 |
 |
 |
| |
Reduction in the incidence of hepatitis C-related decompensated cirrhosis associated with
national scale-up of direct-acting antiviral therapies targeting patients with advanced liver
fibrosis
|
| |
| |
EASL 2018 April 11-15 Paris France
Program Abstract
Sharon Hutchinson1, Heather Valerio1, John Dillon2, Raymod Fox3, Hamish Innes1, Amanda Weir4,
Stephen Barclay5, Scott Mcdonald1, Nicholas Kennedy6, Andrew Fraser7, Adrian Stanley8, Peter
Bramley9, Peter Hayes10, David Goldberg4
1Glasgow Caledonian University, School of Health and Life Sciences, Glasgow, United Kingdom;
2University of Dundee, Department of Molecular and Clinical Medicine, Dundee, United Kingdom;
3Brownlee Centre for Infectious Diseases, Glasgow, United Kingdom; 4Health Protection Scotland,
Glasgow, United Kingdom; 5Glasgow Royal Infirmary, Glasgow, United Kingdom; 6Monklands
Hospital, Airdrie, United Kingdom; 7NHS Grampian, Aberdeen, United Kingdom; 5Glasgow Royal
Infirmary, Glasgow, United Kingdom; 9Stirling Royal Infirmary, Stirling, United Kingdom; 10Royal
Infirmary of Edinburgh, Liver Unit, Edinburgh, United Kingdom
Background and Aims: Direct-acting antivirals (DAAs) for chronic hepatitis C virus (HCV) have been
shown to be highly effective in terminating active infection. Evidence of the impact of DAAs in averting
severe liver morbidity at the population level is however lacking. Scotland, like most other countries,
prioritized DAAs (following first licensing in May 2014) to patients with advanced liver fibrosis, but also
set an ambitious target to reduce the incidence of HCV-related decompensated cirrhosis (DC) by 75%
by 2020. Unlike most other countries, Scotland has national surveillance of HCV treatment and
disease; thus, we aimed to examine the early impact of DAAs on HCV-related DC at the population
level.
Method: Data on the number and characteristics of persons initiated on HCV therapy in Scotland up
to March 2017 were obtained from the Scottish HCV Clinical database. Record-linkage of Scotland's
HCV Diagnosis database to the national inpatient hospital database generated data on the numbers of
persons with a chronic HCV diagnosis that had presented and been admitted to hospital for the first
time with DC (defined as ascites, hepatic encephalopathy, hepatorenal syndrome or bleeding varices)
during 2000-16.
Results: In the three years since the introduction of DAAs (April-14 to March-17), 4,800 people were
initiated on HCV therapy in Scotland, involving: 54% with genotype 1 and 38% genotype 3; 24% with
F2/3 fibrosis stage, 27% compensated and 5% decompensated cirrhosis; 83% treated with DAAs; and
94% with a sustained viral response (SVR) (based on 3240 with available data to date). The number
initiated on therapy was 1.6-fold and 2.8-fold higher in this period (April-14 to March-17), compared to
the preceding three years, for all patients and those with compensated cirrhosis, respectively.
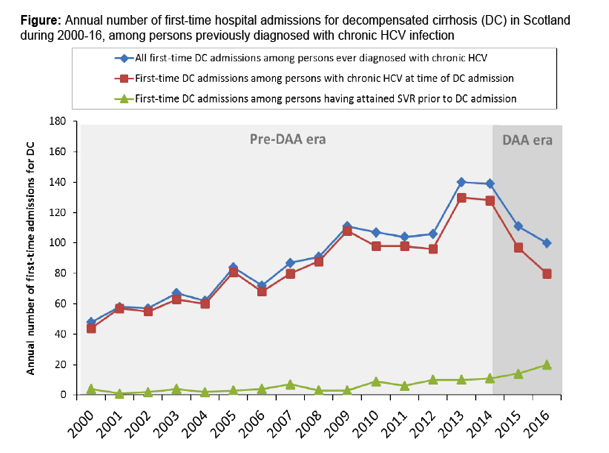
Between 2013 and 2016, we observed a 29% reduction in first-time presentations for DC among all
persons previously diagnosed with chronic HCV (Fig 1); a larger reduction (39%) was observed
among those with chronic HCV at the time of DC admission. However, first-time admissions for DC
doubled from 10 in 2013 to 20 in 2016 among persons who had attained SVR prior to DC
presentation.
Conclusion: These data provide the first country-level evidence of the immediate impact that DAAs
can have in averting HCV-related DC. Greater emphasis needs to be placed however on addressing
comorbidities that pose a continued risk of liver disease progression among those attaining SVR.
|
| |
|
 |
 |
|
|