 |
 |
 |
| |
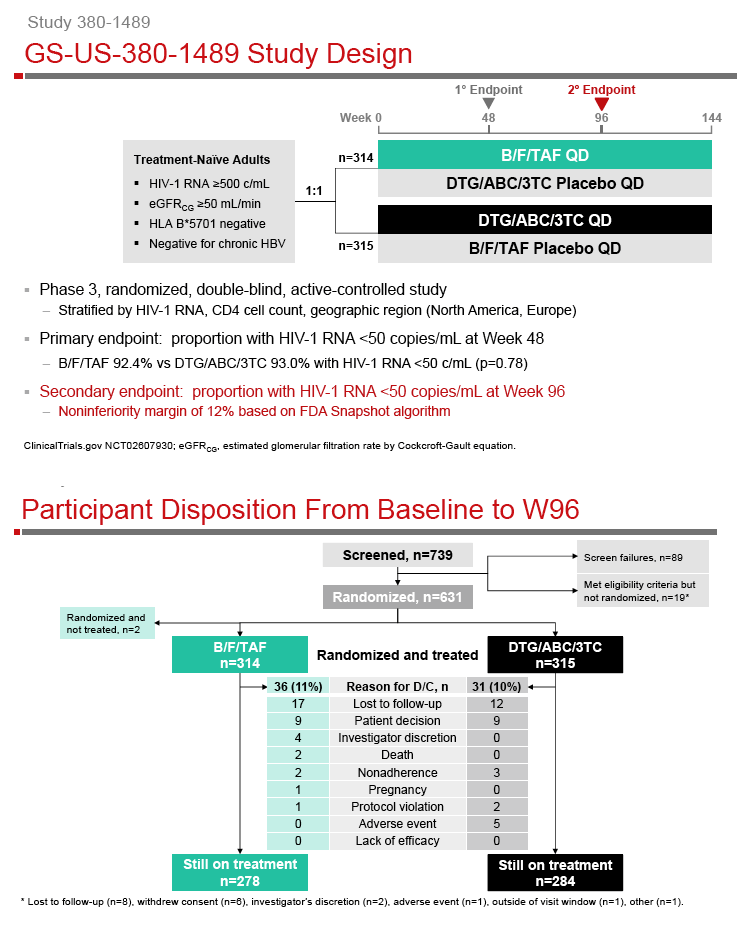
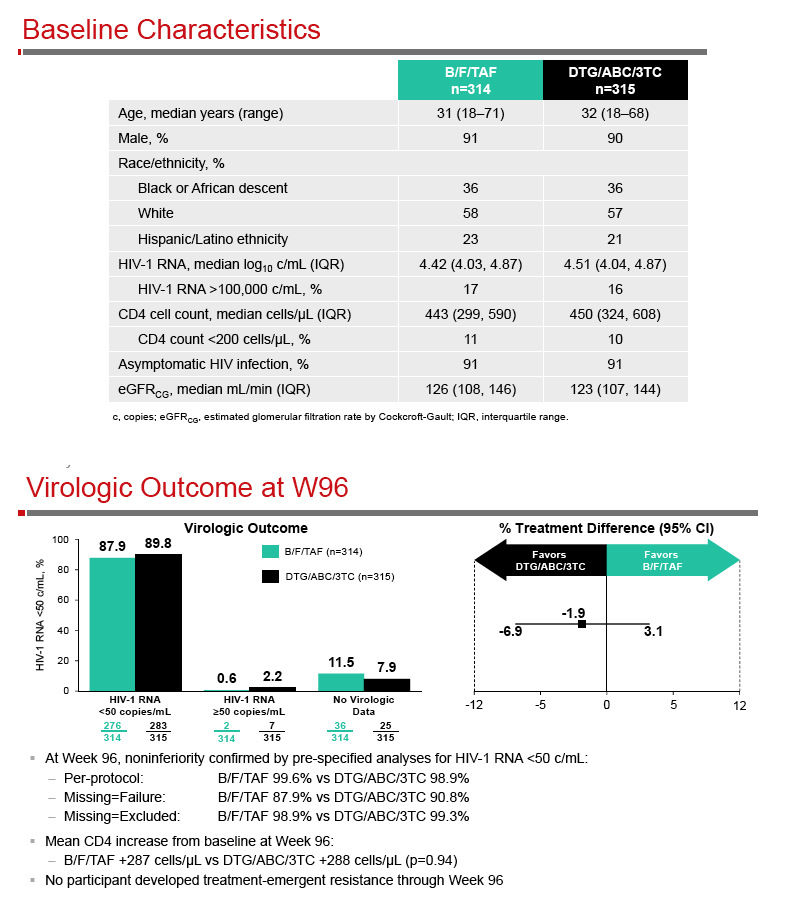
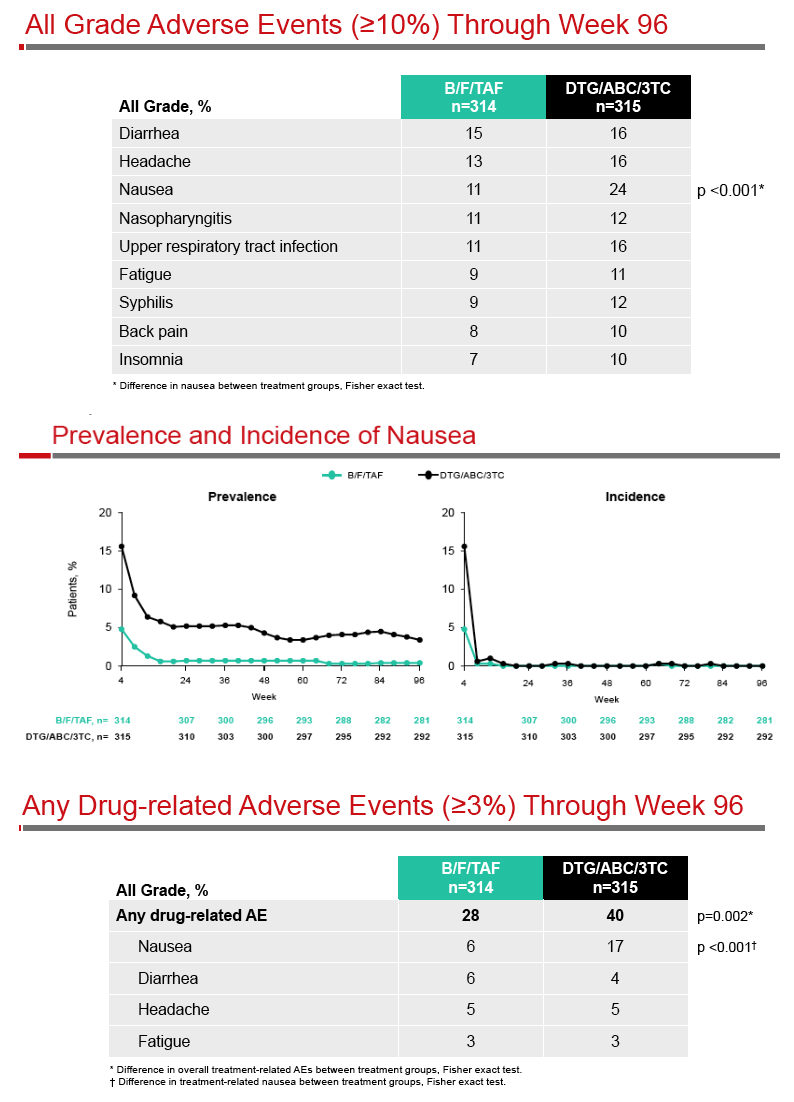
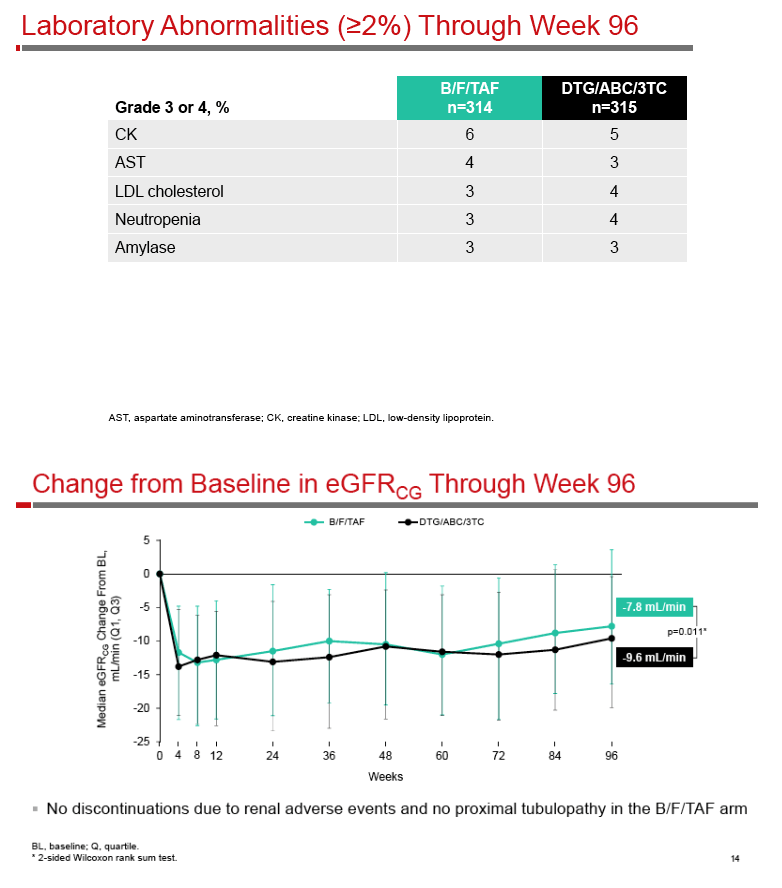
A Phase 3, Randomized, Controlled Clinical Trial of Bictegravir in a Fixed-Dose Combination, B/F/TAF, vs DTG/ABC/3TC in Treatment-Naïve Adults at Week 96
|
| |
| |
Reported by Jules Levin
IDWeek 2018, October 3-7, 2018, San Francisco
David Wohl,1 Yazdan Yazdanpanah,2 Axel Baumgarten,3 Amanda Clarke,4 Melanie A. Thompson,5 Cynthia Brinson,6 Debbie Hagins,7 Moti N. Ramgopal,8 Andrea Antinori,9 Xuelian Wei,10 Kirsten White,10 Sean Collins,10 Andrew Cheng,10 Hal Martin10
1University of North Carolina at Chapel Hill, NC; 2Hopital Bichat Claude Bernard, Paris, France; 3Zentrum fur Infektiologie Berlin Prenzlauer Berg, Berlin, Germany; 4Royal Sussex County Hospital, Brighton, UK; 5AIDS Research Consortium of Atlanta, GA; 6Central Texas Clinical
Research, Austin, TX; 7Georgia Department of Public Health, Chatham Care Center, Savannah, GA; 8Midway Immunology Center, Ft. Pierce, FL; 9National Institute for Infectious Diseases Lazzaro Spallanzani-IRCCS, Rome, Italy; 10Gilead Sciences, Inc., Foster City, CA


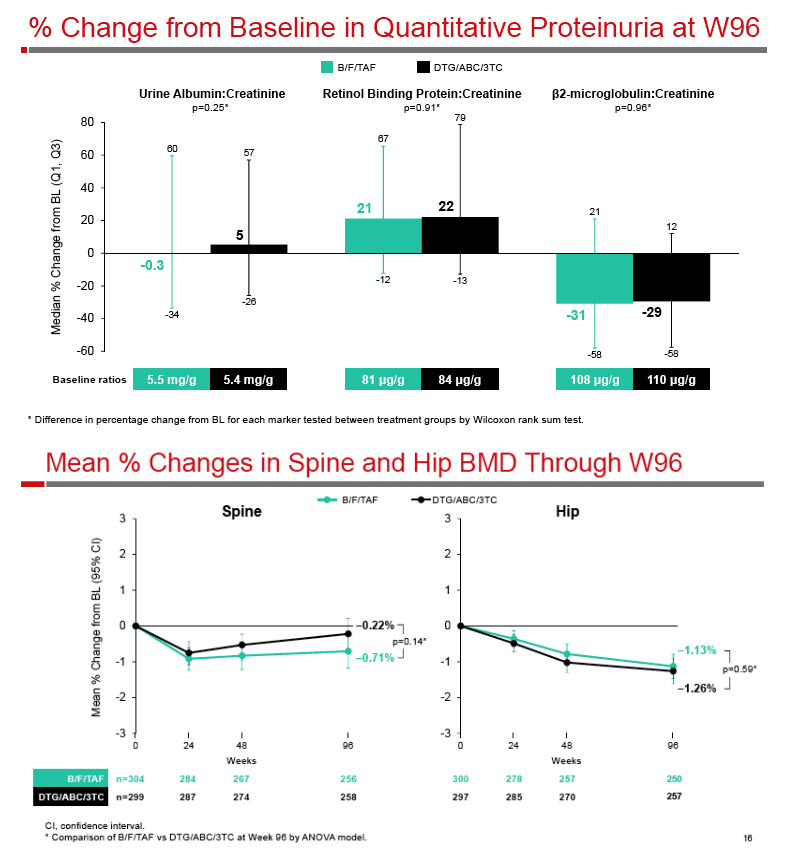
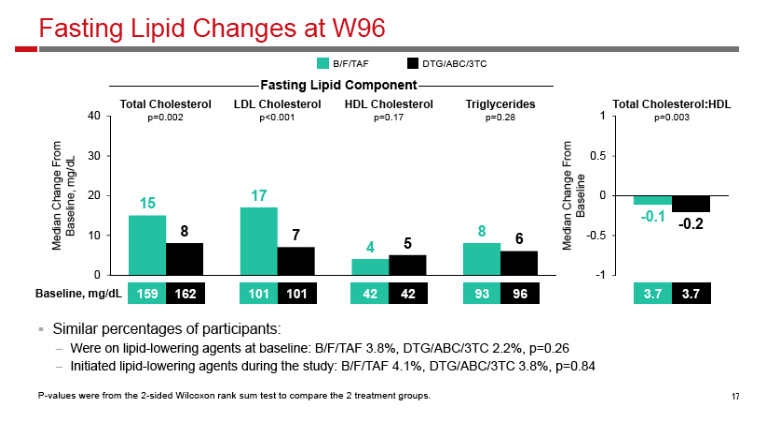
|
| |
|
 |
 |
|
|