| |
|
Acute HCV - Duration of Treatment?
|
| |
| |
Download the PDF here
Download the PDF here
Download the PDF here
Download the PDF here
EASL: Six weeks of sofosbuvir/ledipasvir (SOF/LDV) are sufficient to treat acute hepatitis C virus genotype 1 monoinfection: The HepNet Acute HCV IV Study- (05/27/16) see publication below.
4 & 8 weeks Acute HCV Trestment:
SLAM C study Sofosbuvir and Ledipasvir versus Sofosbuvir and Simeprevir combination therapy in the management of acute hepatitis C: A randomized open label prospective clinical pilot study. (drug rehabs in NYC]
A5327 Sofosbuvir-Containing Regimens Without Interferon for Treatment of Acute HCV in HIV-1-infected Individuals (SWIFT-C) - (10/24/17)
----------------------------------
Comment
Direct-acting antivirals for acute HCV: how short can we go?
27 February 2017
*Marianne Martinello, Jason Grebely, Gail V Matthews
Treatment for individuals with hepatitis C virus (HCV) infection has evolved rapidly, with combinations of two or more different classes of direct-acting antivirals for 8-12 weeks achieving sustained virological response (SVR) in more than 95% of treatment-naive, non-cirrhotic individuals with chronic HCV infection.1, 2, 3
In this context, the optimal management of acute HCV infection is uncertain. With little data available, recent international guidelines support a fairly conservative approach, with the American Association for the Study of Liver Diseases and Infectious Diseases Society of America (AASLD-IDSA) guidelines advocating the same regimens for acute HCV as recommended for chronic HCV, while the European Association for the Study of the Liver (EASL) guidelines suggest sofosbuvir plus an NS5A inhibitor for 8 weeks, with a longer duration of 12 weeks for those with HIV or baseline HCV RNA higher than 1 000 000 IU/mL.
Trials are underway to fill this evidence gap, assessing short duration direct-acting antiviral regimens in acute (<6 months) and recent (<12 months) HCV infection.4, 5, 6Preliminary results following 4 weeks and 6 weeks of sofosbuvir-ledipasvir in acute genotype 1 HCV monoinfection have been encouraging.4, 6
In The Lancet Gastroenterology & Hepatology, Jurgen Rockstroh and colleagues7 have further advanced the field by examining the efficacy and safety of 6 weeks of sofosbuvir-ledipasvir for acute HCV infection in HIV coinfection.7 All 26 participants were men who have sex with men with well-controlled HIV and HCV genotypes 1a or 4. Most were asymptomatic, with only two presenting with jaundice. Data for HCV transmission risk behaviours, including injection drug use and sexual behaviour, were not collected.4 Overall, 20 (77%; 95% 56-91) of the participants had an SVR at 12 weeks (SVR12). Of the six participants who did not achieve SVR12, three had virological relapse, one was reinfected before post-treatment week 4, and two were lost to follow-up after achieving SVR at 4 weeks. The three participants with relapse had high baseline HCV RNA (>6⋅96 log10 IU/mL).
While this study7 and other pilot studies4, 6 show promising efficacy with ultra-short duration therapy in acute HCV, many questions remain unanswered. The effect of clinical, virological, and immunological factors on efficacy-eg, HIV infection, baseline HCV RNA, method of HCV transmission (injection drug use, sexual exposure), clinical presentation, and duration of infection-remain to be adequately determined. Excellent efficacy (SVRs of 100%) has been noted in largely symptomatic, acute HCV-monoinfected patients,6 whereas SVR was lower in Rockstroh and colleagues' study of, predominantly asymptomatic, HIV-coinfected participants. Baseline HCV RNA seems to affect response to short duration direct-acting antiviral therapy;4, 5 high baseline HCV RNA in the three relapsed cases could suggest that, even with a potent direct-acting antiviral regimen, viral suppression is protracted in individuals with a high HCV burden. It is unclear whether any people who inject drugs (PWID; a population in whom targeted therapy might prevent onward transmission) were included in the study by Rockstroh and colleagues7 and these individuals were excluded in the study by Deterding and colleagues.6 The effect of duration of HCV infection is also unclear. In the study by Deterding and colleagues,6 duration of infection at screening was short (≤4 months), whereas in this study by Rockstroh and colleagues,7 the protocol specified a duration of infection of 6 months or less. However, the estimated duration of HCV infection could not be determined for all patients, raising the possibility that treatment began in the early chronic phase of infection. Because the distinction between acute and early chronic infection is somewhat arbitrary, further research regarding timing of treatment initiation within 1 year of infection is warranted and will allow broader clinical application. Combined, these studies offer exciting potential, but are restricted by small sample sizes and selected populations.
Moving forward, optimal direct-acting antiviral regimen choice will be crucial. Mathematical modelling has suggested that rapid on-treatment second-phase viral decline should allow shorter treatment durations.8 The use of a potent dual-class or triple-class direct-acting antiviral regimen, including an HCV NS3/4A protease inhibitor, NS5A inhibitor, or both (with or without a nucleotide analogue) seems justified.8 Pilot studies to date have been genotype-specific; however, a pangenotypic strategy would be ideal for clinical implementation. Further large studies of pangenotypic direct-acting antiviral therapy in well-characterised populations (including people with HIV and PWID) would greatly enhance our understanding of the use of direct-acting antiviral therapy in this field. Several studies are ongoing to assess short-duration direct-acting antiviral regimens in people with acute and recent HCV, including DAHHS-2 (NCT02600325) and REACT (NCT02625909).
Access to HCV treatment for people at high risk of onward transmission, including those with acute and recent HCV infection, should be a priority.9 As we begin to explore the use of ultrashort direct-acting antiviral therapy in acute and recent HCV infection, one question persists: how short can we go?
Pdf attached at top
------------------------------------
Ledipasvir-sofosbuvir for 6 weeks to treat acute hepatitis C virus genotype 1 or 4 infection in patients with HIV coinfection: an open-label, single-arm trial
Overall, 20 (77%; 95% CI 56-91) of 26 patients achieved SVR12: 15 (79%) of 19 with genotype 1a, and five (71%) of seven with genotype 4. Of six patients not achieving SVR12, three relapsed, two achieved sustained virological response 4 weeks after the end of treatment but were lost to follow-up, and one was reinfected.
Of the 14 patients with viral load of less than 800 000 IU/mL at baseline, 12 (86%) had SVR12 compared with eight (67%) of 12 of those with viral load 800 000 IU/mL or higher at baseline. By week 2, 19 (73%) of 26 patients had HCV RNA less than the lower limit of quantification. No relation between undetectable HCV RNA at week 2 and SVR12 rates was noted.
The three patients who relapsed all had pretreatment HCV viral loads above 6⋅96 log10 IU/mL suggesting that all-oral DAA-associated HCV elimination might take longer in patients with a high HCV viral burden. This association is well appreciated with treatment of chronic HCV, where shorter treatment durations of 8 weeks have been possible using ledipasvir-sofosbuvir if patients were treatment-naive and had no signs of advanced liver disease.15 However, treatment durations shorter than 8 weeks, even with the use of three potent DAAs have been associated with high relapse rates in patients with chronic HCV infection.16, 17
Pdf attached at top.
-------------------------------
EASL Guidelines for Acute HCV

-------------------------------------
Comment
Cutting out the roots of acute hepatitis C
Audrey Coilly, *Didier Samuel
Hepatitis C infection is mainly a chronic liver disease with severe complications such as decompensated cirrhosis and liver cancer. Several studies have shown that the consequences of chronic hepatitis C virus (HCV) infection are not confined to the liver but also increase the risk of cardiovascular and renal diseases and cancer.1 Most patients with chronic hepatitis C were infected before 1990, when the HCV antibody testing of blood donors was introduced. The incidence of acute hepatitis C has fallen substantially because of improvements in hospital hygiene and aseptic conditions during invasive medical and surgical techniques, and because of the very low risk of contamination through blood transfusion, at least in high-income countries (<1 per 2 million blood units).2 There nevertheless remains a risk of acute hepatitis, particularly among people who inject drugs, HIV-positive and HIV-negative men who have sex with men, and more rarely in health-care workers, as well as in countries where blood donations are not secure regarding the transmission of HCV infection or where hospital hygiene is suboptimal.
By contrast with acute HIV infection, there are no guidelines for the preventive treatment of patients at risk of acute HCV infection but without documented acute HCV hepatitis. However, the treatment of early HCV during the acute phase is crucial to preventing chronicity and long-term complications. The treatment of acute hepatitis C can be difficult. Mostly asymptomatic, the disease usually goes undiagnosed and is only detected when screening is done in a population at risk or when the disease becomes symptomatic. The risk of evolution from acute to chronic infection is high (50-80%), but not constant, so any decision to treat acute hepatitis or not must be balanced; this was notably the case in the early years of interferon use because this drug was poorly tolerated. In 2001, a study showed that interferon-based therapy was highly effective if initiated less than 3 months after the onset of infection,3 but its potential adverse effects led to many patients refusing the drug.
In The Lancet Infectious Diseases, Katja Deterding and colleagues4 report the first prospective study investigating the efficacy and safety of a 6-week course of ledipasvir plus sofosbuvir for treatment of acute HCV genotype 1 monoinfection. Such a study was eagerly awaited. 20 patients, recruited from ten centres in Germany, were treated, and all achieved a sustained virological response 12 weeks after completion of study treatment. This result might have been predicted in view of the efficacy of all-oral interferon-free regimens to treat chronic hepatitis C, even in patients with advanced liver disease.5 The findings support the preliminary data of the SLAM C study in which 29 people who inject drugs and had acute HCV genotype 1 infection were treated with sofosbuvir and ledipasvir (14 patients) for 4 weeks, or sofosbuvir and simeprevir (15 patients) for 8 weeks. In a per-protocol analysis, the proportion of patients who achieved a sustained virological response was 100% in both groups.6
Another important factor highlighted by the study by Deterding and colleagues is the duration of treatment. If hepatitis C is treated early, before the occurrence of underlying liver fibrosis, this will facilitate viral clearance and enable a shorter treatment duration, which at present is between 8 and 12 weeks for patients with chronic hepatitis C.6 A 6-week course seems to be sufficient to treat acute HCV genotype 1 monoinfection using the combination of ledipasvir and sofosbuvir. However, in the absence of data, no recommendations can be made for other combinations of direct-acting antiviral agents. During the DARE-C study, 19 patients (including 14 with HIV co-infection) were treated for acute hepatitis C with sofosbuvir and ribavirin for 6 weeks, and the proportion of patients who achieved a sustained virological response only reached 32%.7Treatment combining sofosbuvir and ribavirin could not be recommended. In the SWIFT-C study, 17 HIV-infected patients were treated for acute hepatitis C with a combination of sofosbuvir and ribavirin for 12 weeks, and 59% of patients achieved a sustained virological response.8 Finally, no data have as yet been published on patients with an acute infection of HCV genotype 3.
The good tolerability of interferon-free combination therapies is an argument in favour of their use to treat acute hepatitis C. Deterding and colleagues reported only one serious adverse events with ledipasvir plus sofosbuvir, which was deemed unrelated to treatment, although 22 potentially treatment-related events occurred, the most frequent being gastrointestinal symptoms in four patients.
The most important effect of treating acute hepatitis C is the reduction of the spread of this disease, which is difficult to demonstrate. This approach is crucial to preventing the appearance of new cases, and forms part of efforts to achieve eradication of the virus through the universal treatment of hepatitis C. However, to be effective, this approach must be accompanied by a comprehensive care package that includes routine screening in high-risk populations, preventive measures, and health education. Indeed, it is important that patients should be aware of how HCV infection can be transmitted and implement behaviour to avoid any recontamination.9 The role of patient organisations and structures that support people who inject drugs and at-risk individuals is particularly important in this situation.
Potential limitations of the study by Deterding and colleagues include the small number of patients and the high rates of re-infection in at-risk populations receiving high-cost treatment. However, other studies will be done to establish the optimum strategy, and the costs of treatment will subsequently fall. Finally, most patients with acute HCV infection in high-risk populations will be treated, so that the number of patients able to transmit HCV will decrease as a result, although this should not obviate the importance of preventive measures.
---------------------
Ledipasvir plus sofosbuvir fixed-dose combination for 6 weeks in patients with acute hepatitis C virus genotype 1 monoinfection (HepNet Acute HCV IV): an open-label, single-arm, phase 2 study
Katja Deterding,, MD
Pdf attached at top of report
Summary
Background
Early treatment of acute hepatitis C virus (HCV) infection with interferon alfa is highly effective, but can be associated with frequent side-effects. We investigated the safety and efficacy of an interferon-free regimen for treatment of acute HCV infection.
Methods
In this prospective, open-label, multicentre, single-arm pilot study, we enrolled adults (≥18 years) with acute HCV genotype 1 monoinfection from ten centres in Germany. Patients were given ledipasvir (90 mg) plus sofosbuvir (400 mg) as a fixed-dose combination tablet once daily for 6 weeks. The primary efficacy outcome was the proportion of patients with sustained virological response (defined as undetectable HCV RNA 12 weeks after the end of treatment; other primary outcomes were safety and tolerability of ledipasvir plus sofosbuvir. The primary analysis population consisted of all patients who received at least one dose of study drug. Safety was also assessed in all patients who received at least one dose of the study drug. This trial is registered with ClinicalTrials.gov, number NCT02309918.
Findings
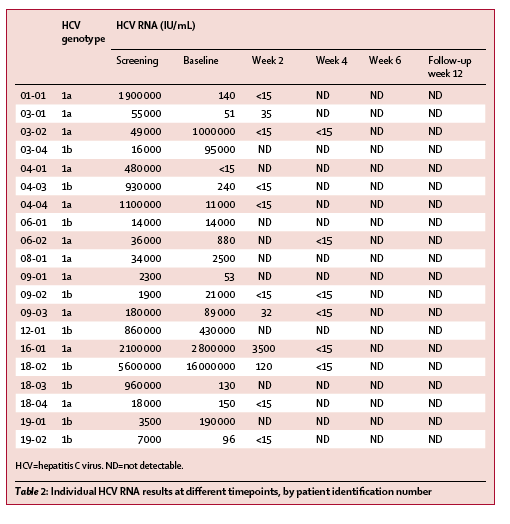
Between Nov 19, 2014, and Nov 10, 2015, we enrolled 20 patients. Median HCV RNA viral load at baseline was 4⋅04 log10 IU/mL (1⋅71-7⋅20); 11 patients were infected with HCV genotype 1a and nine patients with genotype 1b. All patients achieved a sustained virological response 12 weeks after the end of treatment (20 [100%] of 20 patients). Treatment was well tolerated; there were no drug-related serious adverse events. Up to 12 weeks after treatment, 22 possible or probable drug-related adverse events were reported. There was one serious adverse event, which was judged unrelated to the study drug; one patient was admitted to hospital for surgery of a ruptured cruciate ligament.
Interpretation
Treatment for 6 weeks with ledipasvir plus sofosbuvir was well tolerated and highly effective in patients with acute HCV genotype 1 monoinfection. Short-duration treatment of acute hepatitis C might prevent the spread of HCV in high-risk populations.
| |
| |
| |
|
|
|