 |
 |
 |
| |
REAL-LIFE MANAGEMENT OF DRUG-DRUG INTERACTIONS BETWEEN ANTIRETROVIRALS AND STATINS
|
| |
| |
CROI 2020
Perrine Courlet1, Françoise Livio1, Susana Alves Saldanha1, Alexandra Scherrer2,3, Manuel Battegay4,5, Matthias Cavassini6, Marcel Stoeckle4,5, Laurent Arthur Decosterd1,Catia Marzolini4,5,7,§and the Swiss HIV Cohort Study
1Service of Clinical Pharmacology, University Hospital of Lausanne and University of Lausanne, Lausanne, Switzerland; 2Division of Infectious Diseases and Hospital Epidemiology, University Hospital of Zurich, University of Zurich, Zurich, Switzerland; 3Institute of Medical Virology, University of Zurich, Zurich, Switzerland 4Division of Infectious Diseases and Hospital Epidemiology, Departments of Medicine and Clinical Research, University Hospital of Basel, Basel, Switzerland; 5University of Basel, Basel, Switzerland; 6Service of Infectious Diseases, University Hospital of Lausanne and University of Lausanne, Lausanne, Switzerland; 7Department of Molecular and Clinical Pharmacology, Institute of Translational Medicine, University of Liverpool. Liverpool, UK

abstract
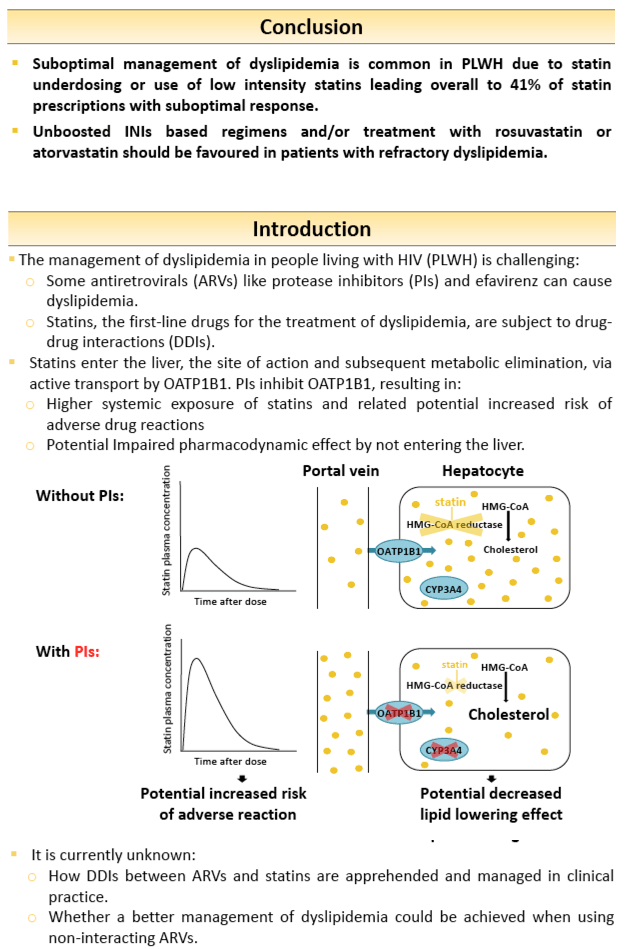
Protease inhibitors (PIs) cause drug-drug interactions (DDIs) with statins due to inhibition of drug metabolizing enzymes and/or the hepatic uptake transporter OATP1B1, which may alter the pharmacodynamic (PD) response to statins. There is a lack of data on real-life management of DDIs between antiretrovirals (ARVs) and statins.
Patients of the Swiss HIV Cohort Study followed-up in the centres of Lausanne and Basel were eligible if they received a statin concomitantly to ARVs. Low-density lipoprotein (LDL), total cholesterol (TC) and plasma concentration of the statin were measured during a follow-up visit. Individual LDL target values were set using the Framingham score whereas TC target values were set according to the 2018 European AIDS Clinical Society recommendations. Statins concentrations were interpreted using published plasma concentration time curves. DDIs management was evaluated based on the statin dose adjustment considering coadministered ARVs and the PD response on the lipid profile.
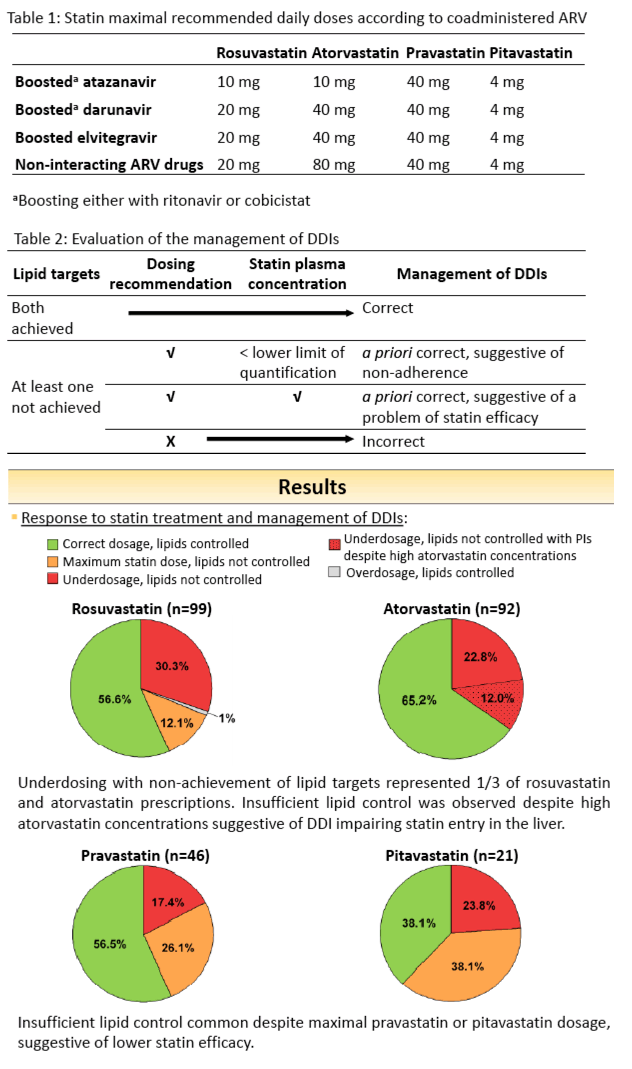
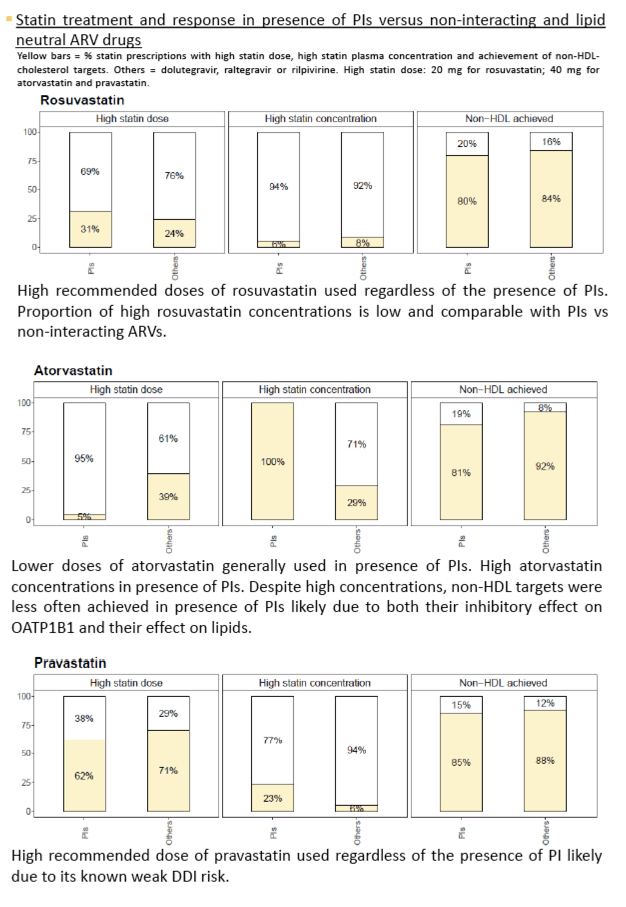
Data were collected for 99 rosuvastatin, 93 atorvastatin, 46 pravastatin and 21 pitavastatin. DDIs management and PD response varied according to the statin (figure 1). Statin underdosing leading to suboptimal PD response was frequent with rosuvastatin and atorvastatin. However, the lipid target values were not always achieved in presence of PIs despite using the maximal recommended rosuvastatin dose. Similarly, suboptimal lipid control was observed with PIs despite high atorvastatin concentrations likely explained by inhibition of OATP1B1 resulting in less statin uptake in the liver, the site of action. Target lipid values were more often achieved with unboosted integrase inhibitors due to both their favourable DDIs profiles and neutral effect on lipids. Underdosing was less frequent with pravastatin and pitavastatin, nevertheless suboptimal lipid control was common regardless of coadministered ARVs and despite using maximal recommended pravastatin and pitavastatin doses. This is likely due to their lower efficacy compared to rosuvastatin or atorvastatin.
Suboptimal management of DDIs with statins underdosing was observed in overall 30% of cases. Management of dyslipidemia in patients on PIs is challenging due to this ARVs class negative impact on lipid profile and DDIs potentially impairing the effect of statins. Integrase inhibitors based regimens and/or treatment with rosuvastatin or atorvastatin should be favoured in patients with refractory dyslipidemia.
|
| |
|
 |
 |
|
|