| |
HIV/Safe PD-1 Cancer Treatment / Studies Should Include HIV+
|
| |
| |
Download the PDF here
Download the PDF here
Download the PDF here
Original Investigation
June 2, 2019
https://www.fredhutch.org/en/news/center-news/2019/06/hiv-cancer-clinical-trials.html
Hutch News - Breaking down HIV exclusions in cancer clinical trials - As an eligibility barrier cracks, a lung cancer patient gets a new lease on life - June 7, 2019 - By Fred Hutch News Service staff. For reasons that date back to the earliest days of the AIDS epidemic, HIV-positive people with cancer have often been excluded from cancer clinical trials. Now, that may be changing. During an ASCO presentation concurrent with the release of the study in JAMA Oncology, Hutch researcher and lead author Dr. Tom Uldrick said that in nearly all cases it was safe to use the drug in patients with cancer and HIV.
The "adverse events profile," a measure of the safety of the drug in the study, was not substantially different from prior studies that excluded such patients. The results, study authors said, are likely applicable to five similar drugs that block receptors known as PD-1 or PD-L1 on the surface of T cells. "Our conclusion is that anti-PD-1 therapy is appropriate for cancer patients with well-controlled HIV, and that patients with HIV and cancer can be treated with the drug and should be included in future immunotherapy studies," Uldrick said in an interview prior to the conference. "Exclusion of people with HIV in clinical trials is a longstanding problem that grew out of the poor outcomes of AIDS patients with cancer, before there were effective antiviral therapies for HIV," Uldrick said. In prior research, Uldrick surveyed 46 recent clinical trials that led to the approval of cancer drugs, and he found 30 contained explicit exclusions for patients with HIV, and nine others where an exclusion was implied. The study findings strengthen the case for opening the door to cancer clinical trials for people with HIV, a door that has begun to crack open.
The National Cancer Institute has encouraged inclusion of HIV-infected patients on clinical trials since 2008, and the Food and Drug Administration in March circulated for comment a draft of non-binding guidelines calling for appropriate inclusion of people with HIV, hepatitis B or hepatitis C in cancer clinical trials. This is the first of two NCI-sponsored trials specifically testing whether a class of cancer immunotherapy drugs called checkpoint inhibitors is safe in patients with HIV. The 30-patient trial studied KEYTRUDA, an anti-PD-1 therapy. Keytruda is manufactured by Merck, which provided the study drug to the trial sponsor, the NCI. HIV-positive patients with different cancers that might respond to the drug were included in the trial. In addition to lung cancer, other cancers treated were Kaposi sarcoma, or KS; non-Hodgkin lymphoma; liver cancer; anal cancer and advanced squamous cell skin cancer. One participant in the trial, enrolled for the treatment of KS, died of a noncancerous condition involving KSHV, the virus that causes KS, and overproduction of certain white blood cells. Although a link between the patient's death and use of the drug is unclear, the study authors recommend that patients with HIV who have high levels of KSHV and symptoms of a condition known as multicentric Castleman disease - which is also linked to the virus and affects white blood cells - be tested before considering the cancer drug. Safety issues addressed in the trial include whether the known side effects of the drug, such as gastrointestinal distress and skin rash, were more problematic in HIV patients. The researchers were also watching for unanticipated disturbances in their immune system, such as an unhealthy or uncontrolled surge in T cells. "Many cancer researchers just assumed that checkpoint inhibitor therapy, which relies on patients' T cells to fight cancer, would not be effective in HIV-infected individuals," noted Dr. Robert Yarchoan, chief of NCI's HIV and AIDS Malignancy Branch. The trial allayed those worries.
---------------------
Assessment of the Safety of Pembrolizumab in Patients With HIV and Advanced Cancer-A Phase 1 Study
As this population ages, the burden of certain cancers such as lung cancer, anal cancer, and cHL continues to increase.[Shiels 2011]Cancer is estimated to be responsible for over one-third of all deaths in this patient population.[Bonnet 2009].....
However, lack of knowledge about the use of cancer therapies in patients with HIV, health care disparities,7 and biologic factors, including advanced HIV-associated immunosuppression,8-10 may affect outcomes.
It is unknown whether anti-PD-1 therapy has an acceptable safety profile in people with HIV.
The current study has demonstrated that pembrolizumab has a similar AE profile for people with HIV and advanced cancer who have suppressed HIV on ART to that observed in published studies of participants without HIV.15,16 The proportion of grade 3 and 4 irECI was generally similar to that previously described in patients receiving anti-PD-1 therapy for FDA-approved indications. The commonest irECI was hypothyroidism, which was noted in 6 (20%) participants and was successfully managed through monitoring of TSH and administration of levothyroxine using routine guidelines.17
Through prospective evaluation in participants with HIV, the current study demonstrates that pembrolizumab monotherapy does not appear to have a detrimental effect on CD4+ T-cell counts......HIV viral loads remained suppressed below the limit of detection on commercial assays in 23 (77%) participants, and low-level viremia, generally blips less than 400 copies/mL (of no clinical significance), was noted in only 7 patients. No participant required a change of ART. Correlative studies evaluating the effects of pembrolizumab on HIV latency reversal and measures of persistence are ongoing.
Conclusions: Data from the present study strongly support the use of monoclonal antibodies targeting the PD-1 pathway in people with HIV on ART and CD4+ T-cell counts of more than 100 cells/μL for FDA-approved cancer indications. These data also demonstrate the feasibility of including patients with HIV in immunotherapy trials with appropriate eligibility criteria and study design.11 Tumor regression in participants with a range of tumor types and CD4 counts supports activity of anti-PD-1 therapy in people with HIV. Evaluation of pembrolizumab as a first-line systemic therapy for HIV-associated KS is ongoing.
JAMA Oncol. Published online June 2, 2019
Thomas S. Uldrick, MD, MS, et al.
for the Cancer Immunotherapy Trials Network (CITN)-12 Study Team
Key Points
Question Is anti-PD-1 (anti-programmed cell death 1) therapy safe to administer in people with HIV with a range of CD4+ T-cell counts and cancer?
Findings In this multicenter, open-label, nonrandomized, phase 1 study of 30 participants with HIV, a CD4 count of greater than 100 cells/μL, and advanced cancer, pembrolizumab had an acceptable safety profile, although an unexpected treatment-emergent adverse event of Kaposi sarcoma herpesvirus-associated polyclonal B-cell lymphoproliferation was noted. Clinical benefit was observed in participants with Kaposi sarcoma, primary effusion lymphoma, diffuse large B-cell lymphoma, and lung cancer.
Meaning Anti-PD-1 therapy is appropriate for US Food and Drug Administration-approved indications and clinical trials in people with HIV.
Abstract
Importance Anti-PD-1 (anti-programmed cell death 1) and anti-PD-L1 (anti-programmed cell death ligand 1) regimens are preferred therapies for many cancers, including cancers associated with HIV. However, patients with HIV were excluded from most registered trials.
Objective The primary objective was to evaluate the safety of pembrolizumab in people with HIV and advanced cancer; the secondary objective was to evaluate tumor responses.
Design, Setting, and Participants Open-label, nonrandomized, phase 1 multicenter study conducted at 7 Cancer Immunotherapy Trials Network sites. Patients with HIV and advanced cancer as well as a CD4 count greater than or equal to 100 cells/μL, antiretroviral therapy (ART) for 4 or more weeks, and an HIV viral load of less than 200 copies/mL were eligible. Exclusion criteria included uncontrolled hepatitis B or C infection, active immunosuppressive therapy, or a history of autoimmune disease requiring systemic therapy.
Interventions Pembrolizumab, 200 mg, administered intravenously every 3 weeks for up to 35 doses in 3 CD4 count-defined cohorts. Participants continued ART.
Main Outcomes and Measures Safety and tolerability were assessed using current NCI Common Terminology Criteria for Adverse Events. Immune-related adverse events grade 2 or higher were considered immune-related events of clinical interest (irECI). Tumor responses were evaluated using standard tumor-specific criteria.
Results
Thirty participants (28 men and 2 women; median [range] age, 57 [39-77] years) were enrolled from April 2016 through March 2018; 6 had Kaposi sarcoma (KS), 5 had non-Hodgkin lymphoma (NHL), and 19 had non-AIDS-defining cancers.
Safety was observed over 183 cycles of treatment with pembrolizumab. Most treatment-emergent adverse events at least possibly attributed to pembrolizumab were grade 1 or 2 (n = 22), and 20% (n = 6) were grade 3.
The irECI included hypothyroidism (6 participants), pneumonitis (3 participants), rash (2 participants), an elevated aminotransferase/alanine aminotransferase level (1 participant), and a musculoskeletal event (1 participant).
One participant with pretreatment KS herpesvirus (KSHV) viremia developed a polyclonal KSHV-associated B-cell lymphoproliferation and died. HIV was controlled in all participants.
Increases in CD4 count were not statistically significant (median increase, 19 cells/μL; P = .18).
Best tumor responses included complete response (lung, 1 patient), partial response (NHL, 2 patients), stable disease for 24 weeks or more (KS, 2 patients), stable disease for less than 24 weeks (15 patients), and progressive disease (8 patients); 2 patients were not evaluable.
Conclusions and Relevance
Pembrolizumab has acceptable safety in patients with cancer, HIV treated with ART, and a CD4+ T-cell count of greater than 100 cells/μL but may be associated with KSHV-associated B-cell lymphoproliferation. Clinical benefit was noted in lung cancer, NHL, and KS. Anti-PD-1 therapy is appropriate for US Food and Drug Administration-approved indications and clinical trials in this population.
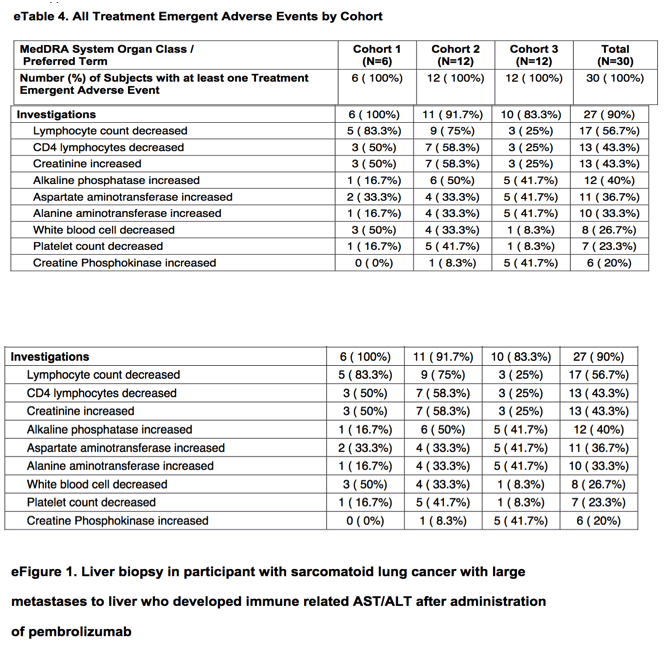
Discussion
Checkpoint inhibitors provide an important treatment option for people with HIV and cancer. Anti-PD-1/PD-L1 therapy has been approved for a variety of cancers that occur with increased incidence in people with HIV, including lung cancer,27,28 squamous cell skin cancer,29 cervical cancer,30,31 HCC,32 cHL,33 head and neck squamous cell carcinoma34 and Merkel cell carcinoma.35 However, the safety of this therapy in people with HIV has not been previously explored prospectively. The current study has demonstrated that pembrolizumab has a similar AE profile for people with HIV and advanced cancer who have suppressed HIV on ART to that observed in published studies of participants without HIV.15,16 The proportion of grade 3 and 4 irECI was generally similar to that previously described in patients receiving anti-PD-1 therapy for FDA-approved indications. The commonest irECI was hypothyroidism, which was noted in 6 (20%) participants and was successfully managed through monitoring of TSH and administration of levothyroxine using routine guidelines.17
Through prospective evaluation in participants with HIV, the current study demonstrates that pembrolizumab monotherapy does not appear to have a detrimental effect on CD4+ T-cell counts. In the setting of relapsed and refractory cancers, CD4+ T-cell counts tended to increase during the study, although the increases were not statistically significant. Additionally, HIV viral loads remained suppressed below the limit of detection on commercial assays in 23 (77%) participants, and low-level viremia, generally blips less than 400 copies/mL (of no clinical significance), was noted in only 7 patients. No participant required a change of ART. Correlative studies evaluating the effects of pembrolizumab on HIV latency reversal and measures of persistence are ongoing.
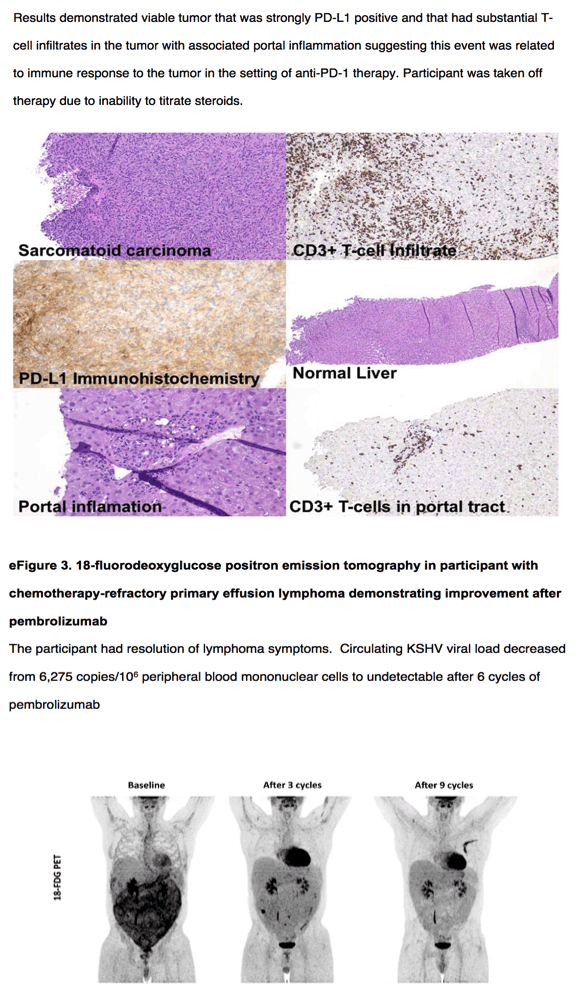
One hesitation in testing anti-PD-1 or anti-PD-L1 therapy in patients with HIV and cancer has been a concern that these patients would not have sufficient underlying T-cell immunity to benefit from therapy. However, although the focus of this trial was safety, the responses in several tumor types across all 3 cohorts were also documented, including 2 AIDS-defining cancers, KS and NHL. Kaposi sarcoma is a highly immune-responsive tumor that is sometimes managed with ART alone,9,36 although additional therapy is often needed. Retrospective reports have described responses to anti-PD-1 therapy in HIV-associated and endemic KS.37,38 Prospective evaluation is warranted to evaluate the efficacy of anti-PD-1 monoclonal antibodies for KS. To date, tumor regression was noted in 5 of 6 participants with relapsed or refractory KS, although this did not meet criteria for partial response at the time of analysis. To better define anti-PD-1 activity in KS, the CITN-12 study team continues participant enrollment in a phase 1b cohort to evaluate pembrolizumab as a first-line systemic therapy in addition to ART for HIV-associated KS. Additionally, data from the present study demonstrated meaningful activity against primary effusion lymphoma, a KSHV-associated cancer with few good treatment options (eFigure 3 in Supplement 2). Although anti-PD-1 therapy may be promising in KSHV-associated cancers, a previously undescribed KSHV-associated B-cell lymphoproliferation was observed in a patient with KS and a history of circulating cell–associated KSHV that was at least possibly attributable to pembrolizumab.
Initial CITN-12 KS eligibility criteria include at least 3 months on ART, the timeframe in which patients with KS are at the greatest risk of immune reconstitution inflammatory syndrome.39 Because death from generalized polyclonal KSHV–associated B-cell lymphoproliferation potentially represents KSHV-multicentric Castleman disease (MCD), the protocol was amended to exclude patients with a history of KSHV-MCD in the last 5 years. In patients with KS and unexplained symptoms concerning for KSHV-MCD, an assessment of KSHV viral load and a computed tomography scan are warranted, and enlarged lymph nodes should be biopsied. In general, KSHV-MCD is successfully managed with rituximab,40,41 which should be considered if KSHV-MCD is observed in the setting of anti-PD-1 therapy.
Limitations
To our knowledge, this is the first and largest prospective study evaluating the safety of anti-PD-1 therapy in people with HIV and cancer; however, the phase 1 design and sample size did not allow for a formal comparison of rates of specific AEs, such as hypothyroidism, with those noted in the general population. Because this was not a randomized study, we were unable to compare TEAE rates associated with the use of pembrolizumab vs alternative cancer interventions or observation alone in this population. Lastly, although the present study demonstrated that pembrolizumab had activity in several cancers, the study did not have enough participants with any given tumor to accurately estimate response rates or to compare response rates with those of people with the same cancers but no HIV.
Conclusions
Data from the present study strongly support the use of monoclonal antibodies targeting the PD-1 pathway in people with HIV on ART and CD4+ T-cell counts of more than 100 cells/μL for FDA-approved cancer indications. These data also demonstrate the feasibility of including patients with HIV in immunotherapy trials with appropriate eligibility criteria and study design.11 Tumor regression in participants with a range of tumor types and CD4 counts supports activity of anti-PD-1 therapy in people with HIV. Evaluation of pembrolizumab as a first-line systemic therapy for HIV-associated KS is ongoing.
|
|
| |
| |
|
|
|