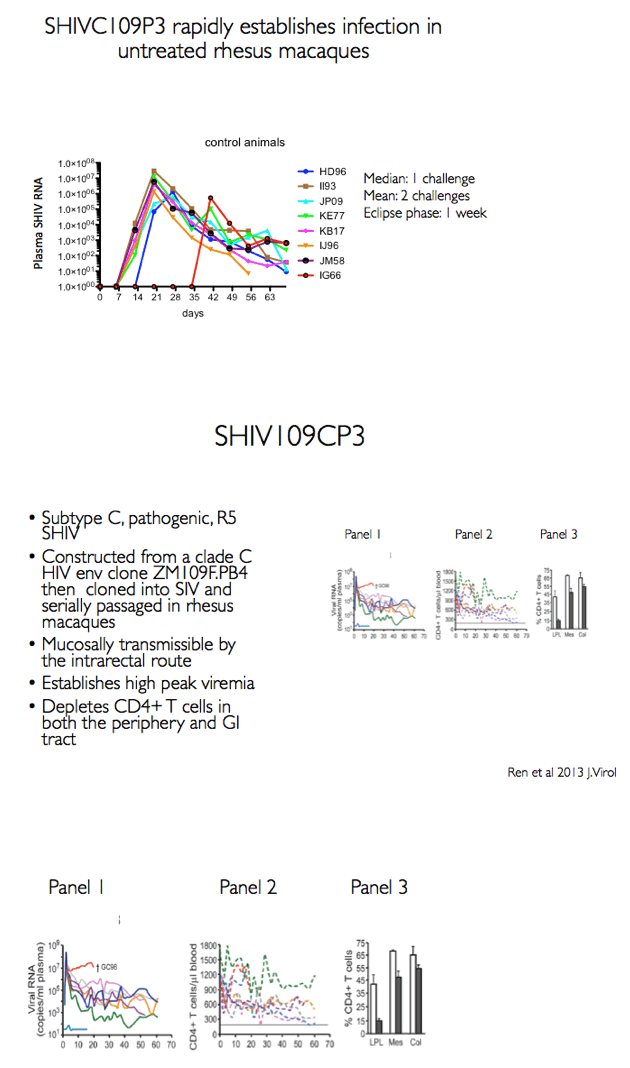
MK-8591 (EFdA) inhibits HIV-1 reverse transcriptase via multiple mechanisms [2,3]. In lab studies, the NRTI remains active against an array of NRTI-resistant clones and patient isolates. Studies in macaques and humans indicate strong antiviral activity with weekly oral dosing, a trait suggesting MK-8591 could find use as preexposure prophylaxis (PrEP). Previous work by researchers at the Aaron Diamond AIDS Research Center and collaborators found that a weekly oral MK-8591 dose of 3.9 mg/kg completely protected 8 macaques from weekly rectal challenges of SHIV 109CP3, a pathogenic R5-using subtype C virus [4].
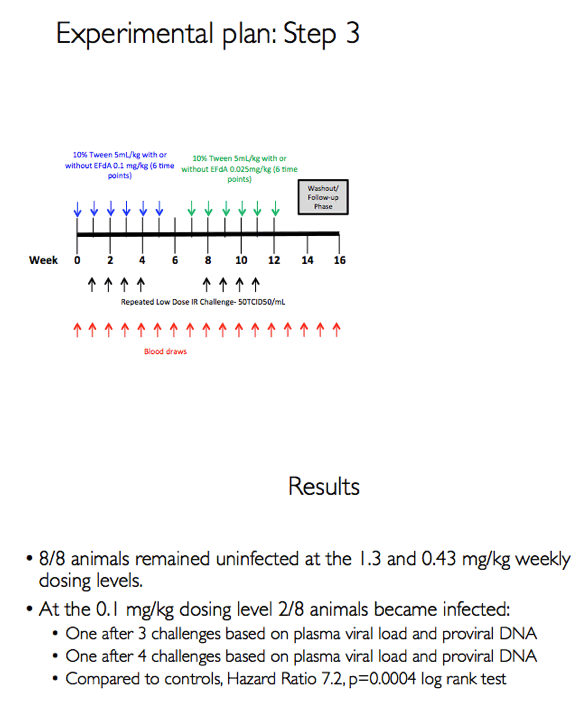
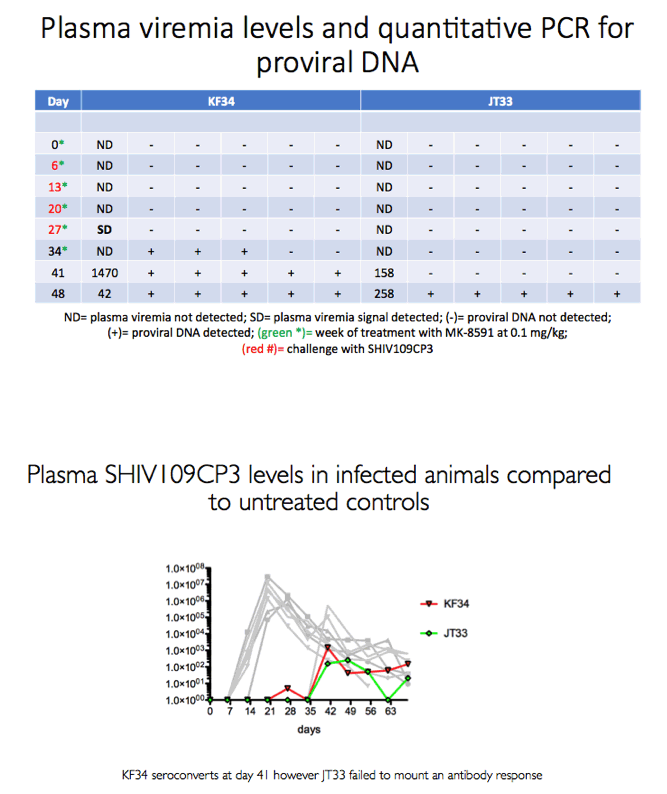
Aiming to determine the MK-8591 dose at which MK-8591 loses its protective capacity, the new study involved 8 male macaques rectally exposed weekly to a 50 TCID dose SHIV 109CP3 for up to 4 challenges. The animals received MK-8591 or inert vehicle (no MK-8591) weekly by oral gavage up to 6 times starting 1 week before the SHIV challenge and ending 1 week after the last challenge. Researchers sequentially tested three doses: 1.30, 0.43, and 0.10 mg/kg weekly.
Eight of 8 macaques receiving a dose of 1.30 or 0.43 mg/kg weekly remained uninfected after SHIV challenges. Two of 8 animals receiving a dose of 0.10 mg/kg became infected, 1 after 3 SHIV challenges and 1 after 4 challenges. Combining these results with those of the earlier study, in which macaques got MK-8591 at a dose of 3.9 mg/kg weekly [4], the researchers determined that animals receiving the NRTI had a 41.47-fold lower risk of SHIV infection than animals not getting the drug (P < 0.0001).
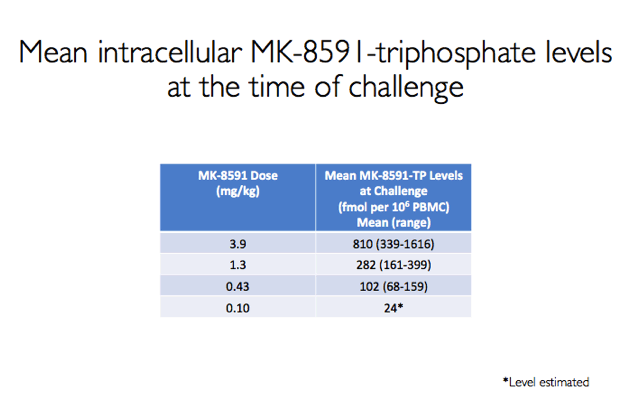
At the time of the SHIV challenge, average intracellular levels of MK-8591 triphosphate (the active form of the drug) were 810 fmol/million cells with 3.9 mg/kg of MK-8591, 282 fmol/million cells with 1.3 mg/kg, 102 fmol/million cells with 0.43 mg/kg, and an estimated 24 fmol/million cells with 0.10 mg/kg.
The researchers concluded that MK-8591 remains completely protective against rectal SHIV at doses of 1.3 and 0.43 mg/kg weekly. The 0.1-mg/kg dose "remains highly protective," they added, at intracellular levels affording 92% protection from SHIV. In the 2 animals who became infected with the 0.1-mg/kg dose, viral loads were lower than those seen in infected animals not receiving MK-8591.
Protective MK-8591 levels attained in this study can be reached in humans, the investigators estimated, at doses below 250 ug weekly or 10 ug daily, calculations "suggesting MK-8591 utility in extended-duration prophylaxis against HIV infection."
References
1. Markowitz M, Gettie A, St. Bernard L, et al. Low dose MK-8591 protects rhesus macaques against rectal SHIV infection. 25th Conference on Retroviruses and Opportunistic Infections (CROI). March 4-7, 2018. Boston. Abstract 89LB.
2. Michailidis E, Marchand B, Kodama EN, et al. Mechanism of inhibition of HIV-1 reverse transcriptase by 4'-ethynyl-2-fluoro-2'-deoxyadenosine triphosphate, a translocation-defective reverse transcriptase inhibitor. J Biol Chem. 2009;284:35681-35691.
3. Michailidis E, Huber AD, Ryan EM, et al. 4'-Ethynyl-2-fluoro-2'-deoxyadenosine (EFdA) inhibits HIV-1 reverse transcriptase with multiple mechanisms. J Biol Chem. 2014;289:24533-24548.
4. Markowitz M, Gettie A, St. Bernard L, et al. Weekly oral MK-8591 protects male rhesus macaques against repeated low dose intrarectal challenge with SHIVC109P3. 9th IAS Conference on HIV Science (IAS 2017), July 23-26, 2017, Paris. Abstract MOAX0203LB. http://www.natap.org/2017/IAS/IAS_10.htm
------------------------------------------
LOW DOSE MK-8591 PROTECTS RHESUS MACAQUES AGAINST RECTAL SHIV INFECTION
New Long Acting Potent Nuke MK-8591 + Doravirine Phase 2B Study Starts - (09/27/17)
New: MK-8591 With Doravirine and Lamivudine in Participants Infected With Human Immunodeficiency Virus Type 1 (MK-8591-011)

Program Abstract
89LB LOW DOSE MK-8591 PROTECTS RHESUS MACAQUES AGAINST RECTAL SHIV INFECTION
Martin Markowitz1, Agegnehu Gettie1, Leslie St. Bernard1, Hiroshi Mohri1, Brooke Grasperge2, James Blanchard2, Li Sun3, Kerry Fillgrove4, Daria Hazuda4, Jay Grobler4
1Aaron Diamond AIDS Research Center, New York, NY, USA, 2Tulane National Primate Research Center, Covington, LA, USA, 3Merck & Co, Inc, Upper Gwynedd, PA, USA, 4Merck & Co, Inc, West Point, PA, USA
Background: MK-8591 (4'-ethynyl-2-fluoro-deoxyadenosine; EFdA), a nucleoside reverse transcriptase translocation inhibitor (NRTTI), was previously shown to completely protect rhesus macaques (RM) against SHIV infection following intrarectal (IR) challenge when dosed at 3.9 mg/kg weekly. At this dose, the mean intracellular MK-8591-triphosphate (TP) concentration of 805 fmol/10Λ6 PBMCs at time of challenge exceeds the lowest MK-8591-TP C168hr that demonstrated potent antiviral activity in HIV infected patients. To determine the lowest drug levels that confer protection, RMs were challenged at progressively lower doses until protection was no longer observed.
Methods: The eight male RM that had been successfully treated with 3.9 mg/kg MK-8591 after 20 weeks (w) were dosed with 1.3 mg/kg MK-8591 orally on day 0 and qw for 6 doses. All animals were again challenged IR with 1 mL of 50TCID50 of SHIVC109P3 on day 6 and weekly thereafter for up to 4 challenges or until infection was confirmed. Prior to challenge, blood was drawn for virology and PK. Infection was confirmed by real-time RT PCR in plasma. Proviral DNA was measured qw by PCR and virus-specific antibody responses were assessed. Intracellular levels of MK-8591- TP were measured by LC/MS/MS. After a 4 w washout, the treatment sequence was repeated on uninfected RM, (0.43 mg/kg weekly for 6 doses) with challenge on day 6 and weekly for up to 4 challenges or until infection was confirmed. After 10 w, this was repeated on the remaining uninfected RM at a dose of 0.1 mg/kg.
Results: All 8 animals remained uninfected after challenges at the 1.3 & 0.43 mg/kg dosing levels. At 0.1 mg/kg, 2/8 animals became infected, with one animal infected after 3 challenges and the other after 4 challenges. Mean levels of intracellular MK-8591-TP at the time of challenge were 282 and 102 fmol/10Λ6 PBMCs at the 1.3 and 0.43 mg/kg dosing levels, respectively. At the 0.1 mg/kg dose MK-8591-TP levels are estimated to be ~24 fmol/10Λ6 PBMCs.
Conclusion: In rhesus macaques MK-8591 completely protects against SHIV infection at weekly doses of 1.3 and 0.43 mg/kg and is partially protective at 0.1 mg/kg (HR 7.2, p=0.0004). MK-8591-TP levels that are protective in this model are achievable in humans at weekly doses of less than 250 μg weekly or 10 μmg daily, suggesting MK-8591 utility in extended duration prophylaxis against HIV infection.

|
| |
|
 |
 |
|
|