 |
 |
 |
| |
Delayed Weight Gain in Immediate Versus Deferred ART Arm in START - "Antiretroviral therapy and body weight in the START (Strategic Timing of Antiretroviral Treatment) trial"
|
| |
| |
17th European AIDS Conference, November 6-9, 2019, Basel
Mark Mascolini
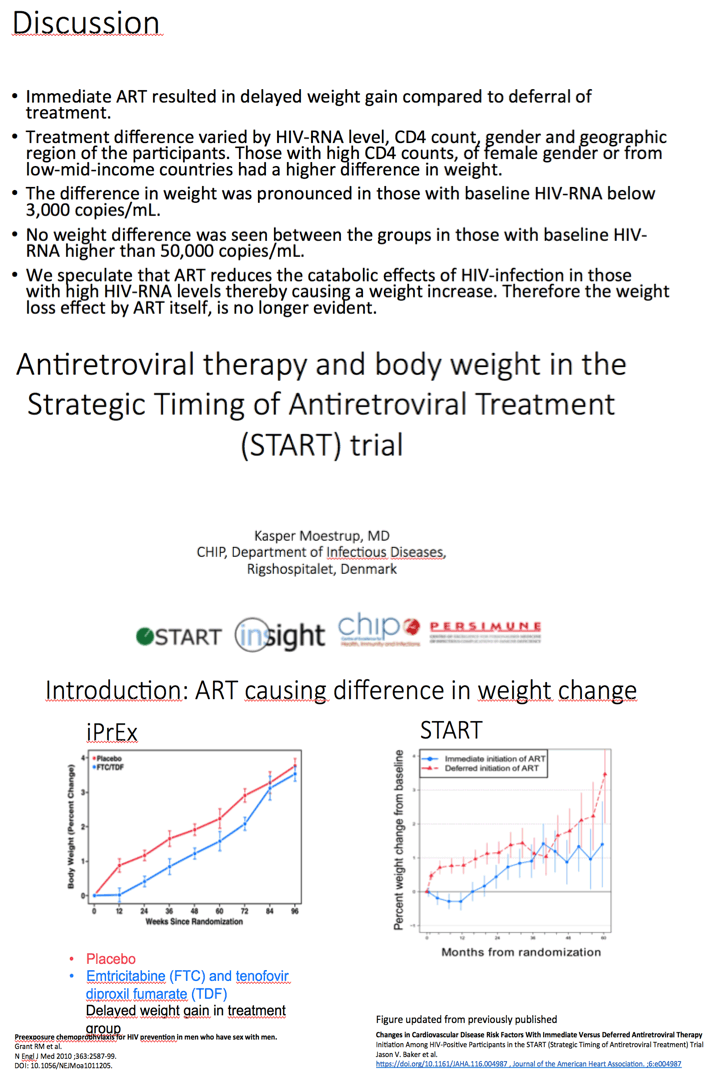
People randomized to begin immediate antiretroviral therapy (ART) in the START trial had slower weight gains than those assigned to deferred ART [1]. But weight change did not differ between START treatment arms in people who began therapy with a viral load above 50,000 copies. And in a finding at odds with other research, being a woman was associated with lower weight gains in the immediate START arm.
START enrolled antiretroviral-naive adults with a CD4 count above 500 and randomized 4684 of them to begin ART immediately or to defer treatment until the CD4 count fell below 350 or AIDS developed [2]. START had participants in 35 countries in Africa, Asia, Australia, Europe, North America, and South America. In a previous analysis of START results, the researchers reported a slower weight gain through 60 months in people randomized to immediate versus deferred ART [3].
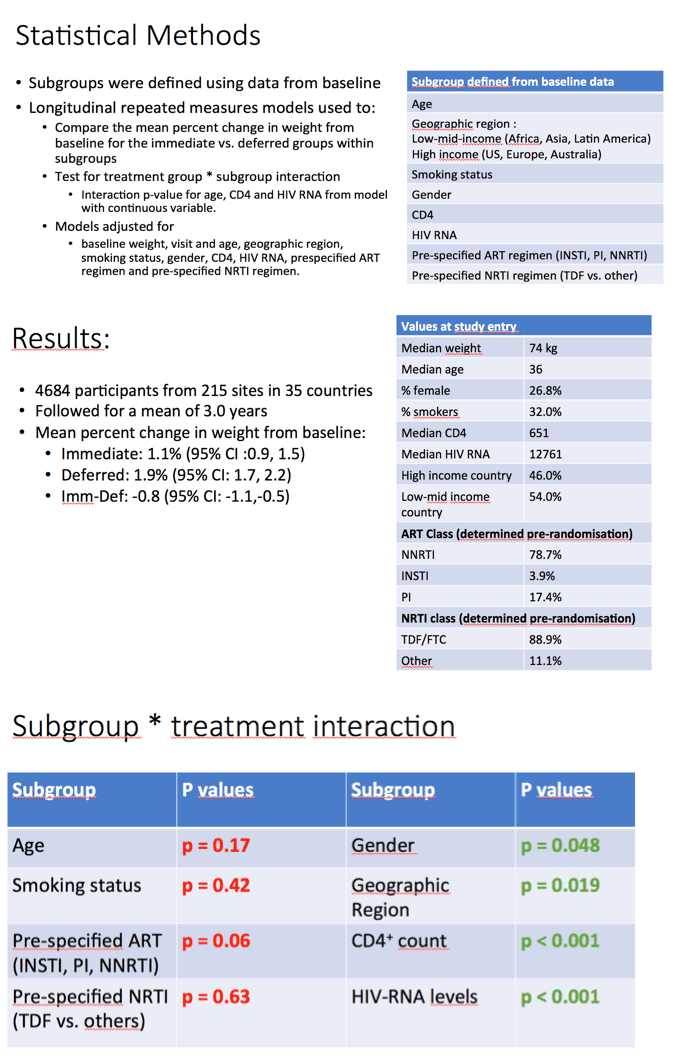
For the new analysis the START team used longitudinal repeated measures models to compare the average percent change in weight from study entry in subgroups of the immediate versus the deferred treatment arms. Models adjusted for baseline weight and for each of the subgroups: age, geographic region, smoking status, gender, CD4 count, viral load, prespecified ART regimen, and prespecified nucleoside regimen.
At the baseline visit, median weight averaged 74 kg, median age 36 years, median CD4 count 651, and median viral load 12,761 copies. About one quarter of participants (26.8%) were women, 54% lived in low-mid-income countries, and 46% lived in high-income countries. Nonnucleosides dominated as the predetermined anchor antiretroviral class (78.7%), followed distantly by protease inhibitors (17.4%) and integrase inhibitors (3.9%). A large majority of participants, 88.9%, started tenofovir disoproxil fumarate/emtricitabine.
Through a median 3 years of follow-up, mean percent change in weight from the baseline visit was 1.13% in the immediate-ART arm (95% confidence interval [CI] 0.86 to 1.48) versus 1.92% in the deferred-ART arm (95% CI 1.65 to 2.19). Average immediate-versus-deferred difference in percentage change in weight measured -0.79% (95% CI -1.08 to -0.50), a highly significant difference (P < 0.001).
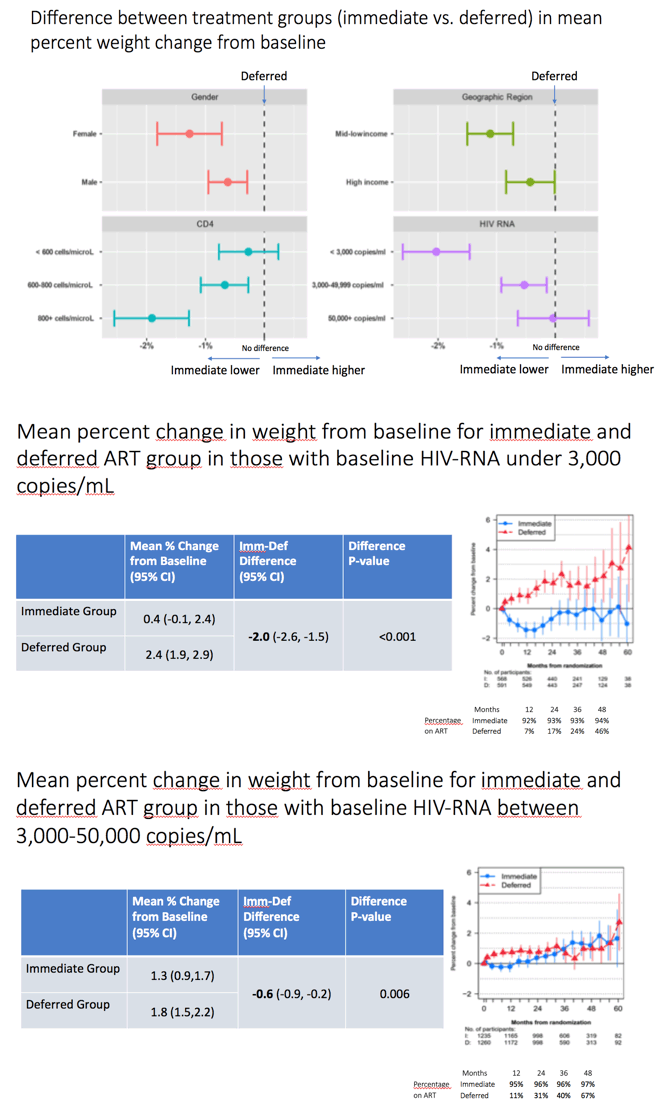
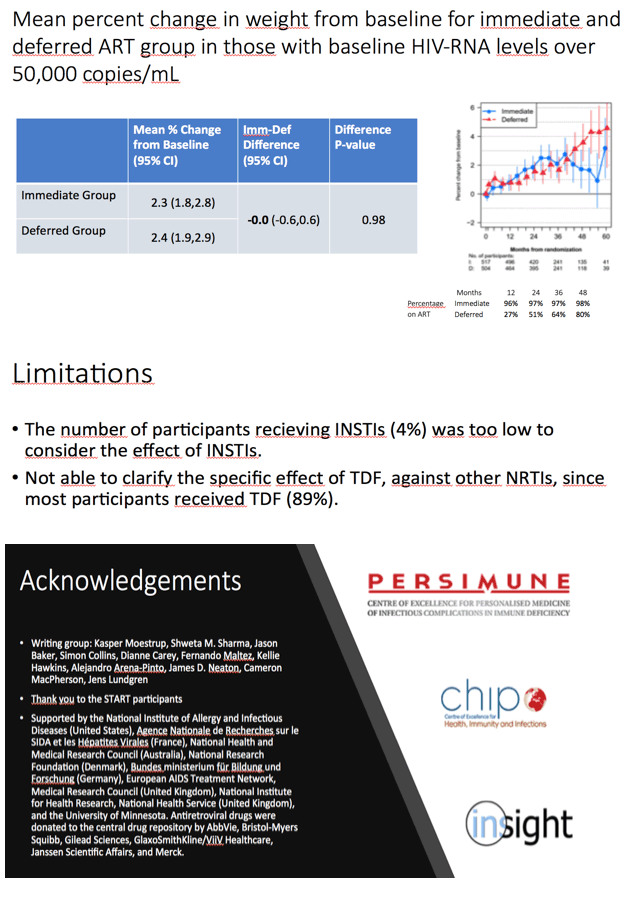
The weight difference between the immediate and deferred arms was greatest in people with a pretreatment viral load below 3000 copies. The weight-change difference between the immediate and deferred ART groups disappeared in people with a pretreatment viral load of 50,000 copies or more. The immediate-versus-difference difference in mean percent weight change from baseline was -2.0% (95% CI -2.6 to -1.5, P < 0.001) for people with a pretreatment load below 3000 copies, -0.55% (95% CI -0.9 to -0.2, P = 0.006) for people with a pretreatment load between 3000 and 50,000 copies, and -0.0% (95% CI -0.6 versus 0.6, P = 0.98) for those with a pretreatment load above 50,000 copies.
Three other factors were associated with less weight gain in the immediate versus deferred ART groups: higher CD4 count (subgroup x treatment interaction P < 0.001), female gender (subgroup x treatment interaction P = 0.048), and living in a low-mid-income country (subgroup x treatment interaction P = 0.019). People taking a nonnucleoside appeared to gain less weight than those taking a protease inhibitor or an integrase inhibitor, but these differences lacked statistical significance. Too few people started an integrase inhibitor in START to allow researchers to say anything about how this class affects weight.
START investigators speculated that "ART reduces the catabolic effects of HIV infection in those with high HIV RNA levels thereby causing a weight increase."
References
1. Moestrup KS, Sharma S, Baker J, et al. Antiretroviral therapy and body weight in the START (Strategic Timing of Antiretroviral Treatment) trial. 17th European AIDS Conference, November 6-9, 2019, Basel. Abstract 3/1.
2. INSIGHT START Study Group, Lundgren JD, Babiker AG, Gordin F, et al. Initiation of antiretroviral therapy in early asymptomatic HIV infection. N Engl J Med. 2015;373:795-807.
3. Baker JV, Sharma S, Achhra AC, et al. Changes in cardiovascular disease risk factors with immediate versus deferred antiretroviral therapy initiation among HIV-positive participants in the START (Strategic Timing of Antiretroviral Treatment) trial. J Am Heart Assoc. 2017;6:pii e004987.

|
| |
|
 |
 |
|
|