 |
 |
 |
| |
SUSTAINED HIV-1 REMISSION FOLLOWING HOMOZYGOUS CCR5 DELTA32 ALLOGENIC HSCT
|
| |
| |
HIV-1 remission following CCR5Δ32/Δ32 haematopoietic stem-cell
Transplantation
Webcast
http://www.croiwebcasts.org/console/player/41065?mediaType=slideVideo&




Second patient free of HIV after stem-cell therapy
The breakthrough suggests first case was not a one-off and could pave way for future treatments.
A person with HIV appears to be free of the virus after receiving a stem-cell transplant that replaced their white blood cells with HIV-resistant versions. The patient is only the second person ever reported to have been cleared of the virus using this method. But researchers warn that it is too early to say that they have been cured.
The patient - whose identity hasn't been disclosed - was able to stop taking antiretroviral drugs, with no sign of the virus returning 18 months later. The stem-cell technique was first used a decade ago for Timothy Ray Brown, known as the 'Berlin patient', who is still free of the virus.
So far, the latest patient to receive the treatment is showing a response similar to Brown, says Andrew Freedman, a clinical infectious diseases physician at Cardiff University who was not involved in the study. "There's good reason to hope that it will have the same result," he says.
Like Brown, the latest patient also had a form of blood cancer that wasn't responding to chemotherapy. They required a bone-marrow transplant, in which their blood cells would be destroyed and replenished with stem cells transplanted from a healthy donor.
But rather than choosing just any suitable donor, the team, led by Ravindra Gupta, an infectious diseases physician at the University of Cambridge, UK, picked a donor who had two copies of a mutation in the CCR5 gene, which gives people resistance to HIV infection. This gene codes for a receptor which sits on the surface of white blood cells involved in the body's immune response. Normally, the HIV binds to these receptors and attacks the cells, but a deletion in the CCR5 gene stops the receptors from functioning properly. About 1% of people of European descent have two copies of this mutation and are resistant to HIV infection.
Gupta's team describes the results in a paper due to be published in Nature on 5 March. The researchers report that the transplant successfully replaced the patient's white blood cells with the HIV-resistant variant. Cells circulating in the patient's blood stopped expressing the CCR5 receptor, and in the lab, the researchers were unable to re-infect these cells with the patient's version of HIV.
The team found that the virus completely disappeared from the patient's blood after the transplant. After 16 months, the patient stopped taking antiretroviral drugs, the standard treatment for HIV. In the latest follow-up, 18 months after stopping medication, there is still no sign of the virus.
Aggressive treatment
Gupta says that it's not yet possible to say whether the patient has been cured. This can only be demonstrated if the patient's blood remains HIV-free for longer, he says.
But the study does suggest that Brown's successful treatment 10 years ago wasn't just a one-off. Gupta says that the latest patient received a less aggressive treatment than Brown to prepare for the transplant. The new patient was given a regimen consisting of chemotherapy alongside a drug that targets cancerous cells, while Brown received radiotherapy across his entire body in addition to a chemotherapy drug.
That suggests that, to be successful, stem-cell transplants in HIV patients would not necessarily need aggressive treatments that might have particularly severe side effects, says Gupta. "The radiation really does knock the bone marrow and make you very sick."
Graham Cooke, a clinical researcher at Imperial College London, points out that this kind of treatment wouldn't be suitable for most people with HIV - who don't have cancer and so don't need a bone-marrow transplant, which is a serious procedure that can sometimes have fatal complications. "If you're well, the risk of having a bone-marrow transplant is far greater than the risk of staying on tablets every day," he says. Most people with HIV respond well to daily antiretroviral treatment.
But Cooke adds that for those who need a transplant to treat leukaemia or other diseases, it seems reasonable to try and find a donor with the CCR5 mutation, which wouldn't add any risk to the procedure.
Gero Hütter, who led Brown's treatment and is now medical director of stem-cell company Cellex in Dresden, Germany, agrees that this kind of treatment could only ever be used for a small group of patients. But he hopes that the paper will stimulate a renewed interest in gene therapies that target CCR5, which could be applied to a much broader group. "The real breakthrough, we are still waiting for," he says.
------------------------
29LB SUSTAINED HIV-1 REMISSION FOLLOWING HOMOZYGOUS CCR5 DELTA32
ALLOGENIC HSCT
Ravindra K Gupta1, Sultan Abduljawad1, Laura McCoy1, H P. Mok2, Dimitra
Peppa1, Helen Lee2, Eleni Nastouli3, Jonathan Lambert3, Matthew Pace4, John
Frater4, Andrew Lever2, Simon Edwards5, Eduardo Olavarria6, Ian Gabriel6, for the
CHERUB and ICISTEM Study Groups
1University College London, London, UK, 2Cambridge University, Cambridge,
UK, 3University College London Hospitals NHS Trust, London, UK, 4University
of Oxford, Oxford, UK, 5Mortimer Market Centre, London, UK, 6Imperial College
Healthcare NHS Trust, London, UK
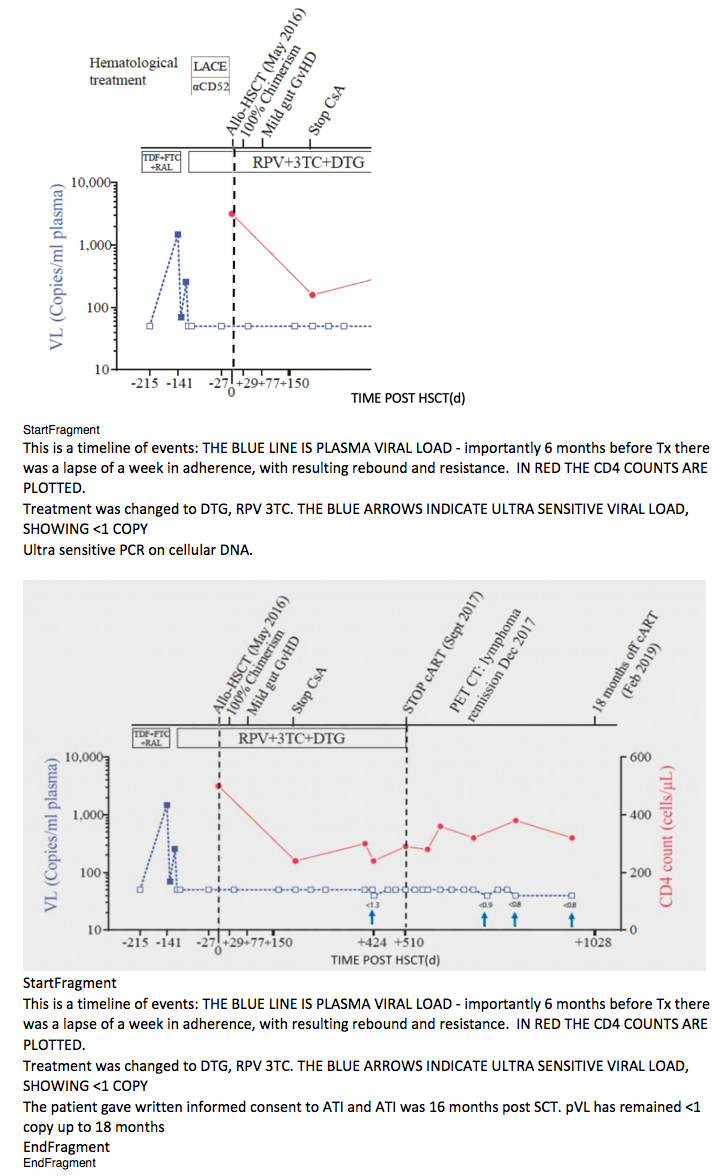
Background: The "Berlin Patient" underwent 2 consecutive HSCTs with total body irradiation. It is unclear which aspects of treatment contributed to this only known case of HIV cure. We report an HIV-infected male diagnosed with Hodgkin's Lymphoma (HL) who underwent allogenic HSCT using a homozygous CCR5d32 donor. Nadir CD4 was 290 cells/mm and baseline VL 180,000 copies/ml. ART (TDF/FTC/EFV) was started in 2012. During episodes of ART interruption viral rebound and selection of NRTI resistance was seen. HL was refractory to 1st line chemotherapy and multiple salvage regimens. An unrelated CCR5d32 homozygous donor was identified with one allelic mismatch at HLA-B. Conditioning was initiated with Lomustine, cyclophosphamide, Ara-C and etoposide followed by 3.6 million CD34+ cells/kg. In vivo T-cell depletion employed anti–CD52 and GvHD prophylaxis was cyclosporine and methotrexate. ART was continued throughout (Rilpivirine, 3TC, dolutegravir).
The patient developed mild gut GvHD. Full donor chimerism was maintained in blood. Six months post-HSCT complete remission was observed.
Methods: Co-receptor tropism was predicted with Geno2Pheno based on single genome sequencing (SGS). Post-HSCT PBMC were analysed by ddPCR and qPCR. Infectious virus was repeatedly analysed by qVOA. Isolated CD4 T cells were experimentally infected with X4 and R5 HIV.
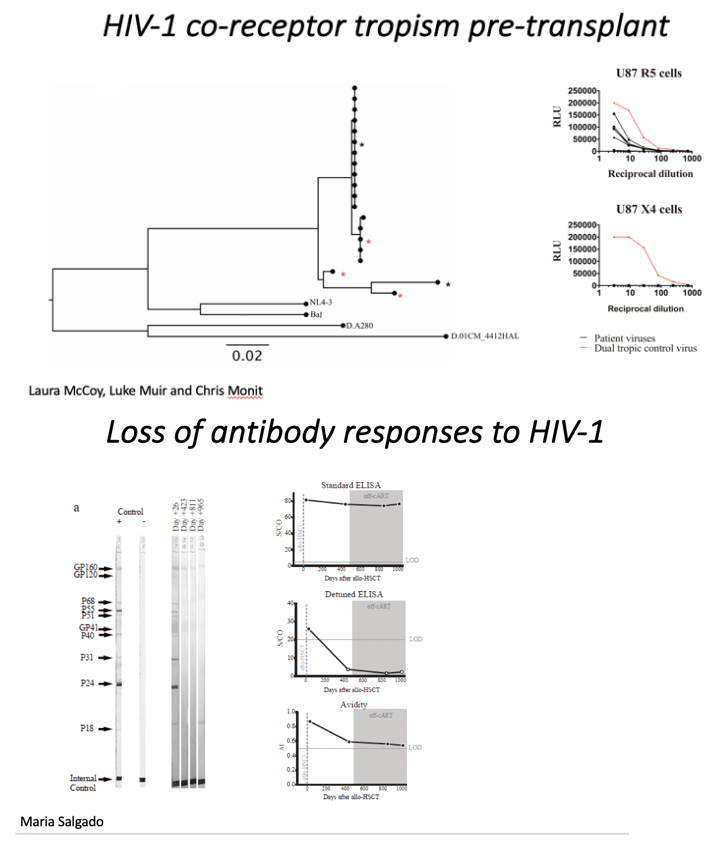
Results: SGS from pre-transplant PBMC identified multiple envelope clones all with predicted R5 tropism. ART was stopped 17 months post-HSCT and plasma HIV VL remained undetectable ( <1.4 copies/ml) at 33 months. ART drugs were not detectable in plasma by LC-MS. Total HIV DNA in CD4+ T-cells at 33 months showed 2 positive droplets in 1 out of 8 replicates (ddPCR HIV LTR, 10^6 cells tested) and no signal in qPCR (<0.69 HIV-gag and <0.65 HIV-LTR copies/million cells). At 16 months post transplant HIV-specific Western blot was positive while p24/p31 bands were absent. VITROS detuned and avidity analysis revealed low quantity and quality of HIV antibody titers. At three time points post-HSCT qVOA showed no reactivatable virus using a total of 24 million resting CD4+ T cells. Post-transplant CD4+ T cells did not express CCR5 and were susceptible in vitro to X4- but not R5-tropic virus.
Conclusion: Absence of viral rebound was observed for 16 months following ART interruption at 17 months after single allogeneic CCR5-d32 HSCT using a no irradiation approach with only mild GvHD. To our knowledge this is the longest adult HIV remission observed since the Berlin patient.
-----------------------
Letter
doi:10.1038/s41586-019-1027-4
HIV-1 remission following CCR5Δ32/Δ32 haematopoietic stem-cell
transplantation
Ravindra K Gupta, Sultan Abdul-jawad, Laura E McCoy, Hoi Ping Mok, Dimitra Peppa, Maria Salgado,
Javier Martinez-Picado, Monique Nijhuis, Annemarie M.J. Wensing, Helen Lee, Paul Grant, Eleni Nastouli, Jonathan Lambert,
Matthew Pace, Fanny Salasc, Christopher Monit, Andrew Innes, Luke Muir, Laura Waters, John Frater, Andrew ML Lever,
SG Edwards, Ian H Gabriel & Eduardo Olavarria
Nature is providing this early version of the typeset paper as a service to our customers.
The text and figures will undergo copyediting and a proof review before the paper is published in its final form. Please note
that during the production process errors may be discovered which could affect the content, and all legal disclaimers apply.
HIV-1 cure remains elusive with only one reported case a decade ago1,2. Termed the 'Berlin patient', the individual underwent two allogeneic haematopoietic stem-cell transplantation (allo-HSCT) procedures using a donor with a homozygous mutation in the HIV coreceptor CCR5 (CCR5Δ32/Δ32) to treat his acute myeloid leukaemia. Total body irradiation was given with each HSCT. Critically, it is unclear which treatment or patient parameters contributed to this only documented case of long-term HIV remission. Here we show that HIV-1 remission may be possible with a less aggressive and toxic approach. An HIV-1-infected adult underwent allo-HSCT for Hodgkin's lymphoma using cells from a CCR5Δ32/Δ32 donor. He experienced mild gut graft versus host disease. Antiretroviral therapy was interrupted 16 months after transplantation. HIV-1 remission has been maintained through a further 18 months. Plasma HIV-1 RNA has been undetectable at less than 1 copy per millilitre along with undetectable HIV-1 DNA in peripheral CD4 T lymphocytes. Quantitative viral outgrowth assay from peripheral CD4 T lymphocytes shows no reactivatable virus using a total of 24 million resting CD4 T cells. CCR5-tropic, but not CXCR4-tropic viruses were identified in HIV-1 DNA from CD4 T cells of the patient prior to transplant. CD4 T cells isolated from peripheral blood post-transplant did not express CCR5 and were only susceptible to CXCR4-tropic virus ex vivo. HIV-1 Gag-specific CD4 and CD8 T cell responses were lost after transplantation, whereas cytomegalovirus (CMV)-specific responses were detectable. Likewise, HIV-1-specific antibodies and avidities fell to levels comparable to those in the Berlin patient following transplantation. Although at 18 months post-treatment interruption it is premature to conclude that this patient has been cured, these data suggest that single allo-HSCT with homozygous CCR5Δ32 donor cells may be sufficient to achieve HIV-1 remission with reduced intensity conditioning and no irradiation, and the findings further support the development of HIV remission strategies based on preventing CCR5 expression.
The HIV-1 epidemic continues with nearly 37 million infected.3 Although over 21 million are accessing lifelong antiretroviral therapy (ART)3, drug resistant HIV in both untreated4 and treated5,6 individuals is significant in low and middle-income countries and sustainability of ART programs is uncertain.7 Drug-free durable HIV-1 suppression is therefore an urgent global priority.
Thus far, the only documented case of sustained HIV remission is the 'Berlin Patient', who received two allogeneic hematopoietic stem cell transplantations (allo-HSCT) using cells from a homozygous CCR5Δ32 (CCR5Δ32/Δ32) donor1. This 32-base pair deletion prevents CCR5 expression rendering these cells resistant to infection with HIV variants utilising the CCR5 co-receptor8. The only other case of an HIV-infected patient transplanted with CCR5Δ32/Δ32 cells who interrupted ART was the 'Essen Patient'9. In this case in which ART was interrupted one week before allo-HSCT, a rapid viral rebound of a pre-existing minority HIV-1 variant able to infect cells via the alternative CXCR4 co-receptor was observed three weeks later9,10. Such pre-existing CXCR4 variants were not observed in the 'Berlin patient'.11 Three other cases transplanted with wildtype CCR5 cells experienced a viral rebound 12, 32 or 41 weeks after ART interruption despite profound reduction of the HIV reservoir.12,13
We report an individual diagnosed with HIV infection in 2003, with a CD4 nadir of 290 cells/mm3 and baseline HIV-1 plasma viral load (pVL) of 180,000 copies/ml. ART was initiated with tenofovir disoproxil fumarate (TDF), emtricitabine (FTC) and efavirenz (EFV) in 2012.
In December 2012 Stage IVB (nodular sclerosing) Hodgkin's Lymphoma (HL) was diagnosed. HL was refractory to first-line chemotherapy (ABVD) and a number of salvage regimens including ESHAP, anti-CD30 monoclonal antibody (Brentuximab) and mini-LEAM were used. ART was switched to TDF/FTC/raltegravir (RAL) during periods of chemotherapy for HL; there was a 5-day episode of ART interruption in late 2015 with HIV-1 pVL of 1,500 copies/ml that did not reach viral set-point. Based on resistance mutations K65R and M184V in reverse transcriptase as well as E157Q in integrase, the regimen was switched to rilpivirine (RPV), lamivudine (3TC) and dolutegravir (DTG), with viral suppression subsequently achieved.
Mobilisation of autologous peripheral blood stem cells failed despite the use of CXCR4 antagonists, thereby precluding standard autologous HSCT. A complete metabolic remission by CT/PET criteria was achieved with IGEV chemotherapy in March 2016.
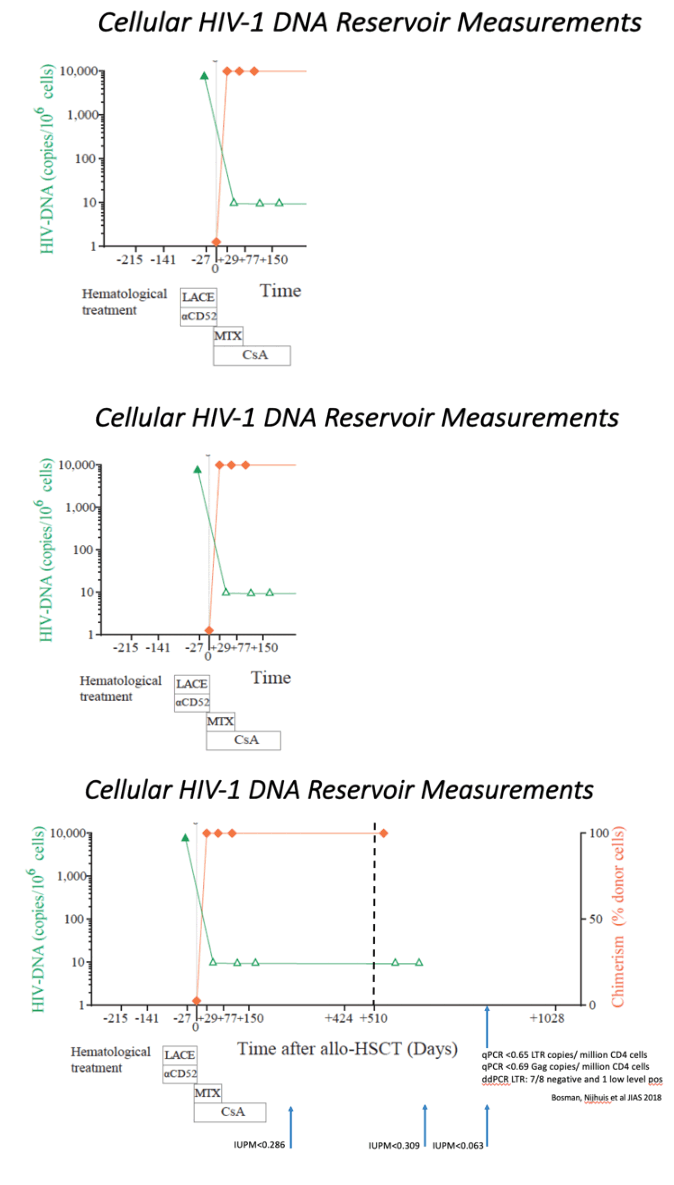
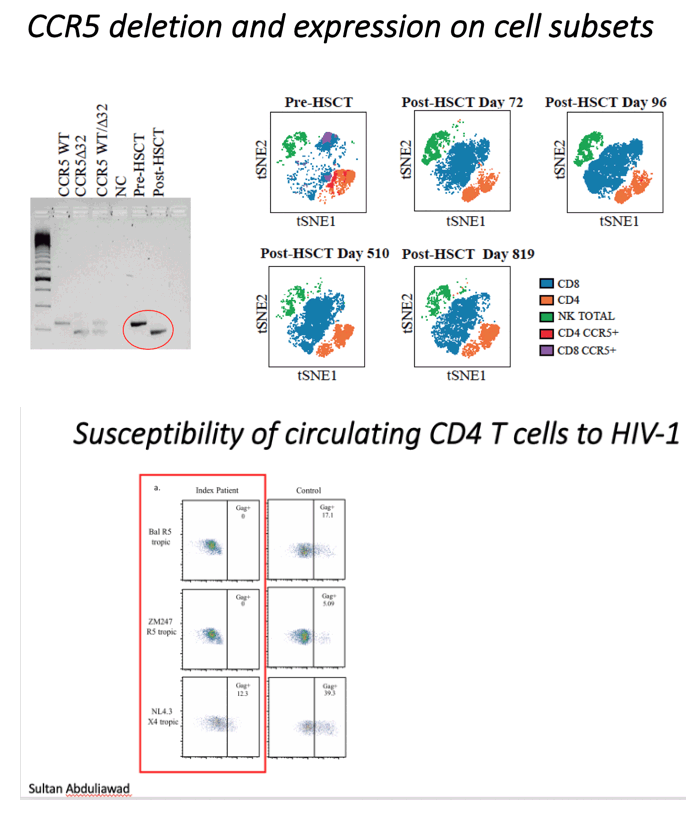
An unrelated (9/10) donor was identified from an international registry with one allelic mismatch at HLA-B by high-resolution HLA typing that also was CCR5Δ32/Δ32 (Extended data table 1 and methods). No fully matched donors were identified in the registry. The patient underwent conditioning with Lomustine, Cyclophosphamide, Ara-C and Etoposide (LACE) followed by infusion of 3.6 Å~ 106 CD34+ cells/kg. In vivo T-cell depletion employed anti–CD52 (Alemtuzumab), 10 mg daily for 5 days (days -7 to -3) and GvHD prophylaxis used Cyclosporine-A (CsA) with a short-course of methotrexate (MTX). ART was continued throughout with RPV/3TC/DTG (Figure 1a). Allo-HSCT was relatively uncomplicated and the patient was discharged on Day+31. Both Epstein-Barr Virus (EBV) and cytomegalovirus (CMV) reactivation occurred at day +85 requiring treatment with anti-CD20 monoclonal antibody (Rituximab) and ganciclovir respectively. At day+77 the patient presented with fever and gastrointestinal symptoms. Gastric, duodenal and colonic biopsies were consistent with grade 1 GvHD, which resolved without intervention. Full-donor chimerism was achieved in the whole leukocyte and in CD3+ T cell fractions from day +30 and maintained in both cell fractions throughout (Figure 1b). Host genotype was CCR5wt/wt before allo-HSCT, and became CCR5Δ32/Δ32 after transplant (Figure 1c), with loss of CCR5 surface expression from circulating CD4 and CD8 T cells (Figure 1d). At +180 days post-transplant CsA was discontinued. CT/PET scan at +120 days and +365 days post-transplant confirmed complete metabolic remission with no subsequent relapse. Post-transplant white cell counts and lymphocyte subsets returned to pre-transplant levels (extended data figure 1), except for CD4 counts which have been slower to recover (Figure 1a).
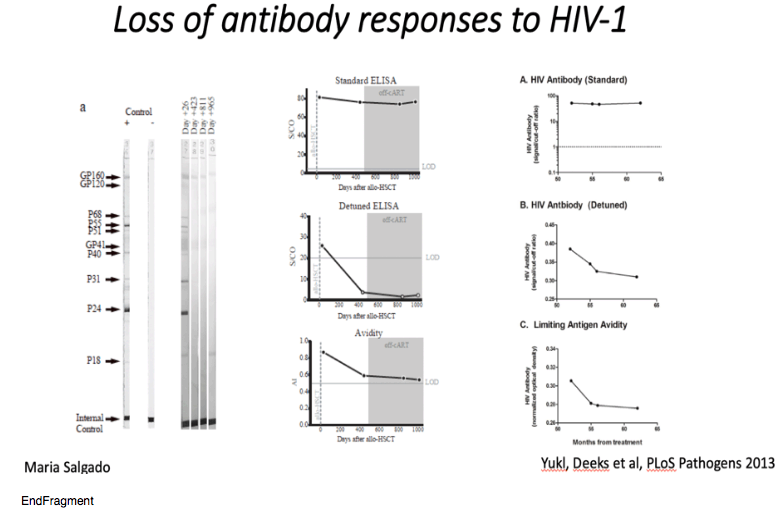
ART was maintained post-HSCT and analytical treatment interruption (ATI) was initiated at day +510 (September 2017). Weekly plasma viral load was performed for the first 3 months and then monthly thereafter. HIV-1 pVL remained undetectable thereafter with limit of detection (LOD) <1 copy RNA/ml (Figure 1a). Plasma concentrations of TDF, 3TC and DTG were negative by HPLC at day +648 and a panel of all currently available antiretroviral drugs tested negative by LC-MS at +973 days. Total PBMC associated HIV-1 DNA fell to below the limit of detection after transplant (Figure 1b). Total DNA in CD4+ T cells at day +876 was undetectable in all replicates by ultra-sensitive qPCR (<0.65 HIV LTR copies/million cells and <0.69 HIV-1 Gag copies/million cells) and in 7/8 replicates of the ultra-sensitive HIV-1 LTR ddPCR14; in one replicate a low-level signal was observed. Such occasional positive signals were also observed in the Berlin patient15 and may reflect a false ddPCR signal, potential contamination, or evidence of very low levels of persistence of HIV infected cells that either did not harbor fully replication competent virus or were unable to lead to recrudescence given that the vast majority of target cells are incapable of being infected with this patient's HIV CCR5 tropic variants (Figure 2). HIV-1 DNA and RNA were also repeatedly undetectable in whole blood when tested with SAMBA II, a CE marked point-of-care isothermal amplification method (LOD: 284 copies/ml; 95% CI: 214-378 copies/ml)16.
Blood was obtained for a modified Quantitative Viral Outgrowth Assay (QVOA)17,18 at three time points post-HSCT at day +217 (on ART), and days +678 and +876 (off ART). QVOA was undetectable on all occasions, giving a reservoir estimation in infectious units per million (IUPM) resting CD4 T cells of <0.286, <0.309 and <0.063 respectively. Pooling the results from the three qVOA tests on a total of 24 million resting CD4 T cells gives an estimate of <0.029 IUPM.
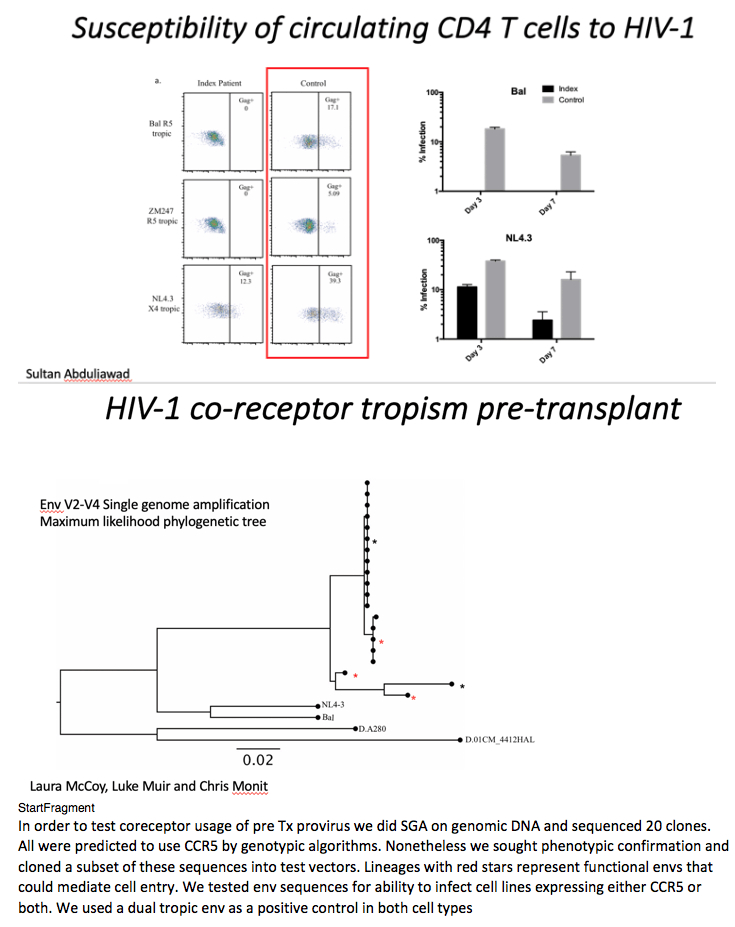
We next sought to confirm that the post-transplant CD4 cells lacked expression of CCR5 and were resistant to HIV-1 infection (Figure 2 and Extended data Figure 2). These CD4 T cells from the study patient were challenged in vitro with the CCR5-tropic viruses Ba-L and ZM247 and productive HIV-1 infection over 7 days was measured by (i) intracellular staining for HIV-1 p24 protein (Figure 2a-b) and (ii) infectivity of culture supernatants on indicator cell lines that respond to expression of HIV Tat protein (Extended data Figure 2). In contrast to a HIV-negative donor, post-transplant cells from the study patient could not be infected with either CCR5 tropic virus (Figure 2a-b). The study patient cells were then challenged with the canonical CXCR4-tropic HIV-1NL4-3. As expected, infection was observed in cells and supernatants from both the patient and an HIV-negative donor (control) (Figure 2a-b and extended data Figure 2).
In order to determine whether the study patient was infected with CCR5- or CXCR4-using virus (or both), we deep-sequenced the V3 loop in HIV-1 envelope (the key determinant of coreceptor usage) and computational algorithms predicted CCR5-tropism19,20 For phenotypic verification we first performed single genome sequencing (SGS) of gp120 from pre-transplant PBMC by limiting dilution PCR21,22. (Figure 2c). These single genomes were cloned into expression vectors and used to generate virions pseudotyped with patient derived gp120 envelope protein (Figure 2d)21, before infection of indicator cells expressing either CCR5 or CXCR4. As predicted from the genotype, robust infection was observed in CCR5 but not CXCR4 expressing cells (Figure 2d).
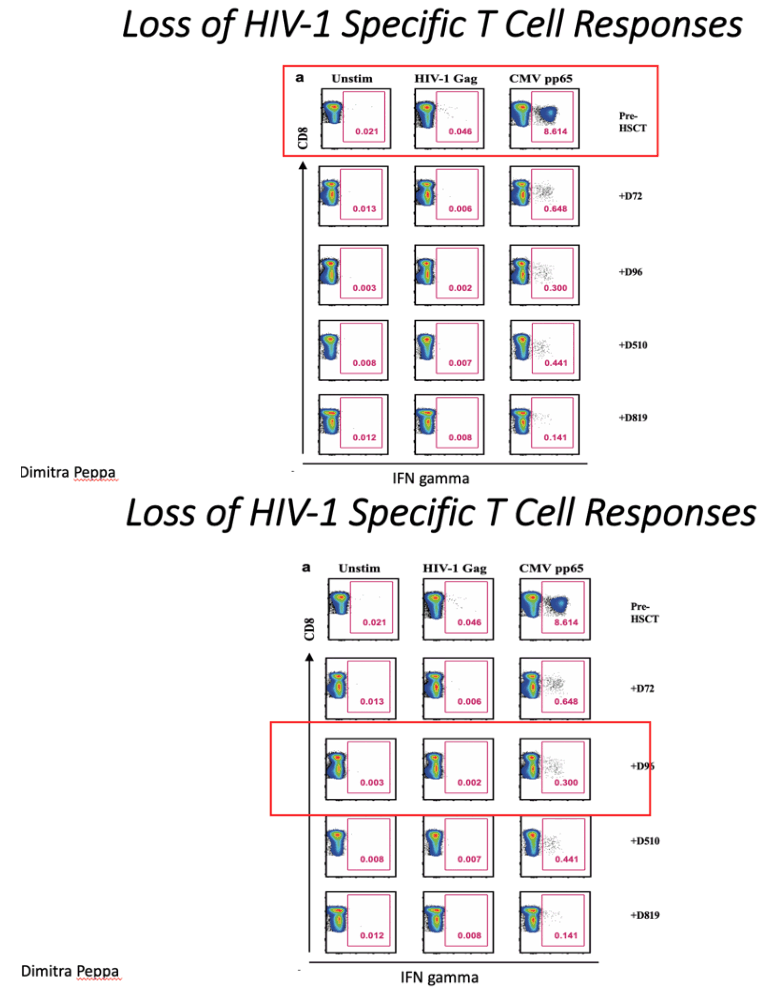
Analyses of both antibody and T cell responses were undertaken in order to further investigate absence of persistent HIV-1 infection and antigenic stimulation. Western blot analysis of antibodies demonstrated loss of p24, p31 and multiple other bands between pre- and post-transplant time points in a similar pattern seen previously in other transplanted patients who have remained on ART23 (Figure 3a and Extended Data table 2), whilst antibodies to envelope protein gp160 and standard ELISA for Env antibodies persisted (Figure 3a-c). Low-sensitivity VITROS analysis and antibody avidity assays were also consistent with loss of HIV antigen following allo-HSCT (Figure 3d-e)15. CD8 and CD4 T cell virus-specific responses were determined following stimulation with HIV-1 Gag and CMV pp65 overlapping peptide pools (Figure 4a). Small Gag-virus specific T-cell responses were identified by intracellular staining and multifunctional responses (IFN-γ, CD107a, TNF-α and IL-2) were detected prior to HSCT (day -35) but not at days +72, 96, 510 and 819 after the procedure (Figure 4a-b). No HIV-1 specific T cell responses were detected to Nef, Pol and Env peptide pools at days +96 and +819 post-transplant (Extended data Figure 3). By contrast CMV- specific T-cell responses were detected both before and after HSCT, albeit at reduced frequencies (Figure 4a-c).
Both antibody and T cell responses observed here are highly reminiscent of observations in the Berlin patient.1,15 Important similarities between the two cases were CCR5-tropic HIV-1 infection and receipt of a CCR5Δ32/Δ32 transplant. The GvHD prophylaxis was very similar and utilized standard regimens (CsA + MMF versus CsA + MTX) that in randomized studies have shown similar outcomes.24 In both cases there was probable mild GvHD, which may have contributed to the loss of HIV-infected cells. Finally, both achieved and maintained full-donor chimerism in peripheral blood that might have contributed to reduced reservoir size23,25.
Notable differences were that before allo-HSCT our patient was homozygous CCR5 wildtype as compared to the heterozygous CCR5WT/Δ32 genotype observed in the "Berlin Patient". Our patient received a reduced intensity conditioning regimen consisting exclusively of chemotherapy agents with known activity against lymphoma26 while the Berlin patient received total body irradiation in conjunction with cyclophosphamide as the conditioning regimen. For lymphodepletion our patient received anti-CD52 Campath while the Berlin patient was treated with antithymocyte globulin. Our patient was treated with a short course of anti CD20 for EBV reactivation. There has been recent interest in the potential for anti-B cell therapy to disrupt B cell follicles that are known to harbour persistent HIV in lymphoid tissues27, though we think this mechanism is unlikely to be related to remission observed here. Finally, this patient achieved full remission after a single allo-HSCT while the Berlin patient experienced a relapse of the AML and received further chemotherapy with an anti-CD33 monoclonal conjugate before a second allo-HSCT.
In terms of a road map forward we speculate that CCR5 gene therapy strategies using stem cells could conceivably be a scalable approach to remission. As significant graft-versus-host effect is likely important, manifesting as early and sustained full donor chimerism in T cells, an autologous approach needs to achieve high levels of re placement by CCR5 depleted cells. Depletion of the CD4+ CCR5+ T cells other potential HIV reservoirs is a major challenge given the toxicities associated with anti-thymocyte globulin and anti-CD52 and the unknown role of anti-proliferative agents such as cyclosporin or methotrexate in clearing infected cells.
This report demonstrates (i) that the 'Berlin' patient was not an anomaly; (ii) that remission of HIV infection can be achieved with reduced intensity drug regimens (iii) that a single CCR5Δ32/Δ32 allo-HSCT is sufficient and (iv) that total body irradiation is not required. Our observation supports the development of HIV cure strategies based on preventing the expression of the CCR5 coreceptor.
Online content
Any methods, additional references, Nature Research reporting summaries, source
data, statements of data availability and associated accession codes are available at
https://doi.org/10.1038/s41586-019-1027-4.
Received: 7 February 2019; Accepted: 26 February 2019;
Published online 5 March 2019.
1. Hutter, G. et al. Long-term control of HIV by CCR5 Delta32/Delta32 stem-cell
transplantation. N Engl J Med 360, 692-698, doi.org/10.1056/
NEJMoa0802905 (2009).
2. Allers, K. et al. Evidence for the cure of HIV infection by CCR5Delta32/Delta32
stem cell transplantation. Blood 117, 2791-2799, doi.org/10.1182/
blood-2010-09-309591 (2011).
3. UNAIDS. UNAIDS DATA, http://www.unaids.org/en/resources/
documents/2017/20170720_Data_book_2017> (2017).
4. Gupta, R. K. et al. HIV-1 drug resistance before initiation or re-initiation of
first-line antiretroviral therapy in low-income and middle-income countries: a
systematic review and meta-regression analysis. Lancet Infect Dis 18, 346-355,
doi.org/10.1016/S1473-3099(17)30702-8 (2018).
5. TenoRes Study, G. Global epidemiology of drug resistance after failure of WHO
recommended first-line regimens for adult HIV-1 infection: a multicentre
retrospective cohort study. Lancet Infect Dis 16, 565-575, doi.org/10.1016/
S1473-3099(15)00536-8 (2016).
6. Barth, R. E., van der Loeff, M. F., Schuurman, R., Hoepelman, A. I. & Wensing, A. M.
Virological follow-up of adult patients in antiretroviral treatment programmes
in sub-Saharan Africa: a systematic review. Lancet Infect Dis 10, 155-166
(2010).
7. Avert. FUNDING FOR HIV AND AIDS, https://www.avert.org/professionals/
hiv-around-world/global-response/funding> (2017).
8. Simmons, G. et al. Primary, syncytium-inducing human immunodeficiency
virus type 1 isolates are dual-tropic and most can use either Lestr or
CCR5 as coreceptors for virus entry. Journal of virology 70, 8355-8360
(1996).
9. Kordelas, L. et al. Shift of HIV tropism in stem-cell transplantation with CCR5
Delta32 mutation. N Engl J Med 371, 880-882, doi.org/10.1056/
NEJMc1405805 (2014).
10. Verheyen, J. et al. Rapid rebound of a preexisting CXCR4-tropic HIV variant after
allogeneic transplantation with CCR5 delta32 homozygous stem cells. Clinical
infectious diseases: an official publication of the Infectious Diseases Society of
America, doi.org/10.1093/cid/ciy565 (2018).
11. Symons, J. et al. Dependence on the CCR5 coreceptor for viral replication
explains the lack of rebound of CXCR4-predicted HIV variants in the Berlin
patient. Clinical infectious diseases: an official publication of the Infectious
Diseases Society of America 59, 596-600, doi.org/10.1093/cid/ciu284
(2014).
12. Henrich, T. J. et al. Antiretroviral-free HIV-1 remission and viral rebound after
allogeneic stem cell transplantation: report of 2 cases. Ann Intern Med 161,
319-327, doi.org/10.7326/M14-1027 (2014).
13. Cummins, N. W. et al. Extensive virologic and immunologic characterization in
an HIV-infected individual following allogeneic stem cell transplant and analytic
cessation of antiretroviral therapy: A case study. PLoS medicine 14, e1002461,
doi.org/10.1371/journal.pmed.1002461 (2017).
14. Bosman, K. J. et al. Development of sensitive ddPCR assays to reliably
quantify the proviral DNA reservoir in all common circulating HIV subtypes
and recombinant forms. J Int AIDS Soc 21, e25185, doi.org/10.1002/
jia2.25185 (2018).
15. Yukl, S. A. et al. Challenges in detecting HIV persistence during potentially
curative interventions: a study of the Berlin patient. PLoS pathogens 9,
e1003347, doi.org/10.1371/journal.ppat.1003347 (2013).
16. Ritchie, A. V. et al. Performance evaluation of the point-of-care SAMBA I and II
HIV-1 Qual whole blood tests. J Virol Methods 237, 143-149, doi.org/10.1016/j.
jviromet.2016.08.017 (2016).
17. Laird, G. M. et al. Rapid quantification of the latent reservoir for HIV-1 using a
viral outgrowth assay. PLoS pathogens 9, e1003398, doi.org/10.1371/journal.
ppat.1003398 (2013).
18. Fun, A., Mok, H. P., Wills, M. R. & Lever, A. M. A highly reproducible
quantitative viral outgrowth assay for the measurement of the replicationcompetent
latent HIV-1 reservoir. Sci Rep 7, 43231, doi.org/10.1038/
srep43231 (2017).
19. Jensen, M. A., Coetzer, M., van 't Wout, A. B., Morris, L. & Mullins, J. I. A reliable
phenotype predictor for human immunodeficiency virus type 1 subtype C
based on envelope V3 sequences. Journal of virology 80, 4698-4704, doi.
org/10.1128/JVI.80.10.4698-4704.2006 (2006).
20. Lengauer, T., Sander, O., Sierra, S., Thielen, A. & Kaiser, R. Bioinformatics
prediction of HIV coreceptor usage. Nature biotechnology 25, 1407-1410, doi.
org/10.1038/nbt1371 (2007).
21. Smith, N. M. et al. Proof-of-Principle for Immune Control of Global HIV-1
Reactivation In Vivo. Clinical infectious diseases: an official publication of the
Infectious Diseases Society of America, doi.org/10.1093/cid/civ219 (2015).
22. Watters, S. A. et al. Sequential CCR5-Tropic HIV-1 Reactivation from Distinct
Cellular Reservoirs following Perturbation of Elite Control. PloS one 11,
e0158854, doi.org/10.1371/journal.pone.0158854 (2016).
23. Salgado, M. et al. Mechanisms That Contribute to a Profound Reduction of the
HIV-1 Reservoir After Allogeneic Stem Cell Transplant. Ann Intern Med 169,
674-683, doi.org/10.7326/M18-0759 (2018).
24. Hamilton, B. K. et al. Long-term follow-up of a prospective randomized trial
comparing CYA and MTX with CYA and mycophenolate mofetil for GVHD
prophylaxis in myeloablative sibling donor hematopoietic cell transplantation. Bone
Marrow Transplant 48, 1578-1580, doi.org/10.1038/bmt.2013.89 (2013).
25. Duarte, R. F. et al. CCR5 Delta32 homozygous cord blood allogeneic
transplantation in a patient with HIV: a case report. Lancet HIV 2, e236-242, doi.
org/10.1016/S2352-3018(15)00083-1 (2015).
26. Pavlu, J. et al. LACE-conditioned autologous stem cell transplantation for
relapsed or refractory diffuse large B-cell lymphoma: treatment outcome and
risk factor analysis from a single centre. Hematol Oncol 29, 75-80, doi.
org/10.1002/hon.956 (2011).
27. Bronnimann, M. P., Skinner, P. J. & Connick, E. The B-Cell Follicle in HIV
Infection: Barrier to a Cure. Front Immunol 9, 20, doi.org/10.3389/
fimmu.2018.00020 (2018)
Acknowledgements This study was funded by a Wellcome Trust Senior
Fellowship in Clinical Science to RKG, research capability funding (RCF) from
UCLH BRC to RKG, as well as funding from Oxford and Cambridge Biomedical
Research Centres (BRC), amfAR (The Foundation for AIDS Research), through
the amfAR Research Consortium on HIV Eradication (ARCHE) program (AmfAR
109858-64-RSRL), the MRC (MR/R008698/1 to LEM and MRM008614/2
to DP). AJI is supported by an NIHR Clinical Lectureship, and acknowledges
support from the NIHR and Imperial Biomedical Research Centre (BRC). We
would like to thank the CHERUB (http://www.cherub.uk.net) and IciStem
Consortia (www.icistem.org) for support and continuous discussion of
results. We would also like to thank Nina Parmahand, Mikaila Bandara, Isobel
Jarvis, Axel Fun, Kirit Ardeshna, Alison Hill, Neha Goel, Richard Szydlo, David
Slade, Sarah Griffith and Cristina Gálvez. Águeda Hernández Rodríguez,
Victoria González Soler and Belén Rivaya Sánchez from the Microbiology
Department of the Hospital Germans Trias i Pujol. Erik van Maarseveen from
the department of Pharmacology, and Laura Huyveneers, Pauline Schipper
and Dorien de Jong from the Translational Virology Group of the department of
Medical Microbiology of the UMC Utrecht. Finally, we would also like to thank
Professor Jane Apperley, Zoe Allwood and Sandra Loaiza from Imperial College
Healthcare NHS Trust and all the nurses in the BMT Unit that looked after the
patient.
Reviewer information Nature thanks Steven Deeks, Timothy Henrich and the
other anonymous reviewer(s) for their contribution to the peer review of this
work.
PREVIEW
ACCELERATED ARTICLE PREVIEW
|
| |
|
 |
 |
|
|