 |
 |
 |
| |
How Low Does Viral Load Have To Be?
|
| |
| |
A popular question and one that has been unanswered is-how low do I have to lower my viral load? We know from several studies that reducing to viral load <50 copies/ml produces more durable viral suppression than reducing viral load to <400 copies/ml. Many have questioned whether more durable viral suppression results if viral load is reduced lower. If viral load is less than 20 or 3, does that produce more durable suppression? This study attempts to answer this question.
At ICAAC held in December 16-19, Luc Perrin, from the University of Geneva, and Abbott researchers reported on a study looking at whether patients taking Kaletra did better over 3 years if they were able to achieve one or more viral load tests of ≤ 3 copies/ml.
BRIEF SUMMARY: This study found there was no greater risk of loss of viral control through 144 weeks of follow-up in patients who did not achieve ≤3 copies/ml during the first 72 weeks of therapy in this study compared to patients who did achieve ≤3 copies/ml during the first 72 weeks. Is 144 weeks of follow-up long enough to feel confident of these results? Some doctors I spoke with about it feel 3 years is long enough follow-up but I am not convinced. Certainly, this is a good question to have been studied, and the results do show evidence that reducing viral load to <50 copies/ml and maintaining that is adequate in maintaining durable HIV suppression. But I would like to see these study results hold up for a longer period of time such as 5 -8 years. Do these results apply to drugs other than Kaletra? We don't know and perhaps not.
Studies reported this year have shown that transient repeated low-level viral load rebounds above 400 increases the risk for viral load rebound.
The objective of this study was to evaluate if the inability to achieve or consistently maintain HIV-RNA ≤3 copies/ml during the first 72 weeks of Kaletra (lopinavir/ritonavir) based therapy was predictive of subsequent loss of viral control through 144 weeks of followup.
To examine this question, they looked at patients in study M97-720, in which 100 treatment-naive patients were randomized to one of 3 Kaletra doses (200/100, 400/100, or 400/200 mg twice daily (bid). D4T and 3TC were also given after 3 weeks of Kaletra monotherapy in one patient group or from the study start in another patient group. After 48 weeks all patients began conversion to 400/100 dose.
In this study, blood samples from patients with HIV-RNA <50 copies/ml at weeks 24, 48, and 72 were analyzed using an experimental modification of the standard Roche Amplicor HIV-RNA previously described by Perrin et al. This modified assay allows for a limit of quantification of ≤3 c/ml.
There were 96 men, 4 women in study. Average age was 35. Before starting the original study the average patient viral load was 63,000 and CD4 was 326. After 144 weeks 76% of patients had <50 c/ml using the stringent ITT analysis (NC=F), and 96% had <50 c/ml using an on-treatment analysis. 79% had <400 copies/ml (ITT, NC=F), and 99% had <400 using an on-treatment analysis. Diarrhea (25%, >3 loose stools per day) was the most common adverse event. Nausea occurred at 15% rate. 17% of patients had cholesterol >300 mg/dL. 16% had triglycerides >750 mg/dL. 10% had AST/ALT >5 times upper limit of normal (ULN). 20 patients discontinued at or before week 144. Study authors reported there were 5 patient discontinuations due to adverse events possibly or probably related to study drugs.
Viral load reductions with Kaletra d4t/3TC After Week 144
|
|
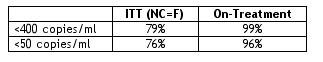 |
| |
RESULTS
This study found there was no greater risk of loss of viral control through 144 weeks of follow-up in patients who did not achieve ≤3 copies/ml during the first 72 weeks of therapy in this study compared to patients who did achieve ≤3 copies/ml during the first 72 weeks.
74% of patients with no HIV-RNA ≤3 copies/ml (n=42) had <50 c/ml at weeks 24, 48, and 72 (ITT, NC=F) had viral load <50 at week 144. 83% of patients with at least 1 HIV-RNA (n=54) <50 c/ml at weeks 24, 48, and 72 had viral load <50 at week 144. The difference between 74% and 83% was not statistically different (p=0.31).
84% of patients (n=32) with multiple HIV-RNA ≤3 c/ml (ITT, NC=F) <50 c/ml at weeks 24, 48, and 72 had viral load <50 at week 144.This compared to 77% of patients with 0 or 1 HIV-RNA ≤3 copies/ml at weeks 24, 48, and 72 who had viral load <50 at week 144. The difference between 84% and 77% was not sifnificant (p=0.44).
Interestingly, patients with >100,000 copies of viral load or <200 CD4s before the study started were less likely to achieve one or multiple HIV-RNA levels ≤3 copies/ml, but the study reported that these patients suppressed viral load (<400 and <50) equally well after 144 weeks as patients with <100,000 c/ml and >200 CD4s. 73% (ITT, NC=F analysis) with <100,000 had <50 c/ml at week 144 vs 80% of patients with >100,000 at week 144. 83% with <200 CD4s had <50 c/ml at week 144 vs 72% with >200 CD4s.
4 patients discontinued prior to week 24 and were not tested with the ≤3 c/ml assay. So the results from the study were based on 96 patients.
- 56% (54/96) on treatment demonstrated a viral load ≤3 copies/ml for at least one test. This compared to 33% (32/96) who had multiple HIV-RNA ≤3 copies/ml.
- 72% (38/53) of patients with HIV-RNA <100,000 achieved at least one HIV-RNA ≤3 copies/ml compared to only 37% (16/43) of patients with baseline viral load >100,000. 53% with <100,000 achieved multiple HIV-RNA <3 c/ml vs 9% with >100,000.
- 67% (41/61) with CD4 >200 achieved at least 1 HIV-RNA ≤3 compared to only 37% (13/35) with CD4 <200.
|
|
| |
|
 |
 |
|
|