 |
 |
 |
| |
Assessment of maternal and fetal dolutegravir exposure by integrating ex vivoplacental perfusion data and physiologically-based pharmacokinetic modeling
|
| |
| |
Reported by Jules Levin
19th International Workshop on Clinical
Pharmacology of Antiviral Therapy 2018
Baltimore, MD, USA
22-24 May 2018
Jolien J.M. Freriksen
PhD student
Radboudumc, Nijmegen, The Netherlands
Program Abstract
Assessment of maternal and fetal dolutegravir exposure by integrating ex vivo placental perfusion data and physiologically-based pharmacokinetic modeling
Freriksen J1,2, Schalkwijk S1,2, Colbers A2, Abduljalil K3, Russel F1, Greupink R1, Burger D2
1Department of Pharmacology and Toxicology, Radboud university medical center, Nijmegen, The Netherlands, 2Department of Pharmacy,
Radboud university medical center, Nijmegen, The Netherlands, 3Simcyp Limited (a Certara Company), Blades Enterprise Centre, Sheffield, United Kingdom
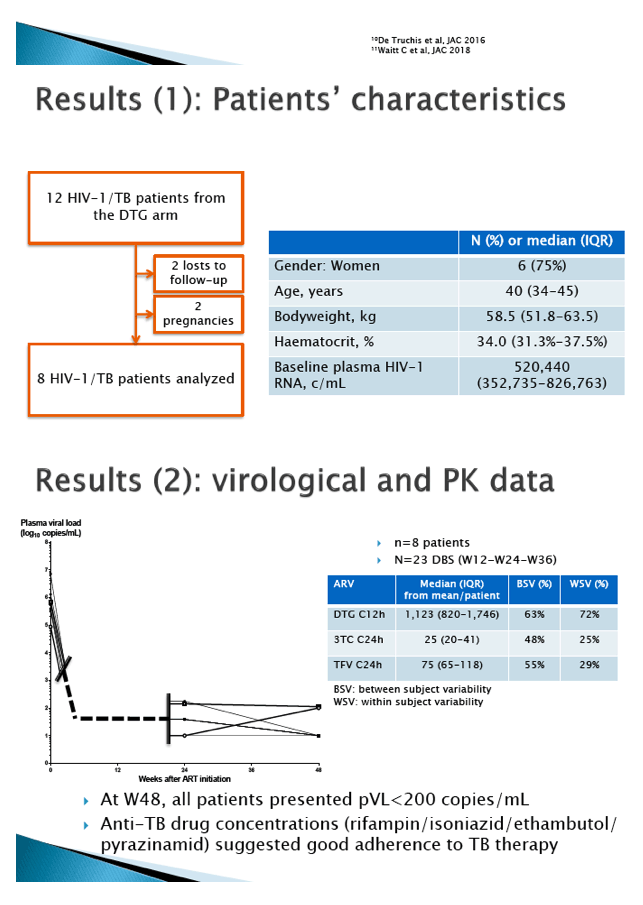
Background: Pregnancy is associated with a variety of physiological and anatomical changes that can alter antiretroviral pharmacokinetics. Subtherapeutic maternal dolutegravir exposure may result in virology breakthrough and mother-to-child transmission of HIV. On the other hand, fetal dolutegravir exposure following maternal dosing could be a determinant of fetal toxicities but may also have potential as preexposure prophylaxis. Pregnancy physiologically-based pharmacokinetic (p-PBPK) models can be of great value to assess maternal and fetal drug exposure. However, to simulate fetal exposure using a p-PBPK model, data on placental drug transfer is necessary. Here, we studied placental dolutegravir transfer via ex vivo dual-side cotyledon perfusion experiments. We integrated these data into the p-PBPK model and simulated maternal and fetal dolutegravir exposure in third trimester pregnant women to explore the clinical implications of standard dolutegravir dosing for mother and child.
Methods: A PBPK model for dolutegravir exposure in healthy volunteers was established and model performance was verified against clinical pharmacokinetic datasets of several dolutegravir dosing regimens. In a next step, physiological parameters were modified to reflect maternal physiological changes during pregnancy. The p-PBPK model was extended with a feto-placental unit and parameterized using dolutegravir transplacental kinetics obtained by performing ex vivo dual side cotyledon perfusion experiments using healthy term human placentas. Subsequently, the model was used to simulate maternal and fetal exposure after maternal dosing and simulations were compared with available clinical pharmacokinetic data from the PANNA network1 and the IMPAACT group2.
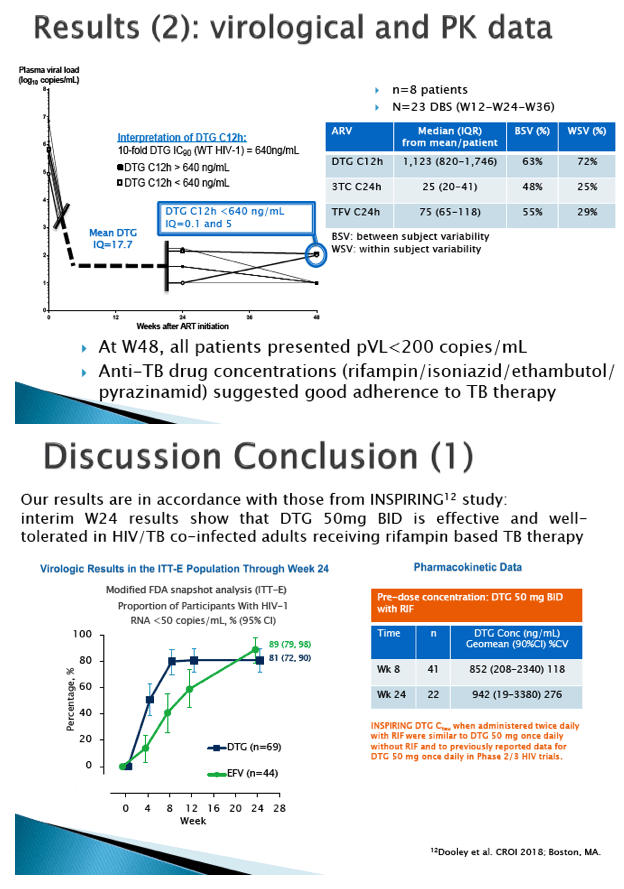
Results: The initial PBPK model was able to adequately capture clinical dolutegravir pharmacokinetics in healthy volunteers. Based on six successful perfusions, dolutegravir cotyledon clearances (mean±SD) were 1.03±0.06 mL/min and 1.03±0.23mL/min for the maternal-to-fetal and the fetal-to-maternal direction, respectively. Scaled placental transfer data were incorporated into the established p-PBPK model. Simulations suggested that administration of 50mg dolutegravir QD would result in a maternal C24h of 0.98mg/L, which is in line with clinical pharmacokinetic data of third trimester HIV-1-infected women obtained by the PANNA network and the IMPAACT group (C24h: 0.7mg/L and 0.93mg/L, respectively). Based on the predicted fetal C24h level of 0.65mg/L, a fetal-to-maternal C24h concentration ratio of 0.66 could be calculated, which is in the range of the clinically observed umbilical cord blood-to-maternal blood concentration ratios (0.32-1.6, n=5)¹. Furthermore, the predicted fetal C24h is above the EC90 for viral inhibition (0.064μg/L)³.
Conclusions: The integrated approach, in which in vitro-to-in-vivo extrapolation of cotyledon perfusion data is incorporated into a p-PBPK model, enables simulation of maternal and fetal pharmacokinetics simultaneously. It provides a tool to guide maternal dosing and ensure safety of the mother and her unborn child. Following this approach, the model suggested that a dose of 50mg dolutegravir QD should result in maternal therapeutic exposure and may have potential for fetal pre-exposure prophylaxis.
1 Bollen P. et al. A comparison of the pharmacokinetics of dolutegravir in pregnancy and postpartum. 18th International Workshop on
Clinical Pharmacology of Antiviral Therapy. June 2017, Chicago
2 Mulligan N. et al. Dolutegravir Pharmacokinetics in Pregnant and Postpartum Women Living with HIV. AIDS, 2018 Jan 23.
³ FDA Clinical Pharmacology Review, Dolutegravir




|
| |
|
 |
 |
|
|