 |
 |
 |
| |
Inflammation, Arterial Stiffness and Directly-Acting
Antivirals in HCV and HIV/HCV, no beneft observed in this study
|
| |
| |
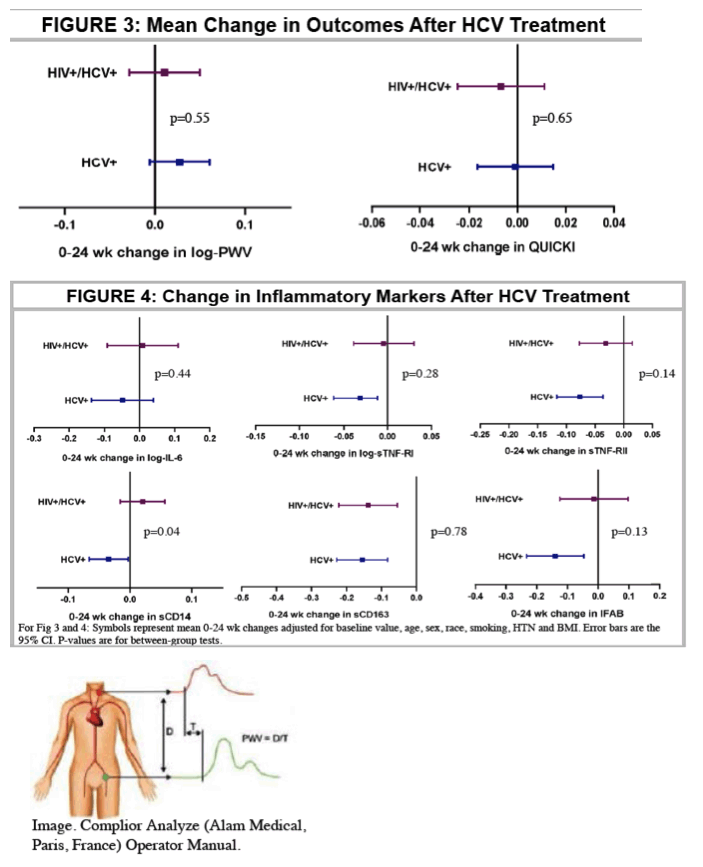
"After DAA treatment, immune activation and gut markers improved in the HCV group; no change was observed in the HIV/HCV group. Further, PWV did not improve in either group. Cardiac risk may remain elevated in HIV/HCV despite SVR with DAAs." See figure 4 below
Reported by Jules Levin
CROI 2019 March 4-7 Seattle
SonikaMalik1, RuchiBisen1, Melissa Osborn Jenkins1,3, Sanjay Gandhi1,3, Grace A McComsey2,3 and Corrilynn O Hileman1,31MetroHealth Medical Center, 2University Hospitals Cleveland Medical Center
3Case Western Reserve University School of Medicine, Cleveland, Ohio, USA

Abstract Body:
Coinfection with HCV increases cardiovascular disease (CVD) risk in HIV. Insulin resistance and heightened inflammation may contribute. While directly-acting antivirals (DAAs) improve glucose homeostasis and CVD risk in HCV-infected persons, the effect in HIV/HCV coinfection is less clear.
This is a 24-week prospective, cohort study to compare baseline and changes in aortic pulse wave velocity (PWV), glucose homeostasis (HOMA-IR), systemic inflammation [interleukin-6 (IL6), soluble tumor necrosis factor α receptors 1 and 2 (sTNF-R1 and -R2)], monocyte activation (soluble CD14 and CD163), and gut integrity [intestinal fatty acid binding protein (IFAB)] among adults with HIV, HCV, HIV/HCV or neither infection (controls) and after HCV treatment in HCV and HIV/HCV. Adults without CVD or diabetes and on stable antiretroviral therapy (HIV and HIV/HCV) were included. Pairwise comparisons of log-transformed outcome variables were made at baseline and absolute changes over 24 weeks were compared within and between groups that underwent HCV treatment. Analysis of covariance (ANCOVA) was used for adjustment.
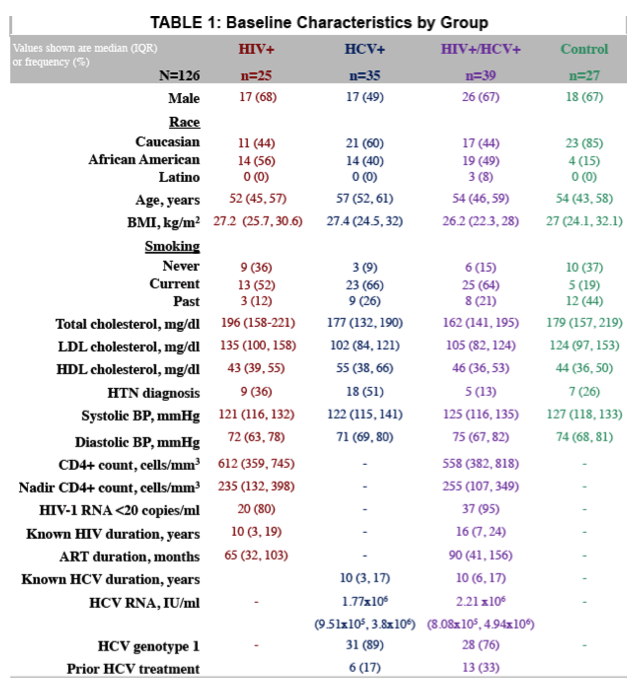
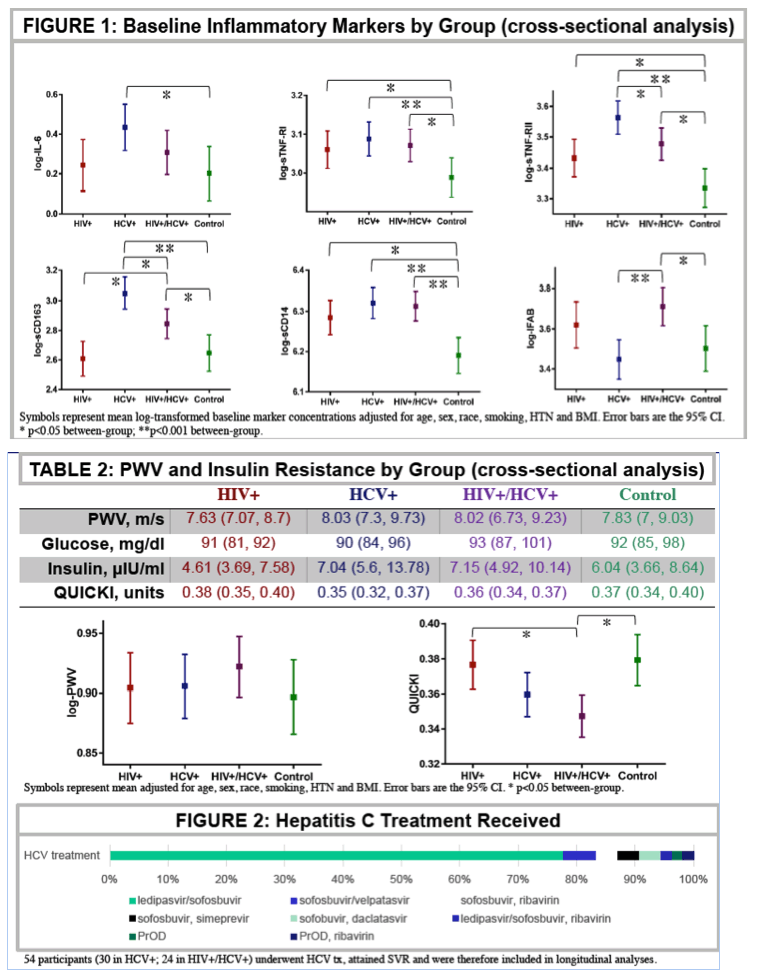
126 subjects (25 HIV, 35 HCV, 39 HIV/HCV, 27 controls) were included. 54 (30 HCV, 24 HIV/HCV) received DAAs and attained sustained virologic response (SVR). Groups were similar except HCV subjects were older (56 vs 51 years) and more likely to have HTN (51 vs 23%); controls were more likely Caucasian (85 vs 48%) and non-smokers (81 vs 38%). Of those who underwent HCV treatment, 77% initiated ledipasvir/sofosbuvir. Baseline PWV was not different among groups and 0-24 week changes were not significant within or between groups treated for HCV (p=0.46 for between group test). Baseline HOMA-IR was higher in HIV/HCV than HIV and HCV trended to be higher than controls, but did not change after DAAs (p=0.89 for between group test). Baseline IFAB and sCD163 were greater in HIV/HCV than HCV and HIV, respectively. Most inflammatory markers were higher in HCV and HIV than controls. The figure shows 0-24 week changes in the markers tested. Most markers improved in HCV, while they did not change in HIV/HCV. Changes in sTNF-R1 and sCD14 tended to be different between groups with improvements in HCV group only.
After DAA treatment, immune activation and gut markers improved in the HCV group; no change was observed in the HIV/HCV group. Further, PWV did not improve in either group. Cardiac risk may remain elevated in HIV/HCV despite SVR with DAAs.

|
| |
|
 |
 |
|
|