| |
The switch from tenofovir disoproxil fumarate to tenofovir alafenamide determines weight gain in patients on rilpivirine-based regimen
|
| |
| |
Download the PDF here
Abstract
Objective:
To investigate whether the switch from tenofovir disoproxil fumarate/emtricitabine/rilpivirine (TDF/FTC/RPV) to tenofovir alafenamide (TAF)/FTC/RPV is associated with weight gain in people living with HIV (PLWHIV).
Design:
Retrospective single-centre study.
Methods:
All PLWHIV on TDF/FTC/RPV who switched to TAF/FTC/RPV from January 2017 to December 2018 were considered if they had at least two weight measures in the year before and two after the switch. The weight trend across the study was evaluated by a generalized linear model for repeated measures, with pair comparison performed by Bonferroni adjustment.
Results:
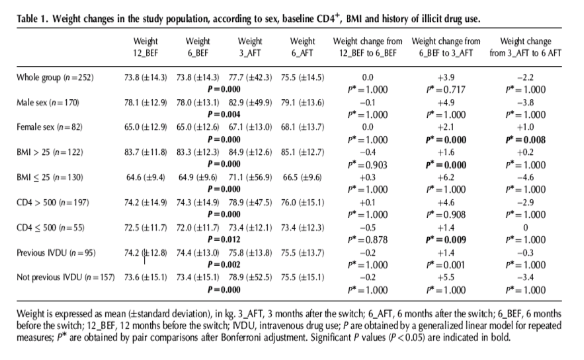
Two hundred and fifty-two patients on TDF/FTC/RPV were included, 65% men, mean age 51.2 years (±9.6), history of 18 (±18.2) years of HIV infection and CD4+T-cell count of 744 (±329) cells/μl. All had HIV-RNA <50 copies/ml. Twelve months before the switch, baseline weight was 73.8 (±14.3) kg, and remained stable to 73.8 (±14.3) kg in the following 6 months. A weight increase was noticed 3 and 6 months after the switch, to 77.7 (±42.3) and 75.5 (±14.5) kg, respectively (P < 0.0001). A significant weight change exactly within the timeframe of the switch (between 6 months before and 3 months after) was found in women, patients with higher BMI (>25 kg/m2), lower CD4+ T-cell count (≤500 cells/μl) and history of previous drug abuse. The frequency of BMI greater than 25 kg/m2 rose from 122/252 patients (48.4%), to 133/252 (52.8%) (P < 0.0001).
Conclusion:
TAF appears to have an impact on weight gain, similarly to what observed in naïve patients, also in experienced PLWHIV with good virologic control.
Introduction
Weight gain has historically been associated with better survival and improved long-term outcomes for people living with HIV (PLWHIV) after the introduction of antiretroviral therapy (ART) [1]. However, weight gain has recently emerged as a primary side effect of ART, potentially increasing the associated cardiovascular risks and negatively impacting the quality of life of PLWHIV [2]. In particular, integrase strand transfer inhibitors (INSTIs), currently considered the first line of therapy for their excellent safety and efficacy profiles, have been linked to increased weight gain compared with other classes of antiretrovirals among naïve patients [3,4], as well as experienced ones [5,6]. Little data is available on the role of backbone drugs in the ART regimen. Tenofovir alafenamide (TAF), a drug characterized by its high efficacy and safety, has recently been associated with this same side effect among naïve patients and was linked to greater weight gain than tenofovir disoproxil fumarate (TDF) [7,8] and abacavir [8]. Moreover, the limb and trunk fat mass increases were pronounced among the naïve patients taking TAF as compared with TDF in the preliminary 96-week results of the ADVANCE trial [9]. Such a weight increase was also noticed in a retrospective study performed with 129 ART-experienced patients who switched from TDF to TAF; however, because of varying drug combinations with TAF and the study's small sample size, the data was not definitive [10]. Further, nonnucleoside reverse transcriptase inhibitors (NNRTIs) are considered to have low or insignificant levels of impact on weight gain [4,11,12], but NNRTI-treated patients taking rilpivirine (RPV) and doravirine have reported to gain more weight compared with those taking efavirenz [8,13] although doravirine and efavirenz had comparable effects on BMI [13]. In this study, we retrospectively analysed the weight changes of all the patients with HIV at the Policlinico San Martino Hospital, Italy, when they switched from TDF/emtricitabine (FTC)/RPV to TAF/FTC/RPV. This study aimed to define the role of TAF, in comparison to TDF, in the weight changes among ART-experienced PLWHIV.
Methods
Information on all the PLWHIV on TDF/FTC/RPV who switched to TAF/FTC/RPV between January 2017 and December 2018 was retrieved from the computerized pharmacy records. The patients were, thereafter, included in the study if they had been treated with TDF/FTC/RPV within, at least, the last 12 months, had less than 200 copies/ml of the HIV RNA in the 6 months preceding the switch and had at least two weight measures each in the years before and after the switch. The preswitch measures were single weight values closest to 12 (12_BEF) and 6 months before (6_BEF) starting TAF. The postswitch weight measures were the weight values closest to 3 (3_AFT) and 6 months after (6_AFT) starting TAF. The patients' weights were also registered 12 months after the switch (12_AFT) for those who had the available data.
The weight trend across the study period was evaluated using a generalized linear model for repeated measures and a pair comparison performed with the Bonferroni adjustment. The sub-groups were then analysed to investigate the possibility of different trends between male and female patients and those of patients with higher CD4+ T cells (>500 cells/μl), BMI greater than 25 kg/m2 or with a history of drug use (IVDU) as compared with PLWHIV without these characteristics [2,8]. Finally, the chi-square test and Fisher's exact test were used to compare the frequencies of the overweight and obesity measures at 12_BEF, 6_AFT, and for patients with the available data, 12_AFT. Patients with a BMI between 25 and 30 kg/m2 were considered as overweight and those with a BMI greater than 30 kg/m2 were considered obese. Incident obesity was defined as a BMI increase to a value greater than 30 kg/m2 among people not obese at the study baseline. All the clinical and laboratory data were retrieved from MedInfo, an online database for anonymous and automatic data collection; its use for scientific purposes was approved by the Ligurian Ethics Committee (date of approval: 28 August 2013).
All the patients signed an informed consent form in which they agreed to the anonymous use of their clinical data for scientific purposes. The study was performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments as well as the Italian national laws.
Results
We identified 305 PLWHIV who switched from TDF/FTC/RPV to TAF/FTC/RPV and had less than 200 copies/ml of the HIV RNA during the 6 months preceding the switch. Of them, 53 patients were excluded for having insufficient records of their weights. The remaining 252 patients were primarily men (N = 170, 65.5%) with a mean age of 51.2 years (standard deviation, ±9.6) and mean history of 18 (±18.2) years of HIV infection. Almost all the study participants were Caucasian (N = 249, 98.8%). The risk factor for HIV acquisition was sexual exposure in 150 cases (59.5%), intravenous drug use (IVDU) in 95 (37.7%) and other/unknown in 7 (2.8%). The mean CD4+ T-cell count was 744 (± 329) cells/μl.
At the time of the first evaluation (12_BEF), the mean weight was 73.8 (± 14.3) kg and the mean BMI was 25.3 (± 4.3) kg/m2. The mean weight did not change during the 6 months before the switch and remained 73.8 (± 14.3) kg at 6_BEF. A weight increase was noticed 3 and 6 months after the switch to TAF, becoming 77.7 (± 42.3) and 75.5 (± 14.5) kg, respectively (P < 0.0001 for trend). However, the pair comparison after the Bonferroni adjustment indicated that the weight difference between 6_BEF and 3_AFT was not significant for the entire population. The analysis was subsequently repeated to take into account the sex, baseline BMI, CD4+ T-cell count and previous IVDU (Table 1) of the patients.
The general trend in the weight gain after switching to TAF/FTC/RPV was confirmed among all the identified sub-groups (P for trend <0.005 for all, Table 1), and a significant change within the timeframe of the switch (between 6_BEF and 3_AFT) was found among the women, patients with higher BMI (>25 kg/m2), lower CD4+ T-cell count (≤500 cells/μl) and history of previous IVDU (Table 1). Two hundred and nineteen patients also had a weight measure available for 12 months after the switch. In this group, the general trend in the weight gain was confirmed, with the weights for 12_BEF, 6_BEF, 3_AFT, 6_AFT and 12_AFT, respectively, being 74.4 (± 14.2), 74.3 (± 14.2), 78.5 (± 45.0), 75.9 (± 14.7) and 76.7 (± 14.9) kg (P < 0.0001 for trend). The weight change resulting from the Bonferroni adjustment was significant between 6_AFT and 12 AFT (P < 0.0001).
Finally, the frequency of overweight and obesity was evaluated at the study's baseline and follow-up stages. At 12_BEF, 122/252 patients (48.4%) had a BMI exceeding 25 kg/m2, of which 87/252 (34.5%) were overweight and 35/252 (13.9%) obese. The proportions of overweight and obese patients rose to 94/252 (37.3%) and 39/252 (15.5%), respectively, at 6_AFT (P < 0.0001 for both), with six cases of incident obesity among all the patients who were overweight at 12_BEF and two patients who were obese at baseline but lost weight at 6_AFT. Similarly, among the 219 patients who were followed up with at 12_AFT, the proportion of overweight and obese patients, in comparison to 12_BEF, changed from 73/219 (33.3%) to 88/219 (40.2%) and 31/219 (11.8%) to 34/219 (15.5%), respectively (P < 0.0001 for both), with four cases of incident obesity in the previously overweight patients as well as one patient who was obese at the baseline but lost weight at 12_AFT.
Discussion
In this retrospective single-centre study, we found a significant weight gain among the patients who switched from TDF/FTC/RPV to TAF/FTC/RPV. This effect is likely attributable to TAF as the patients examined before and after the switch were exactly the same and did not change any other component of their ART regimen, so the involvement of an external confounding factor was little probable. The observed trend was the same in all the considered sub-groups: a stable weight was recorded before the switch, followed by an increase during the timeframe in which the switch was made, that is, 6_BEF-3_AFT. Moreover, among the patients with longer follow ups, the weight continued to increase from 6 to 12 months after the switch, suggesting the effect continued after the switch, in the year following TAF introduction. However, the change during the time of the switch, although present, was not significant for the entire study population but only for female patients, those with lower CD4+ T-cell counts and with a history of previous IVDU. This may be because of the more evident effects of the drugs on these patients' weights, in accordance with the previous data for the naïve patients [8,12], or because of the higher dispersion of the data observed among the patients without these characteristics in our study; the lack of statistical significance does not exclude that the effect exists. Among naïve patients, the weight-gain phenomenon may be, at least in part, attributable to a 'return to health', explaining the effect seen in patients with lower CD4+ T-cell counts and a history of IVDU. Less clear is the mechanism among ART-experienced patients, among whom the effects of single drugs seem less incisive [2,14] and the association with this possible mechanism appears less clear. In addition, we found that in our population of patients with long histories of HIV infection, those with a higher baseline BMI experienced a significant weight increase at the moment of the switch, a context in which the residual impact of the return-to-health phenomenon seems little probable. On the other hand, overweight patients are those more likely to have a lifestyle that influences weight gain, per se, and have been already found to be at risk of gaining weight during ART [8,15] in the absence of a direct drug effect; thus, the true mechanism responsible for the weight gain among these patients remains unclear. In this context, however, the increase in total cholesterol and cholesterol fractions that has been described after the switch from TDF to TAF [16,17] may increase the concerns linked to weight gain among people with other risk factors for cardiovascular diseases, such as those who are overweight and obese. Finally, 99% of the study participants were Caucasian, which confirms the weight gain as an adverse event also occurring in this population, also if previously reported as more evident in black people [7,8,12]. This study has several limitations. Because of its retrospective design, there may have been a selection bias when choosing the patients, as the people excluded for insufficient weight measures may have been those whose respective weights did not change at all after the switch. Moreover, the patients included in the study had high baseline CD4+ T-cell counts, and nearly half of them were overweight; thus, they could not be considered as representative of PLWHIV in other contexts. Finally, we did not collect data on the concomitant drug use and lifestyle habits that possibly influenced the weight changes during the study period.
In conclusion, TAF appears to have an impact on weight gain, similar to the effect observed among naïve patients [7,8] also in experienced PLWHIV with a long history of HIV infection and good virologic control. Alongside the choice of a switch from TDF to TAF, the variation in the cardiovascular risk among the single patients after introducing a possible element that favours weight gain should also be taken into account [16,17]. Furthermore, the weight-gain mechanism does not seem to be applicable only in the case of a return-to-health phenomenon and, hence, deserves to be studied among both naïve and ART-experienced patients.
|
|
| |
| |
|
|
|