 |
 |
 |
| |
Abstract 489. Amprenavir (APV) Plasma and Intracellular Concentrations When
Co-Administered with Ritonavir (RTV) in Twice and Once Daily Regimens in
HIV-Infected Patients
Reported by Jules Levin
|
| |
| |
This study was reported by R Garraffo and GlaxoSmithKline. APV/RTV twice and
once daily dosing is being studied in clinical trials. This study at ICAAC
reported the changes in blood levels of APV and RTV when used in combination
at once and twice daily dosing. This was a phase 1, open-label, multiple
dosing study with 3 regimen periods. 10 HIV-infected patients received the
following 3 treatments for a minimum of ten days: APV 1200mg bid (twice
daily), APV 600/RTV 100mg bid, APV 1200/RTV 200mg QD (once daily). Subjects
continued their ART including NNRTIs. However, other PIs were not permitted
during the study. Blood samples for APV and RTV plasma determination were
collected at pre-dose, 0.5, 1, 2, 4, 6, 8 and 10 hours. Additional samples
were performed at 22 and 24 hour for QD regimen. Blood samples for APV
intracellular determinations in PBMCs were collected at pre-dose, 2 and 22
hours (only for QD regimen) after dosing.
Demographic Characteristics of Patients
age: 39
weight: 69 kg
CD4: 419
HIV-RNA: 2.73 log (<1.6-5.90 range)
All patients took NRTIs including NNRTIs: Efavirenz (n=1), NVP (n=1).
RESULTS
APV Steady State PK Parameters
|
|
| |
 |
|
| |
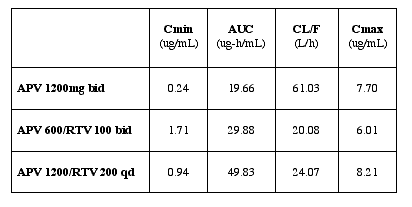
APV Steady State PK Parameters |
|
| |
 |
|
| |
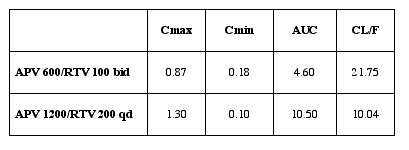
Authors reported APV exposure was highly increased; particularly, Cmin was 7
times higher (APV/RTV bid regimen) and 4 times higher (APV/RTV qd regimen)
than APV 1200mg without modification of APV Cmax and Tmax. APV clearance was
decreased by more than 50%. the principal effect of RTV is seen on APV
hepatic metabolism rather than APV intestinal absorption. Seven subjects
(7/10) receiving APV 1200mg bid alone presented APV intracellular
concentrations under the limit of quantification, vs 1 patient in APV/RTV bid
regimen and none in APV/RTV qd regimen. Cmin ratios were above 1 in the two
regimen using RTV co-administration, and the highest ratio was obtained by
APV/RTV qd. PK parameters were not altered by APV. None of the treatment
combinations were associated with an increased number of adverse events or
clinically significant lab abnormalities.
|
|
| |
|
 |
 |
|
|