 |
 |
 |
| |
Real-life efficacy and safety of Glecaprevir/Pibrentasvir in a large cohort of HCV-infected patients: the MISTRAL study
|
| |
| |
Reported by Jules Levin
CROI 2019 March 4-7 Seattle
1M. Persico, 1A. Aglitti, 2M. Milella, 3C. Coppola, 4V. Messina, 5E. Claar, 6I. Gentile, 7S. De Gioia, 8P. Pierri, 9L.A. Surace, 10F. Morisco, 11P. Tundo, 12G. Brancaccio, 13G. Serviddio, 14P. Gatti, 15A.P. Termite, 16G.
Di Costanzo, 17B. Caroleo, 18R. Cozzolongo, 12N. Coppola, 19A. Longo, and 1M. Masarone.
1Unità di Medicina Interna ed Epatologia, Università di Salerno; 2Unità di Malattie Infettive, Bari; 3Unità di Medicina Interna ed Epatologia, Gragnano; 4Unità di Malattie Infettive, AORN Caserta; 5Unità di Epatologia, Ospedale Evangelico "Villa Betania", Napoli; 6Unità di Malattie Infettive, Università Federico II, Napoli; 7Unità di Medicina Interna, Ospedale "SS Annunziata",
Taranto; 8Unità di Malattie Infettive ed Epatologia, Ospedale Cotugno, Napoli; 9Centro di Medicina del Viaggiatore e delle Migrazioni, ASP Catanzaro; 10Unità di Gastroenterologia ed Epatologia, Università Federico II, Napoli; 11Unità di Malattie Infettive, Ospedale "S. Caterina Novella", Galatina; 12Unità di Malattie Infettive, Università della Campania "Luigi Vanvitelli",
Napoli; 13Centro CURE, Università di Foggia; 14Unità di Medicina Interna, Ostuni; 15Unità di Medicina Interna, Ospedale di Castellaneta, Taranto; 16Unità di Epatologia, AORN Cardarelli, Napoli; 17Unità di Geriatria, Università Mater Domini, Catanzaro; 18Ospedale S. de Bellis – IRCCS - Castellana Grotte; 19Unità di Malattie Infettive, Ospedale "Perrino", Brindisi

Program Abstract:
Data on the effectiveness and safety of Glecaprevir/Pibrentasvir for the treatment of HCV infection in a 'field-practice' scenario are still scant. This study (MISTRAL: MavIret SouTh italy ReAl Life), currently ongoing, evaluates this therapy in a large cohort of HCV-infected patients from Southern Italy.
All HCV-infected patients, consecutively treated with Glecaprevir/Pibrentasvir at 22 Centers all over Southern Italy were considered. Fibrosis was determined histologically or non-invasively, through liver stiffness measurement. We report here baseline characteristics of patients and available data on end of treatment (EOT). Efficacy of treatment was defined as undetectable HCV-RNA in patients' blood sample obtained twelve weeks after the end of therapy (sustained virological response).
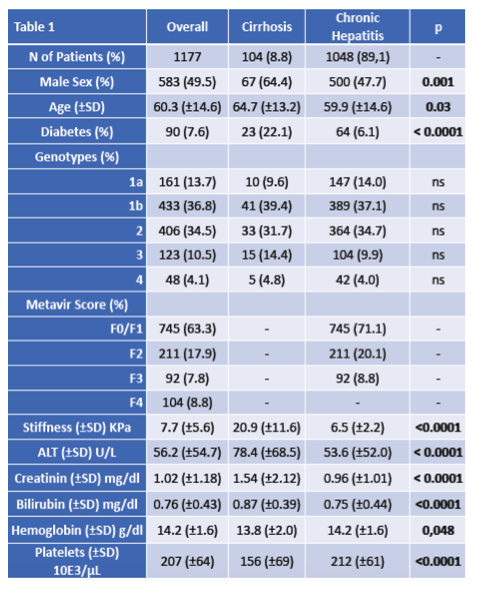
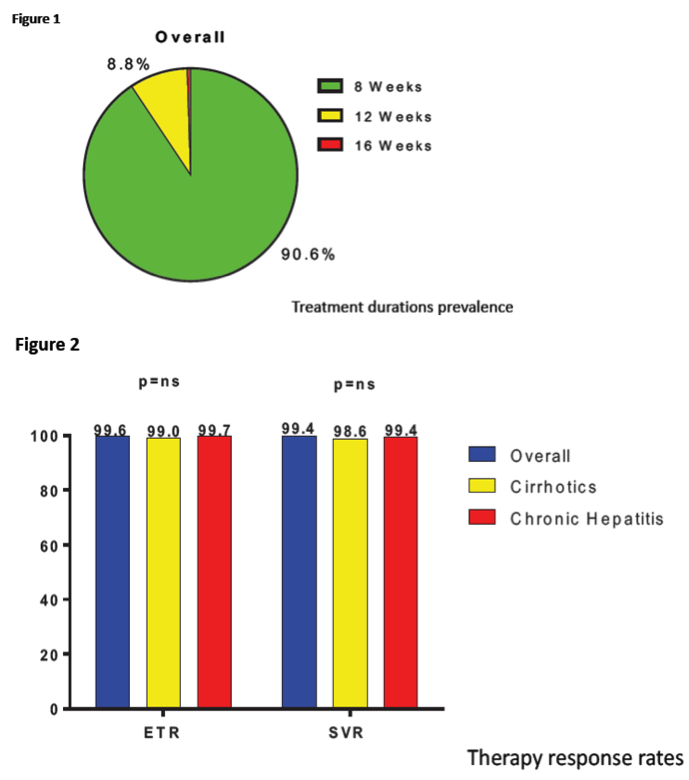
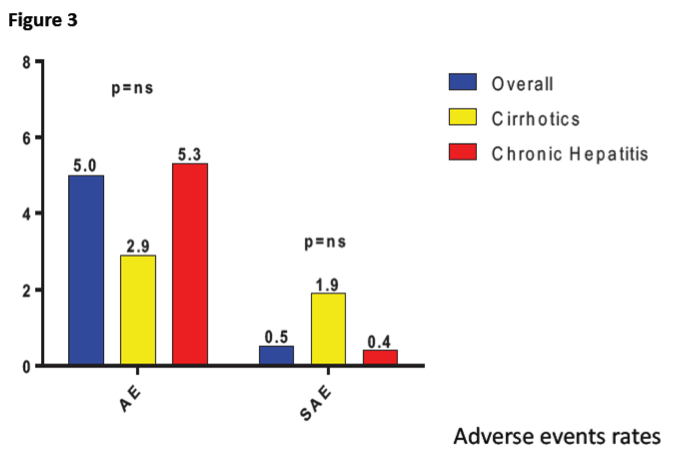
In total, 1178 patients were enrolled (mean age, 60±15 years; 581 males, 49.4%). Most common etiologies were HCV 1b (n=432; 36.8%) and HCV 2 (n=405; 34.5%). One hundred and twenty-three patients (10.5%) were infected from genotype-3 HCV. METAVIR score was F0 in 269 patients (22.9%), F1 in 477 (40.6%), F2 in 209 (17.8%), F3 in 92 (7.8%) and F4 in 103 (8.8%). The wide majority of patients showed normal (CKD1; n=572, 48.7%) or mildly impaired (CKD2; n=472, 40.2%) renal function; 28 subjects had kidney failure (CKD5; 2.4%). Ninety patients (7.7%) were diabetics. Laboratory parameters were as follows: creatinine, 1.02±1.17 mg/dl; bilirubin, 0.76±0.44 mg/dl; hemoglobin, 14.2±1.6 g/dl; platelets, 206874±64111/μl; ALT 45±38 U/L and AST 55±56 U/L. Most patients (n=918; 78.1%) were treatment-naïve. Planned duration of Glecaprevir/Pibrentasvir treatment was 8 weeks for 1067 patients (90.8%), 12 weeks for 102 (8.7%) and 16 weeks for 5 (0.4%). At the time of analysis, data concerning EOT were available for 1178 patients 100% of the total). Almost all of them reached EOT (99.5%). Data on sustained virological response at 12 weeks after EOT are not complete at the time of the present analysis we then here reported data on 885 patients showing a prevalence of 99.3% of SVR. SAE, not related to the drug, were documented in 1% of patients and 8.5% of AE (mostly pruritus).
The large MISTRAL study, conducted in a field-practice scenario, provides a still better prevalence, compared to registration trial, of SVR confirming the extraordinary efficacy and safety of Glecaprevir/Pibrentasvir association also for only 8 weeks treatment. Complete final results will be presented at the CROI meeting.

|
| |
|
 |
 |
|
|