 |
 |
 |
| |
CLINICAL SIGNIFICANCE OF gp120 POLYMORPHISMS, TMR IC50FC AND HIV-1 SUBTYPE IN BRIGHTE
|
| |
| |
CROI 2020
Reported by Jules Levin
Download the PDF here
IAS 2019 - Week 96 safety and efficacy of the novel HIV-1 attachment inhibitor prodrug fostemsavir in heavily treatment-experienced participants infected with multi-drug-resistant HIV-1 (BRIGHTE Study)....http://www.natap.org/2019/IAS/IAS_17.htm .........

Margaret Gartland1, Peter Ackerman2, Frank Mannino3, Louise K. Garside4, Andrew Clark5, Amy Pierce1, Mark Krystal2, Cyril C. Llamoso2, Max Lataillade2
1ViiV Healthcare, Research Triangle Park, NC, USA,2ViiV Healthcare, Branford, CT, USA,3GlaxoSmithKline, Collegeville, PA, USA,4GlaxoSmithKline, Uxbridge, UK,5ViiV Healthcare, London, UK
Abstract Body:
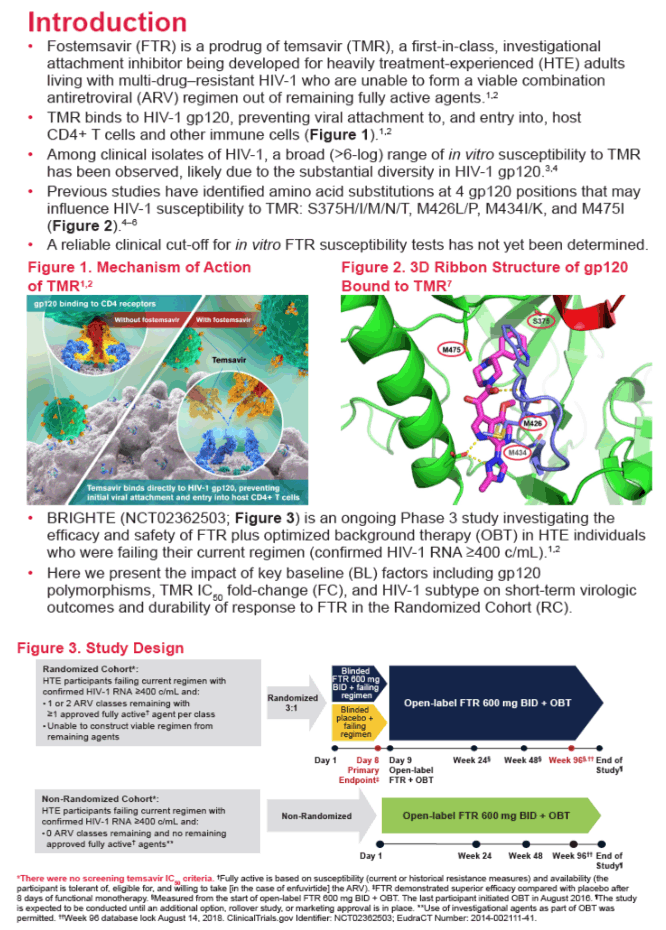
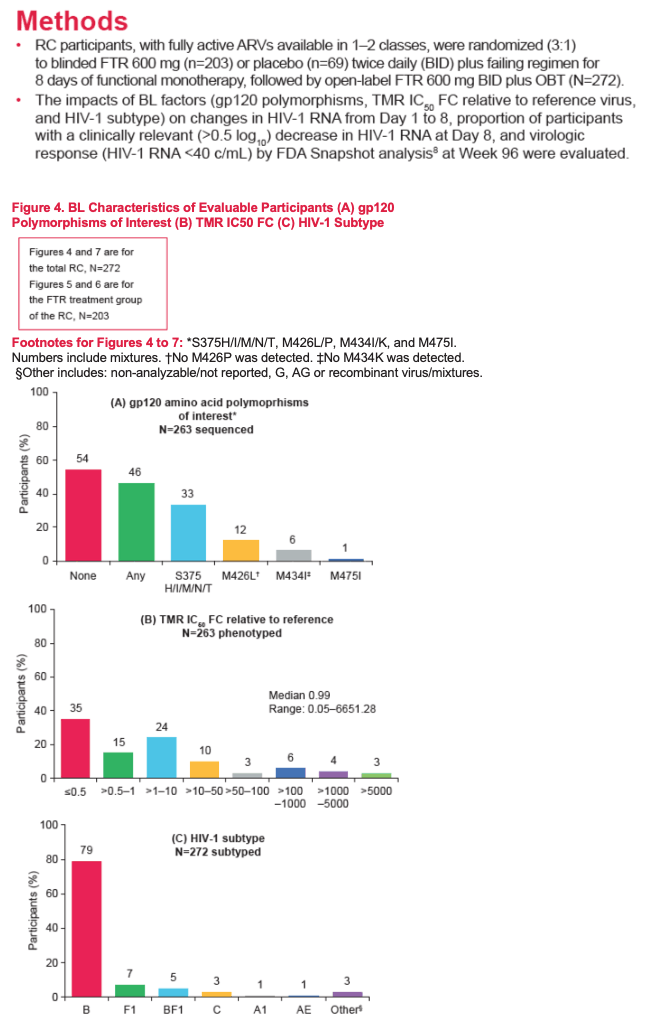
The ongoing Ph3 BRIGHTE study is evaluating Fostemsavir (FTR), an investigational prodrug of the first-in-class attachment inhibitor temsavir (TMR), in heavily treatment-experienced (HTE) participants with multi-drug resistant HIV-1 infection who are unable to form a viable regimen from fully active ARV agents. We present the impact of key baseline (BL) factors on short-term virologic outcome and durability of response to FTR in the Randomized Cohort (RC).
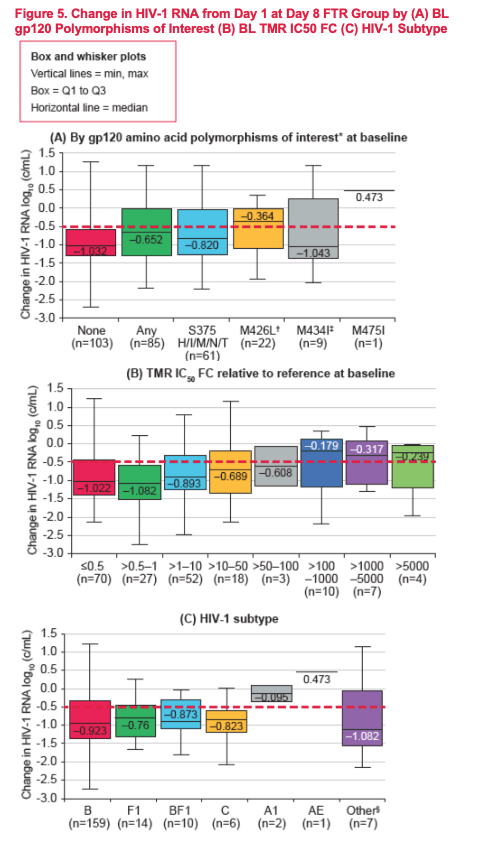
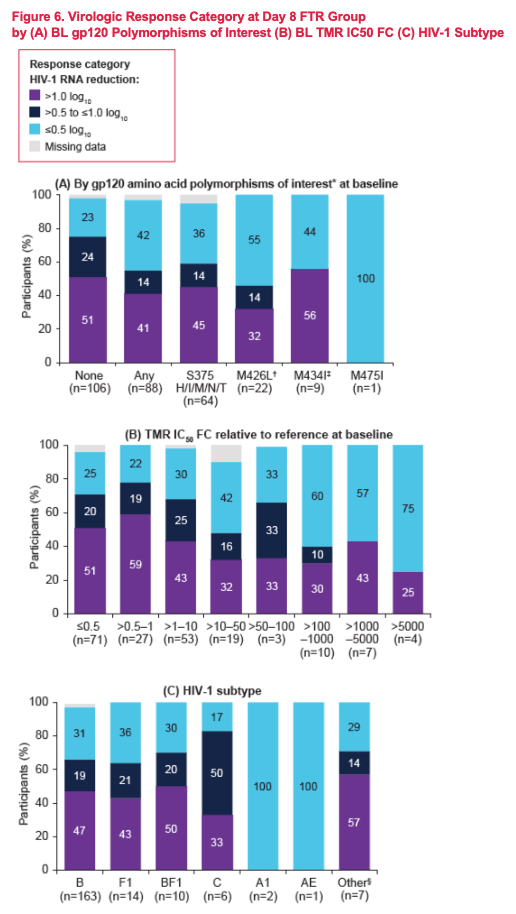
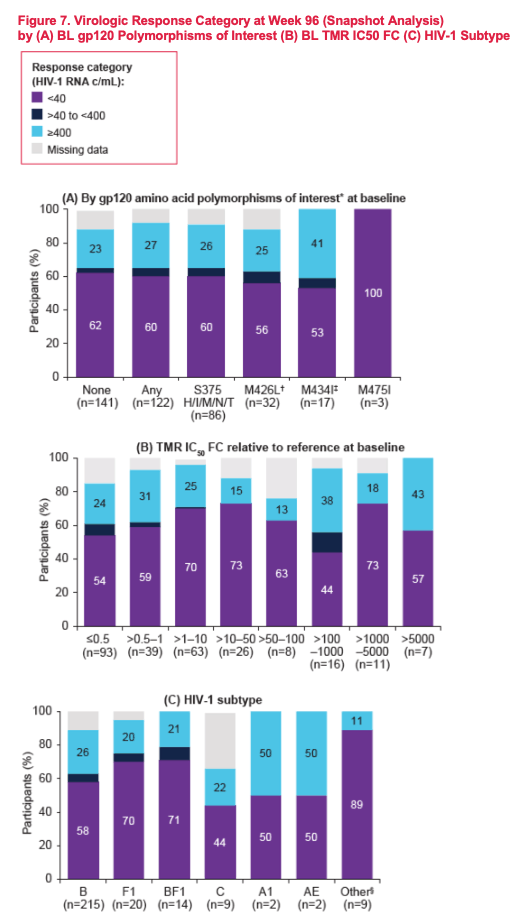
RC participants, with 1-2 fully active ARVs were randomized (3:1) to blinded FTR 600 mg (n=203) or placebo (n=69) BID plus failing regimen for 8 days of functional monotherapy, followed by open-label FTR 600mg BID plus optimized background therapy (OBT; n=272). The impact of BL factors: gp120 polymorphisms, TMR IC50 fold-change (FC), and HIV-1 subtype, on change in HIV-1 RNA from Day 1 to 8, proportion of participants with a clinically relevant (>0.5log10) decrease in HIV-1 RNA at Day 8, and HIV-1 RNA <40 c/mL at W96, was evaluated.
Overall, 46% (122/263) of evaluable RC participants had a relevant gp120 polymorphism present at BL. Median change in HIV-1 RNA at Day 8 was lower among monotherapy participants with vs without BL gp120 polymorphisms of interest (-0.65 log10 vs -1.03 log10). However, 55% (48/88) of participants with BL gp120 polymorphisms achieved a viral load reduction >0.5 log10 at Day 8. BL TMR IC50FC from reference was observed over a broad range (0.05 to >5,000-fold; median 0.99-fold) with 74% (195/263) and 87% (229/263) of evaluable participants with TMR IC50FC <10- and <100-fold, respectively. While monotherapy participants with TMR IC50FC >100-fold at BL had a median change in HIV-1 RNA of <0.5 log10 at Day 8, this did not prevent a decline >0.5 log10. In fact, 38% (8/21) of participants with BL TMR IC50FC >100-fold achieved >0.5 log10 decline over this time. The majority of participants in the RC (79%, 216/272) had HIV-1 subtype B virus. Similar proportions of monotherapy participants with subtype B (66%, 108/163) vs non-B (65%, 26/40) virus achieved >0.5 log10 decline in HIV-1 RNA at Day 8; although the number of participants with non-B subtype, including AE, was small (n= 40 and 1, respectively).
In BRIGHTE, BL gp120 polymorphisms of interest, TMR IC50FC, and HIV-1 subtype did not reliably predict virologic outcome at Day 8 of FTR functional monotherapy and did not impact durability of response (HIV-1 RNA <40 c/mL) to FTR + OBT through 96 weeks of therapy, among HTE participants in the RC.


|
| |
|
 |
 |
|
|