 |
 |
 |
| |
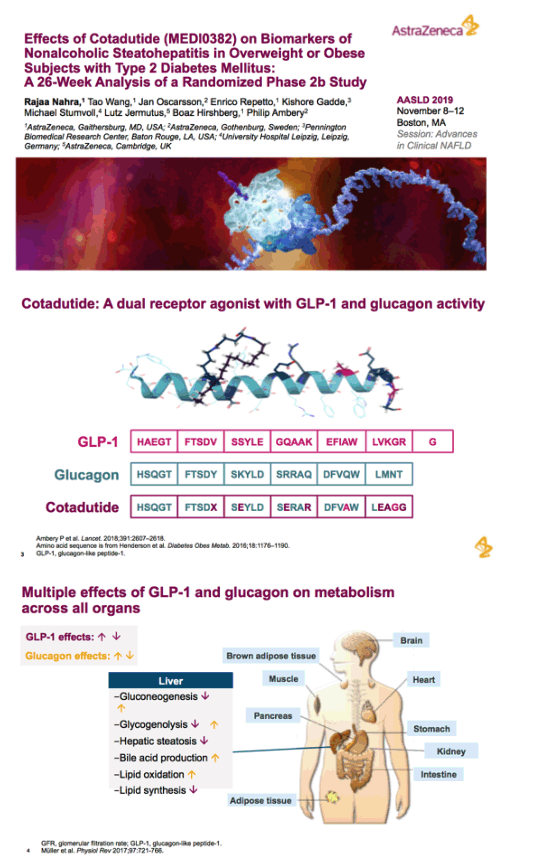
Effects of Cotadutide (MEDI0382) on Biomarkers of Nonalcoholic Steatohepatitis in Overweight or Obese Subjects with Type 2 Diabetes Mellitus:
A 26-Week Analysis of a Randomized Phase 2b Study
|
| |
| |
AASLD 2019 Nov 8-12 Boston
Rajaa Nahra,1 Tao Wang,1 Jan Oscarsson,2 Enrico Repetto,1 Kishore Gadde,3 Michael Stumvoll,4 Lutz Jermutus,5 Boaz Hirshberg,1 Philip Ambery2
1AstraZeneca, Gaithersburg, MD, USA; 2AstraZeneca, Gothenburg, Sweden; 3Pennington Biomedical Research Center, Baton Rouge, LA, USA; 4University Hospital Leipzig, Leipzig, Germany; 5AstraZeneca, Cambridge, UK
--------------------------------
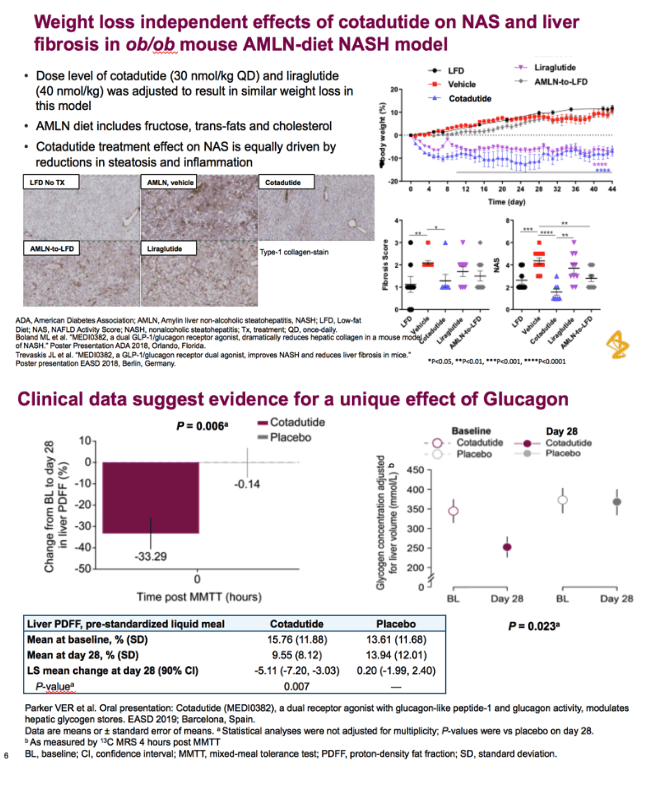
glucagon-like peptide-1 (GLP-1) and glucagon receptor dual agonist. Once-daily cotadutide, an investigational dual receptor agonist with glucagon-like peptide-1 (GLP-1) and glucagon receptor activity, was associated with a reduction in hepatic glycogen in obese and overweight patients with type 2 diabetes (T2D)
https://www.univadis.co.uk/viewarticle/easd-2019-cotadutide-reduces-glycogen-in-patients-with-t2d-693476
Efficacy, safety, and mechanistic insights of cotadutide a dual receptor glucagon-like peptide-1 and glucagon agonist
.....Journal of Clinical Endocrinology & Metabolism.....14 October 2019...
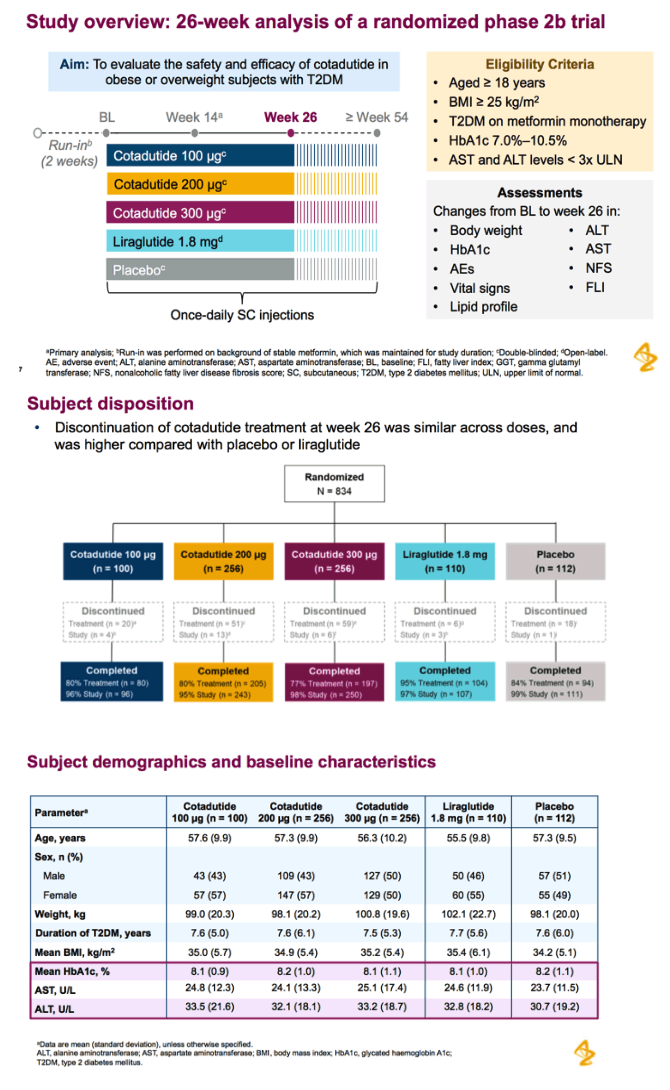
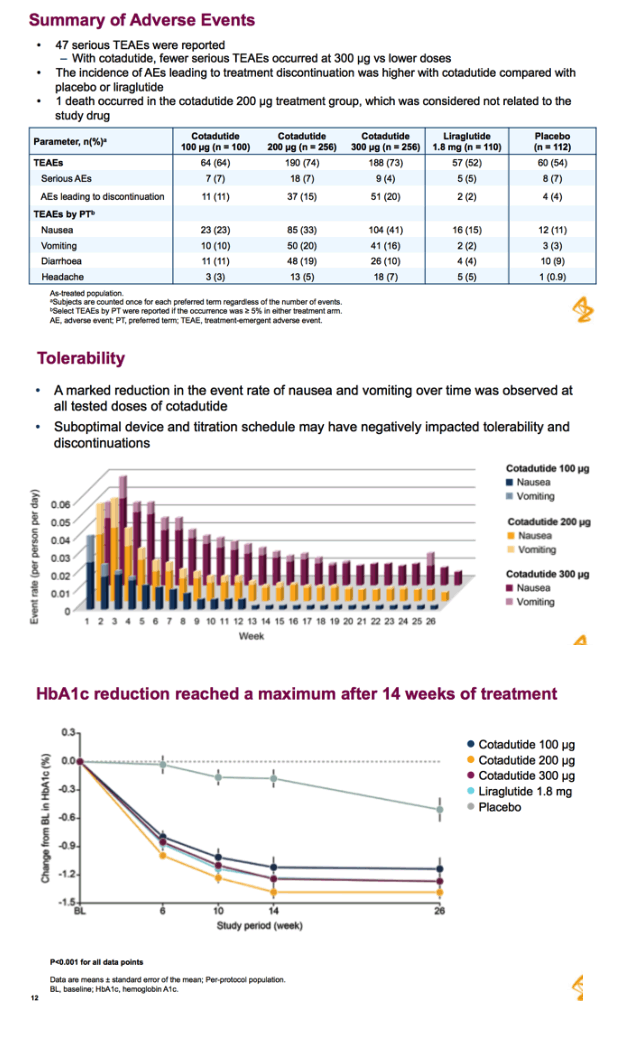
Abstract...Cotadutide is a dual receptor agonist with balanced GLP-1 and glucagon activity that is under development for T2DM, obesity, and nonalcoholic steatohepatitis. Clinical and preclinical studies in T2DM have shown that cotadutide improves glycemic control and promotes body weight loss (23,24). In a randomized, controlled, phase 2a study, treatment with cotadutide resulted in a significant reduction in postprandial and fasting glucose levels, and body weight, compared with placebo in obese or overweight patients with T2DM (24).
Context: Cotadutide is a dual receptor agonist with balanced glucagon-like peptide-1 (GLP-1) and glucagon activity.
Objective: To evaluate different doses of cotadutide and investigate underlying mechanisms for its glucose-lowering effects.
Design/setting
Randomized, double-blind, phase 2a study conducted in two cohorts at five clinical trial sites
Patients: 65 adult overweight/obese patients with type 2 diabetes mellitus; 63 completed the study; 2 were withdrawn due to AEs.
Intervention
Once-daily subcutaneous cotadutide or placebo for 49 days. Doses (50-300 µg) were uptitrated weekly (cohort 1) or biweekly (cohort 2).
Main outcome measures: Coprimary end points (cohort 1) were percentage changes from baseline to end of treatment in glucose AUC0-4h post-mixed-meal tolerance test (MMTT) and weight. Exploratory measures included postprandial insulin and gastric emptying time (GET; cohort 2).
Results
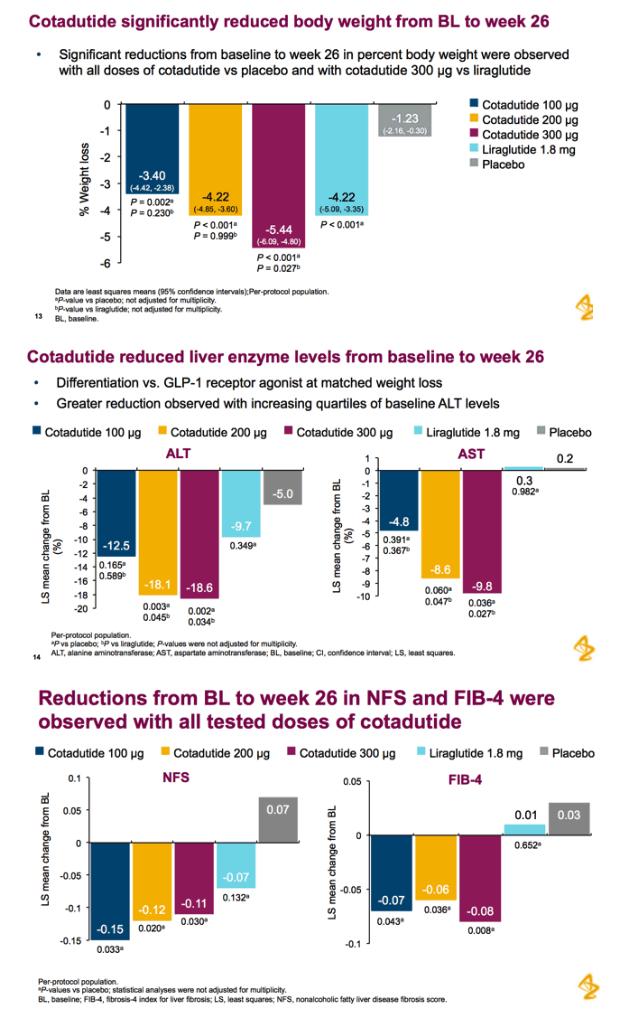
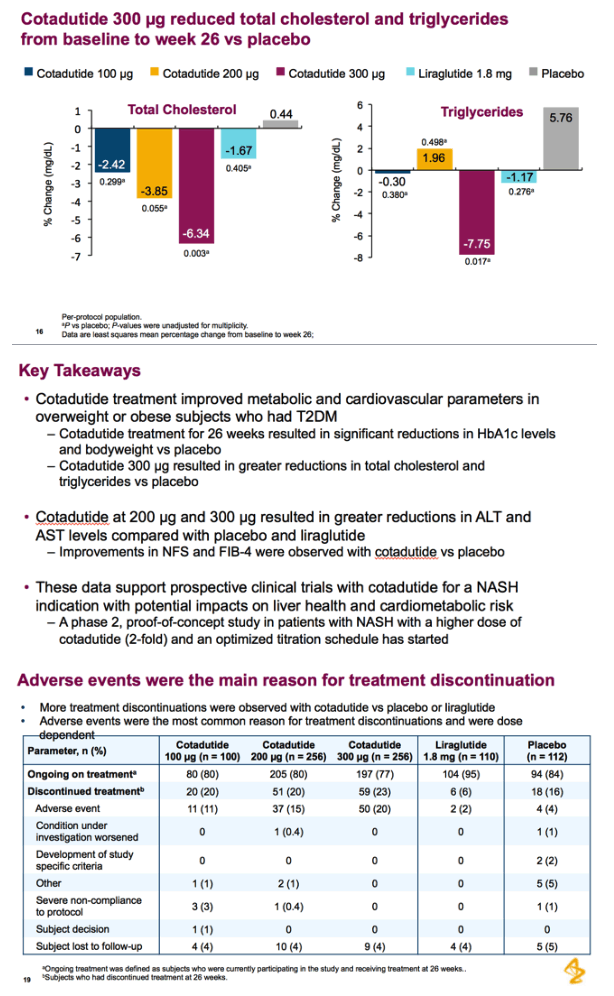
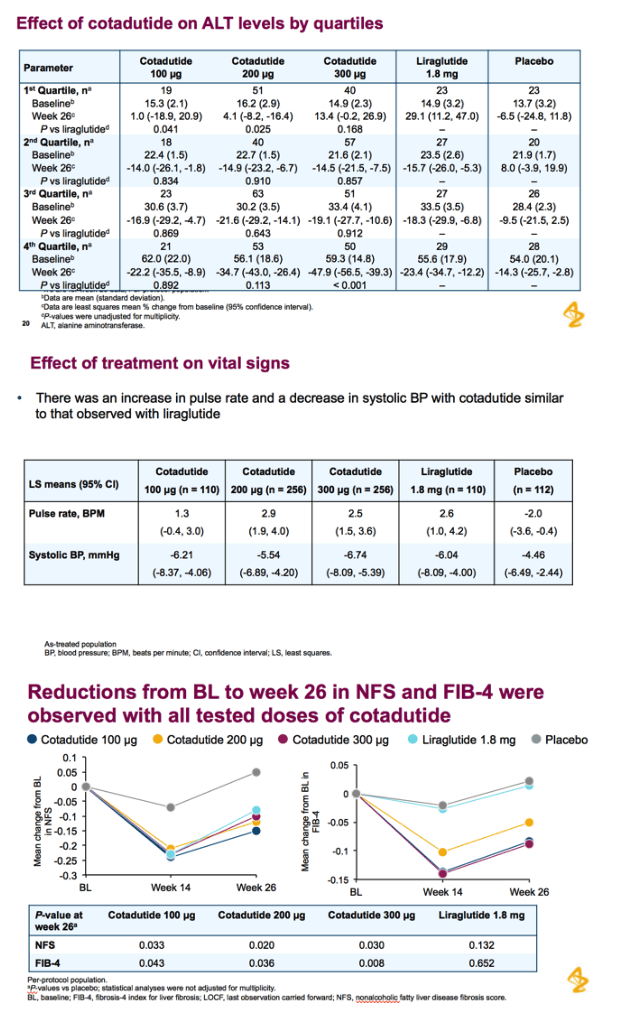
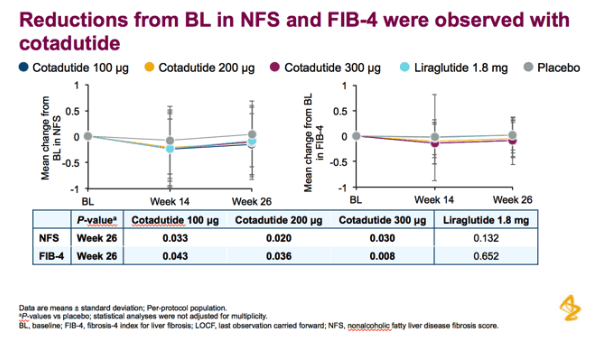
Patients received cotadutide (cohort 1, n=26; cohort 2, n=20) or placebo (cohort 1, n=13; cohort 2, n=6). Significant reductions were observed with cotadutide vs placebo in glucose AUC0-4h post MMTT (LS mean [90% CI]: -21.52% [-25.68, -17.37] vs 6.32% [0.45, 12.20]; P<0.001) and body weight (-3.41% [-4.37, -2.44] vs -0.08% [-1.45, 1.28]; P=0.002). A significant increase in insulin AUC0-4h post MMTT was observed with cotadutide (19.3 mU.h/L [5.9, 32.6]; P=0.008) and GET was prolonged on day 43 with cotadutide vs placebo (t½: 117.2 minutes vs -42.9 minutes; P=0.0392).
Conclusion: These results suggest that the glucose-lowering effects of cotadutide are mediated by enhanced insulin secretion and delayed gastric emptying.
----------------------
|
| |
|
 |
 |
|
|