 |
 |
 |
| |
MK-8504 and MK-8583 (Tenofovir Prodrugs)
Single-Dose PK and Antiviral Activity in HIV Infection
|
| |
| |
CROI 2020
Reported by Jules Levin
Randolph P. Matthews1; Sarah J. Hsieh1; Jesse C. Nussbaum1; Jonathan A. Robbins1; Guido H. Jajamovich1; Tian Zhao1; Saijuan Zhang1;
Diana Selverian1; Michaelanne Wasenius1; Liesbeth Haspeslagh2; Jean-François Denef2; Caroline Cilissen2; Kristien Van Dyck2;
Dirk Schürmann3; Sylvie Rottey4; Marta Boffito5; Tom Reynders2; Deanne Jackson Rudd1; S. Aubrey Stoch1; Marian Iwamoto1
1MRL, Merck & Co., Inc., Kenilworth, NJ, USA; 2MSD, Belgium, Brussels, Belgium; 3Charité Research Organisation GmbH, Berlin, Germany; 4Ghent University, Ghent, Belgium; 5Chelsea and Westminster Hospital, London, UK
Abstract
There is a need for Human Immunodeficiency Virus (HIV-1) treatments with improved safety profiles and increased ease of administration. MK-8504 and MK-8583 are nucleoside reverse transcriptase inhibitors (NRTIs) that are novel tenofovir (TFV) prodrugs, with a potential for weekly dosing due to the long t1/2 of the active diphosphate (TFV-DP). Single-dose pharmacokinetics (PK) and antivirologic activity of these compounds were assessed in two Phase 1 programs.
Single doses of 2-240 mg MK-8504 were tested in healthy adults, and single doses up to 240 mg were tested in ART-naïve persons living with HIV (PLWH). Single doses of MK-8583 2-150 mg were tested in healthy adults, and a single dose of 100 mg was tested in ART-naïve PLWH. Plasma and peripheral blood mononuclear cells (PBMCs) were collected up to 10 days after dosing for PK and viral load (VL).
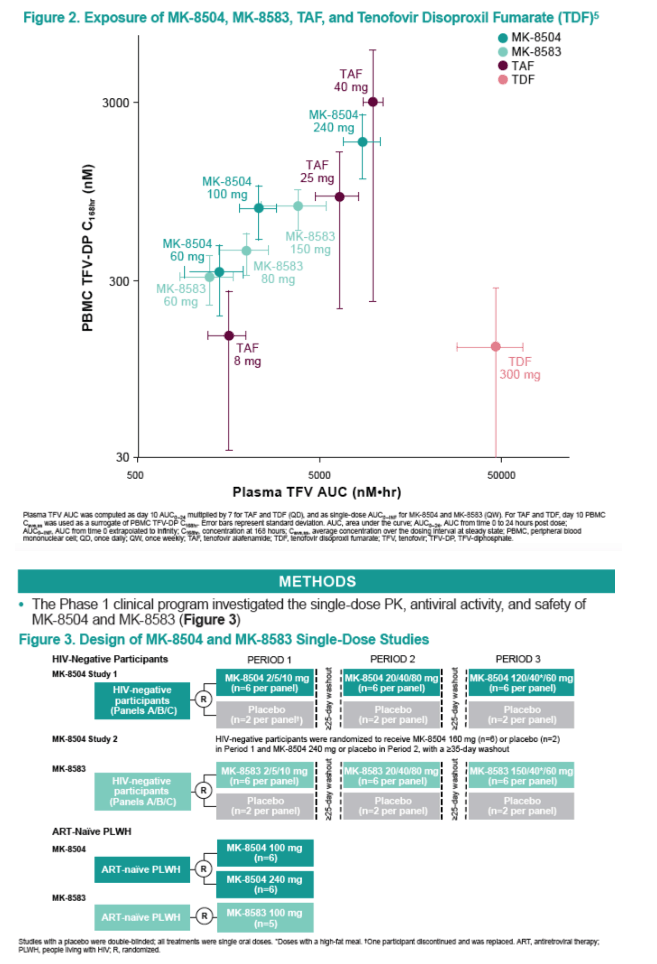
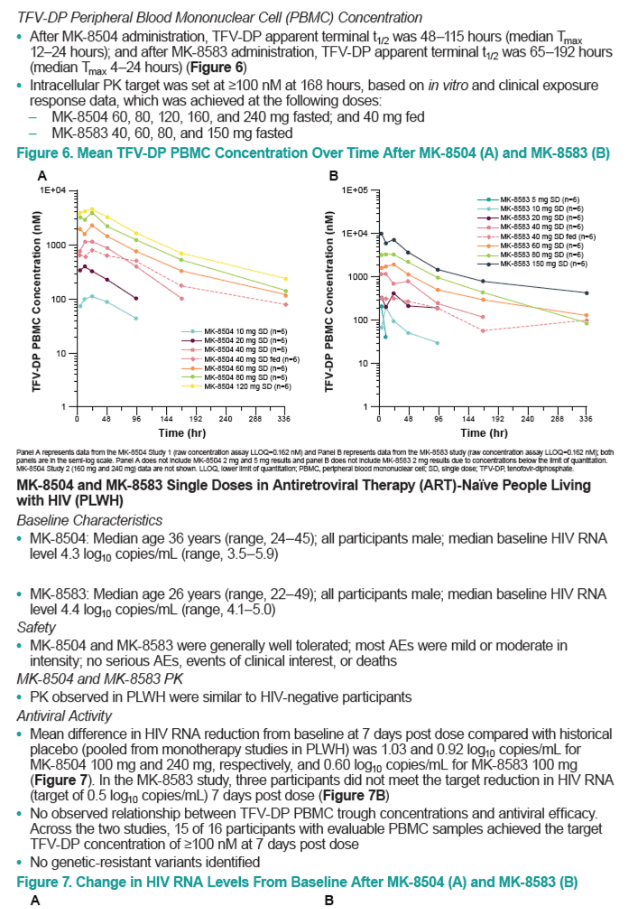
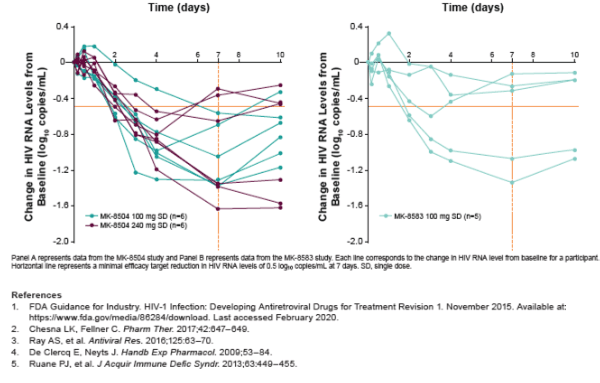
Oral MK-8504 and MK-8583 were rapidly absorbed (Tmax ~ 0.5 hour); MK-8583 was rapidly eliminated from plasma (t1/2 0.2-0.4 hours), while MK-8504 had slower elimination (t1/2 6-8 hours). As expected, plasma TFV concentrations were generally similar, with a median Tmax of 1-4 hours after both MK-8504 and MK-8583 administration, and a t1/2 of 20-38 hours for MK-8504 and 19-30 hours for MK-8583. The levels of TFV-DP in PBMCs exhibited a median Tmax of 4-24 hr for both compounds, with a t1/2 of 48-115 hours for MK-8504 and 65-192 hours for MK-8504. The PK in PLWH and healthy participants were similar. Despite PBMC TFV-DP concentrations consistently above the efficacious trough concentration for marketed TFV prodrugs (100 nM), HIV-1 VL reduction was suboptimal for both compounds. The mean VL reduction at 7 days was 0.6 log10 copies/mL for MK-8583 and 0.9 log10 copies/mL at the top dose of MK-8504, and several participants failed to achieve consistent VL reduction. There was no relationship observed between Day 7 PBMC TFV-DP concentrations and VL reduction for either compound. No genetic resistant variants were identified.
Single doses of MK-8504 and MK-8583 were generally well-tolerated. These TFV prodrugs were rapidly converted to the active form and maintained target concentrations in PBMCs through 7 days. Unlike other TFV prodrugs administered daily in monotherapy trials, the antiretroviral activity of MK-8504 and MK-8583 was modest and transient. The persistent adequate concentrations of TFV-DP belie the poor VL response; it is possible, though, that VL responses could improve with daily administration of MK-8504 or MK-8583. Collectively, these data raise questions about the feasibility of TFV prodrugs in extended-interval dosing regimens.
|
| |
|
 |
 |
|
|