 |
 |
 |
| |
Report from 6th Intl Congress on Drug Therapy in HIV-Infection
Reported by Mike Youle, MD, Royal Free Hospital, London, UK
|
| |
| |
Amprenavir(Agenerase Once Daily
MaxCmin2: saquinavir/r vs lopinavir/r (Kaletra)
Reducing Viral load <3 copies: Kaletra Study
AZT-d4T Cross Resistance
Rising HCV Rates and Sexual Transmission
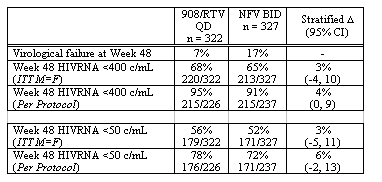
The trials and tribulations of amprenavir have not been a happy story, consisting of a drug with a high-pill burden and frequent gastro-intestinal side effects, mainly diarrhoea and bloating. Fosamprenavir (GW433908, 908) is a new formulation with distinct and now well-characterized resistance profile. The change in formulation allows for once daily dosing (QD) when combined with 200mg of ritonavir (RTV). Schurmann and colleagues from the US, Europe and South Africa presented the SOLO study, a large multi-centre, randomised, open-label study in ART-naive subjects which compared the efficacy and safety of 908/RTV QD vs nelfinavir (NFV) BID over 48 weeks [abstract PL14.4]. Six-hundred and sixty subjects (649 treated) with plasma HIV-1 RNA ≥1000 copies/mL were randomised 1:1 to 908 1400mg/RTV 200mg QD or NFV 1250mg BID in combination with ABC and 3TC BID, and were stratified at entry by viral load >100,000 or <100,000 copies/mL. Subjects generally had advanced HIV disease at baseline: median HIVRNA of 4.8 log10 c/mL with 43% >100,000c/mL; median CD4 170 cells/mm3, 20% had <50 cells/mm3; 22% had a history of AIDS defining events.
|
|
 |
| |
Diarrhea was significantly lower in the 908/RTV group (9% v 16%, p=0.008), but the incidence of other drug-related side effects was comparable. Incidences of severe lipid abnormalities were low despite the use of low dose RTV (triglycerides: 6% v 2%; cholesterol: 1% v 0; 908/RTV v NFV respectively).
These results show that the new version of amprenavir is at least as good as nelfinavir and that has a low pill burden. It would seem logical that those who are currently on amprenavir should switch to fosamprenavir when it becomes available in the clinic.
MaxCmin2: saquinavir/r vs lopinavir/r (Kaletra)
Another protease inhibitor based comparison study that had it's first airing at this Scottish meeting was the MaxCmin 2 trial that compared saquinavir soft gel/ritonavir 1000mg/100mg with lopinavir/ritonavir 400mg/100mg [abstract LB PL14.5]. The study was conducted in a wide range of patients who were naive to treatment or who were undetectable with side effects or failing therapy but who were still sensitive to the boosted protease regimens.
The MaxCmin2 trial was designed to test whether the two protease inhibitors had differences in 48 week efficacy and safety profile among patients with a clinical need for a ritonavir-boosted PI treatment. All patients also received at least two (non) nucleoside reverse transcriptase analogues (NRTI's/NNRTI's). In the protocol, one interim analysis was planned when all patients had completed 24 weeks of follow-up. As per protocol, the Peto method of repeated significance testing was used to test for differences between the treatment arms, with a p-value of 0.001 used as the significance level for the interim analysis presented to the independent data safety and monitoring board (DSMB). Differences, which did not reach this level of significance, were considered not significant.
A total of 339 patients from 28 sites in Europe, South and North America were randomised from June 2001 to January 2002, of which 326 initiated the PI to which they were assigned to receive (the ITT/exposed population - ITT/e).
Forty-nine percent of subjects were PI-naive at entry (32% antiretroviral naive), 32% were PI-experience and with HIV-RNA > 400 copies/mL and the remaining 19% PI-experienced but with HIV-RNA < 400 copies/mL. The median HIV-RNA plasma level was 4.6log10 copies/ml (40,000), and 21% had less than 400 copies/ml. Nadir CD4 cell count was relatively low with a median 98 cells/mm3.
After 24 weeks of therapy, 55 patients (17%) had discontinued the assigned PI without differences between the two treatment arms at the p=0.001 level. The primary reason for discontinuation was clinical non-fatal adverse events (n=22). Only one patient to date has discontinued the assigned PI combination because of virological failure. However a switch from the randomised treatment was not mandatory for patients that experienced protocol defined virological failure. A total of 23 patients withdrew consent or was lost to follow-up, and 93% of the patients remained under follow-up.
The DSMB has recommended that the study be continued to the planned 48 weeks. For the overall study population, the proportion of patients with HIV-RNA < 400 copies/mL (ITT/e) at baseline, week 4, week 12 and week 24 was: 21%, 53%, 72% and 77%. If switch from randomised PI was counted as being not suppressed (ITT/e/switch= failure) then the corresponding numbers were 21%, 52%, 70% and 74%. Finally, in on treatment analysis 21%, 56%, 81% and 90%, respectively, had a HIV-RNA < 400 copies/mL. The median increase of CD4+ lymphocytes from baseline to week 24 was 81 cells/µL.
A total of 90 adverse events (AEs) of at least grade 3 were recorded up to the end of September 2002, of which 24 were considered at least possibly related to either of the study PI's. The time to develop the first adverse event of at least grade 3 was comparable in the two arms. There was no statistical significant difference at the p=0.001 level between the two treatment arms in AEs grade 3 or more.
So it seems that at the present this study has not picked up major statistical differences between these two boosted PI regimens and unlike MaxCmin 1 that compared boosted saquinavir at this dosage with indinavir/ritonavir 800/100mg twice daily there is not a difference in side effect profile. The final analysis of the week 48 data is expected in late 2003.
Reducing Viral load <3 copies: Kaletra Study
One enduring question at the moment is what is the effectiveness of suppression of HIV if you go to the current lower limit of detection of most HIV RNA viral load assays of 50copies/mL. In a study by King and co-workers this was examined using a new version of a viral load test which can measure down to less than 3copies/mL [abstract PL3.3]. Whilst the difference between 3 and 50 does not seem large this represents an entire log of viral load, equivalent to the difference between 30,000 and 500,000, which we know from other studies has a disease progression consequence. At the XI International HIV Drug Resistance Workshop in Seville, Spain, in July 2002, a group from San Diego showed that adding abacavir to the regimen of patients with long-term viral suppression between 2.5 and 50 copies/mL resulted in suppression to < 2.5 copies/mL in all subjects.
One hundred antiretroviral-naive subjects were treated with lopinavir/ritonavir, lamivudine, and stavudine for 204 weeks, and viral load was measured using <400 and <50 copies/mL assays at each visit. In addition, at weeks 24, 48, and 72, viral load was also evaluated using a modified version of the standard Roche Amplicor assay, in which an additional step in the measuring process enabled the detection of as few as 3 copies/mL of HIV-1 RNA. The likelihood of viral load rebound by week 204 was compared between those who achieved viral load suppression to < 3 copies/mL by week 72 (n = 54) and those who did not stay below 3copies/mL (n = 42).
At week 204, 72 subjects had a plasma HIV-1 RNA level < 50 copies/mL (by intention-to-treat analysis, missing equals failure), while 28 had discontinued therapy. There was no significant difference in the likelihood of viral suppression at this time between those with < 3 copies/mL by week 72, compared with those with > 3 copies/mL (86% vs 83%; P = .71, log rank test). The failure rates by year are shown in the Table.
|
|
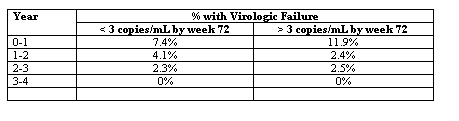 |
| |
Fifteen individuals had a viral load rebound during the follow-up period of which 7 continued therapy with the same regimen and all these had viral load < 50 copies/mL by week 204. Of the 8 subjects with virologic failure who then stopped treatment, genotypic resistance testing showed that no subjects had protease mutations, whilst 2 had had the M184V mutation associated with lamivudine use.
About 30% on this regimen developed grade 3 or 4 adverse events whilst the average increase in CD4+ cell count by week 204 was 440 cells/mm3, and appeared to be independent of baseline CD4+ cell count or starting viral load level.
This study suggests that a decrease in viral load to < 3 copies/mL might not associated with a better long-term virologic outcome to a lopinavir-ritonavir-containing regimen, compared with a decrease to < 50 copies/mL. However, the results do not address whether it may be advantageous either to intensify a regimen that is maintaining viral load at just under 50 copies/mL, or to prospectively aim to maintain viral load continuously below 3 copies/mL for a long period.
AZT-d4T Cross Resistance
Jaques Grassi and his colleagues from Gif-surYvette, France showed an interesting presentation on the intracellular metabolism of nucleoside analogue reverse transcriptase inhibitors (NRTI's) [abstract PL8.3]. Using a novel technique the researchers showed that when zidovudine (AZT) is metabolized not only is the triphosphate active form of AZT made, AZT-TP but also measurable levels occur of d4T-Tp the active form of stavudine (d4T). In 31 subjects blood was taken between 30 minutes and 15 hours after their last dose of AZT and intracellular levels of AZT-TP and d4T-TP were assessed. The later metabolite was found unexpectedly in all samples and represented between 3% and 30% of the total tri-phosphated reverse transcriptase inhibitor compared to AZT-TP. Thus giving one drug appears to produce some levels of another active anti-HIV drug. This work is supported by other observations in the test tube that show that d4T-TP is produced in cells cultured in the presence of AZT while d4T-TP was never detected in patients not receiving d4T or AZT but who were treated with other NRTI's such as lamivudine (3TC) or abacavir.
This observation indicates that d4T-TP could participate to the action and or toxicity of AZT and in any case brings a new light to the cross resistance observed between these two widely used NRTI's.
Rising HCV Rates and Sexual Transmission
Following the enduring challenge of HIV there has been a recent realization that hepatitis C virus (HCV) represents our next viral disease challenge; in fact, surveillance systems are not properly in place in most countries. For instance in Great Britain there are an estimated 300,000 HCV cases, over 90% of which are as yet undiagnosed, in comparison to approximately 33,500 people living with HIV, about 30% of whom are undiagnosed. The significance of sexual transmission in this emerging epidemic has been unclear but it appears now that there is incontrovertible evidence that this is an important method of acquisition, at least among men who have sex with men (MSM).
From the Chelsea and Westminster Hospital in London, Browne and co-workers presented some evidence that sexual transmission is responsible for an increasing incidence of HCV in HIV-infected individuals [abstract P283]. Cases of HCV were identified among subjects with a previously negative HCV antibody result that attended sexual health services from 1997 to 2002. There was a rise in the number of these HCV seroconvertors from zero during 1997 to ten in 2002. A total of 23 cases were identified, 22 of whom were male 21 known to be HIV-infected, including 2 who seroconverted to both viruses concurrently. Although 4 subjects gave a classic history of injection-drug use and needle sharing, 19 did not; these were all MSM, 15 of whom reported recent unsafe sex. Eight subjects in this cohort developed syphilis at the same time as they acquired their HCV.
All of the HIV-infected individuals diagnosed with HCV were identified by screening for HCV RNA among those with abnormal liver function, using stored blood samples to try to identify the date of acquisition. Routine antibody tests were also performed which were initially negative, with a median elapse rate from negative to HCV antibody positivity of 4 months (range 3-9.5 months). The rate of diagnosis of HCV among HIV-infected subjects found to have elevated liver function tests in the general clinic population rose throughout the time-period from 10.7% to 40%, a statistically significant change (P = .035, Chi-squared test). There appeared to be a significantly higher rate of diagnosis of HCV in 2002 compared to 1997, 5.1 cases/1000 patient years (95%CI 2.2-10.1) compared to 0 cases/1000 patient years (95%CI 0-1.2). This suggests that the HCV infection burden within the HIV-infected population has increased during that time.
This study raises a concern that the use of HCV antibody tests alone may not be sufficient to identify individuals who acquire HCV with only sexual risk factors or even in low incidence areas. Further epidemiological work addressing sexual acquisition is required to identify more clearly the risk factors for transmission as well as the outcomes of these infections. From a public health perspective, more aggressive surveillance studies should be performed, and health promotion messages need to be developed to educate those at risk.
|
|
| |
|
 |
 |
|
|