 |
 |
 |
| |
B/F/TAF versus ABC/DTG/3TC or DTG + F/TAF in treatment-naïve adults with high baseline viral load or low baseline CD4 count in two phase III randomized, controlled clinical trials: week 96 results
|
| |
| |
Bictegravir/F/TAF at 96 Weeks With High Baseline Viral Load or Low CD4 Count
HIV Drug Therapy, Glasgow 2018, October 28-31, 2018, Glasgow
Mark Mascolini
Combining results of two phase 3 trials, researchers found similar high 96-week virologic response rates with bictegravir/emtricitabine/tenofovir alafenamide (B/F/TAF) and two dolutegravir (DTG)-based combinations in antiretroviral-naive people with a pretreatment viral load above 100,000 copies or a pretreatment CD4 count below 200 [1].
People who start antiretroviral therapy (ART) with a high viral load or low CD4 count may respond more slowly than people with less advanced HIV infection. In two double-blind phase 3 studies B/F/TAF proved virologically noninferior to single-tablet DTG/abacavir/lamivudine (ABC/3TC) (Gilead trial 1489) or to DTG plus F/TAF (Gilead trial 1490) in previously untreated people at weeks 48 and 96. The new analysis combined 96-week results of those two trials in people starting ART with a viral load above 100,000 copies or a CD4 count below 200 in Europe, North America, Latin America, or Australia.
Bictegravir and dolutegravir are integrase inhibitors.
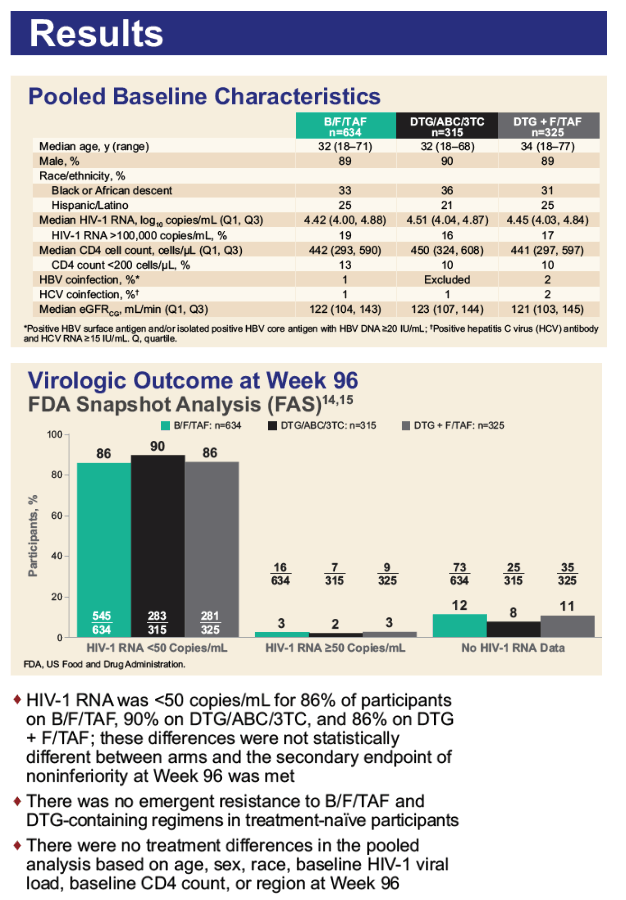
This 96-week per-protocol analysis included everyone with an on-treatment viral load during the 96 weeks (or who stopped treatment for lack of efficacy). The analysis excluded people who violated trial entry criteria or had antiretroviral adherence below the 2.5th percentile. Overall trial populations (regardless of starting viral load or CD4 count) included 634 people randomized to B/F/TAF, 315 to DTG/ABC/3TC, and 325 to DTG+F/TAF. Median ages of the 3 groups were in the low 30s, about 90% were men, about one third black, and about one quarter Hispanic. In the B/F/TAF, DTG/ABC/3TC, and DTG+F/TAF arms, there were 19%, 16%, and 17% with a pretreatment viral load above 100,000 and 13%, 10%, and 10% with a pretreatment CD4 count below 200.
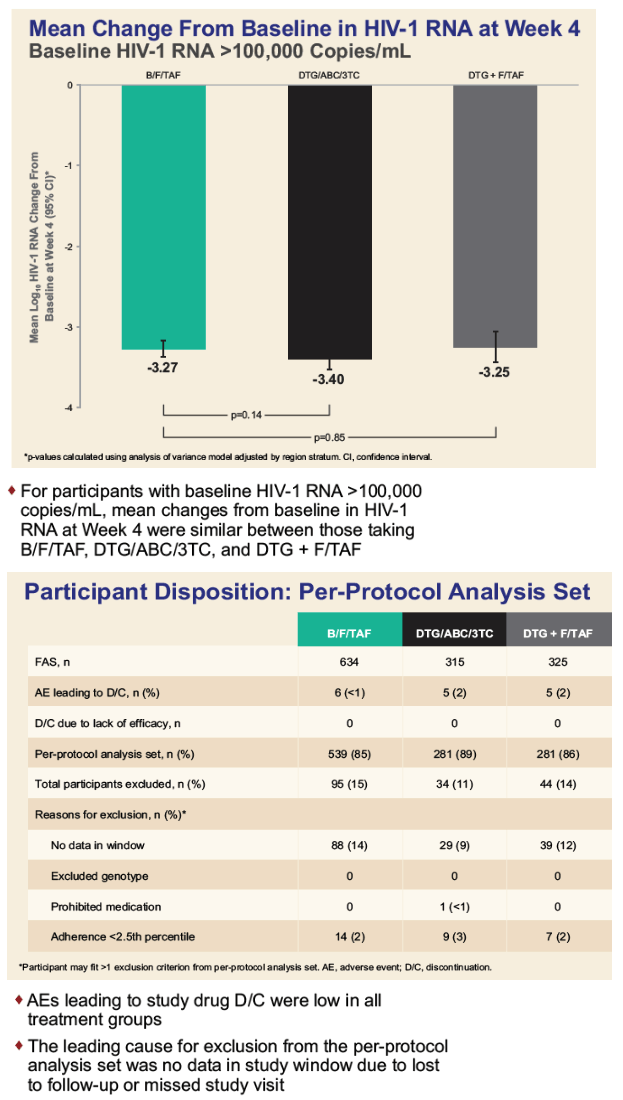
After 4 weeks of treatment, viral load in people with an initial load above 100,000 copies
fell by an average 3.27 log in the B/F/TAF group, 3.40 log in the DTG/ABC/3TC group (not significant versus B/F/TAF`, P = 0.14), and 3.25 log in the DTG+F/TAF group (not significant versus B/F/TAF, P = 0.85). (3.25 log is about 1800 copies.)
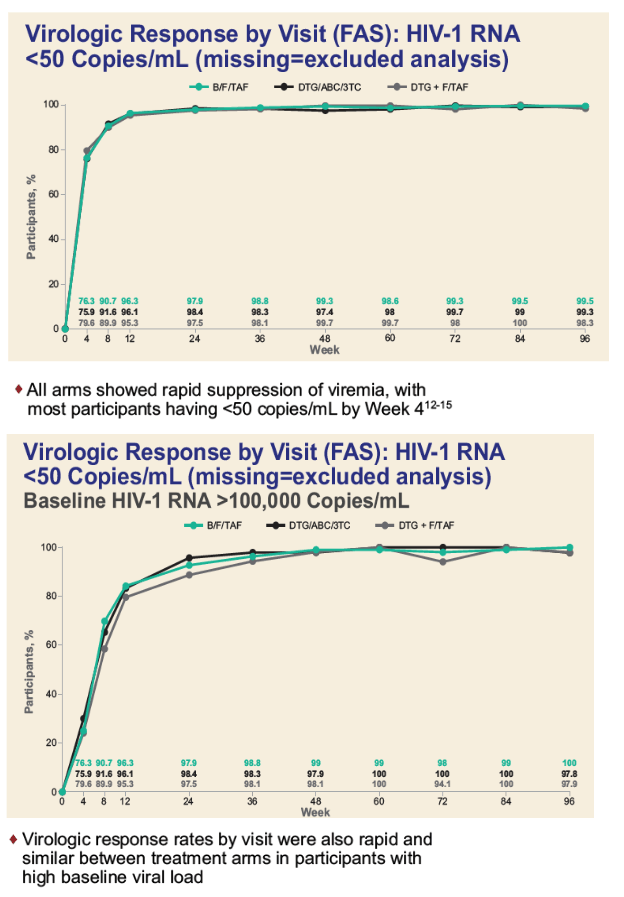
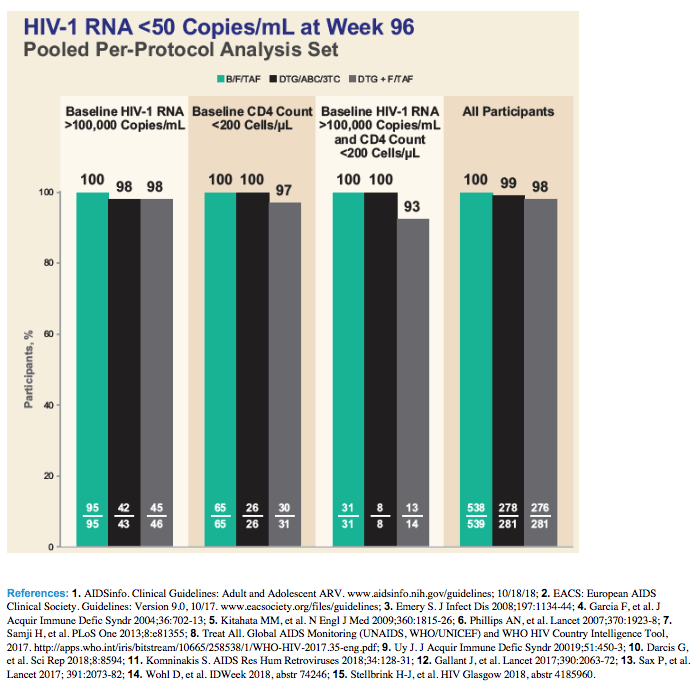
The week-96 per-protocol analysis of proportions with a viral load below 50 copies by the FDA snapshot method showed similar high response rates with all regimens when pretreatment viral load lay above 100,000 copies, pretreatment CD4 count lay below 200, or when both pretreatment conditions applied:
Pretreatment viral load above 100,000 copies
B/F/TAF: 100% (95 of 95)
DTG/ABC/3TC: 98% (42 of 43)
DTG+F/TAF: 98% (45 of 46)
Pretreatment CD4 count below 200
B/F/TAF: 100% (65 of 65)
DTG/ABC/3TC: 100% (26 of 26)
DTG+F/TAF: 97% (30 of 31)
Both pretreatment load above 100,000 and CD4 count below 200
B/F/TAF: 100% (31 of 31)
DTG/ABC/3TC: 100% (8 of 8)
DTG+F/TAF: 93% (13 of 14)
In the per-protocol participant set, proportions with adverse events leading to discontinuation were below1% with B/F/TAF, 2% with DTG/ABC/3TC, and 2% with DTG+F/TAF. Proportions excluded because of adherence below the 2.5th percentile were 2% with B/F/TAF, 3% with DTG/ABC/3TC, and 2% with DTG+F/TAF. No one in any study arm stopped drugs because of lack of efficacy.
Reference
1. Podzamczer D, Stellbrink H, Orkin C, et al. B/F/TAF versus ABC/DTG/3TC or DTG + F/TAF in treatment-naïve adults with high baseline viral load or low baseline CD4 count in two phase III randomized, controlled clinical trials: week 96 results. HIV Drug Therapy, Glasgow 2018, October 28-31, 2018, Glasgow. Abstract P119.
B/F/TAF vs DTG/ABC/3TC or DTG + F/TAF in Treatment-Naïve Adults With High Baseline Viral Load or Low Baseline CD4 Count in 2 Phase 3, Randomized, Controlled, Clinical Trials: Week 96 Results
Daniel Podzamczer,1 Hans-Jürgen Stellbrink,2 Chloe Orkin,3 Anton Pozniak,4 Jose Arribas,5 Ellen Koenig,6 Moti Ramgopal,7 Axel Baumgarten,8 Xuelian Wei,9 Andrew Cheng,9 Devi SenGupta,9 Hal Martin9
1IDIBELL-Hospital Universitari de Bellvitge, L'Hospitalet, Barcelona, Spain; 2ICH Study Center, Hamburg, Germany; 3Ambrose King Centre, Royal London Hospital, Barts Health, London; 4Chelsea and Westminster Hospital, London; 5Hospital Universitario La Paz, Madrid, Spain; 6IDEV: Instituto Dominicano de Estudios Virológicos, Santo Domingo, Dominican Republic; 7Midway Immunology and Research Center, Fort Pierce, Florida, USA; 8ZIBP: Zentrums für Infektiologie Berlin Prenzlauer Berg, Berlin, Germany; 9Gilead Sciences, Inc., Foster City, California, USA
--------------------------------



|
| |
|
 |
 |
|
|