| |
Pegasys + Ribavirin: study results published
|
| |
| |
In the current issue of New England Journal of Medicine, the study of Pegasys + ribavirin conducted by Michael Fried and co-investigators was published. This study was presented publicly at the DDW Conference in the Spring of 2001.
A total of 1121 patients were randomly assigned to treatment and received at least one dose of study medication, consisting of:
--180 µg of peginterferon alfa-2a once weekly plus daily ribavirin (1000 or 1200 mg, depending on body weight), or
--weekly peginterferon alfa-2a plus daily placebo, or
--3 million units of interferon alfa-2b thrice weekly plus daily ribavirin for 48 weeks.
STUDY WITHDRAWAL
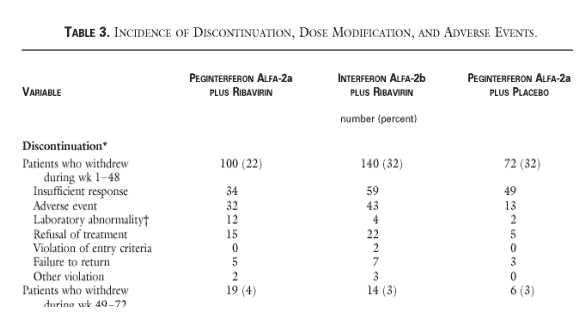
More patients withdrew for insufficient response in IFN/RBV group than Pegasys-ribavirin patients (43 vs 32). Less patients withdrew from Pegasys-ribavirin group than IFN/RBV group (34 vs 59). There were less adverse events for patients receiving Pegasys alone (13 vs 32 Peg/RBV & 43 IFN/RBV).
DOSE MODIFICATION
There were more dose modifications in Pegasys/RBV patients than IFN/RBV patients due to neuropenia. The dose changes were in the interferon: 20% modified Pegasys dose vs 5% modified IFN dose. As well, thrombocytopenia (reduced platelets) led to more dose changes in patients receiving Pegasys than in patients receiving IFN (4% Peg/RBV, 6% in Peg monotherapy arm vs <1% in IFN/RBV group).
ADVERSE EVENTS
Although side effects and adverse events are common with HCV therapy, the patients receiving Pegasys/RBV had significantly less incidence of certain side effects than patients receiving IFN/RBV: depression (22% vs 30%), myalgia (body aches: 42% vs 50%), pyrexia (43% vs 56%), rigors (shakes: 24% vs 35%).
Author's Discussion
Among patients considered to have the most treatment-resistant disease
that is, those with both HCV genotype 1 and high base-line viral levels a substantially higher proportion of those treated with peginterferon alfa-2a plus ribavirin had a sustained virologic response than of those treated with interferon alfa-2b plus ribavirin.
Early prediction of virologic response to interferon-based therapy can help identify patients who are unlikely to have a sustained response and allow clinicians the option to discontinue treatment, saving patients the side effects and cost of additional therapy. In the current study, 97 percent of patients who did not have an early virologic response to peginterferon alfa-2a plus ribavirin by week 12 never had a sustained virologic response. The incremental benefit of continuing therapy beyond 12 weeks for patients who have not had an early virologic response must be considered for each patient individually.
This study was designed to treat patients for 48 weeks, regardless of HCV genotype. Therefore, we cannot comment on shorter treatment periods. (edit note: but the Greek study of Pegasys+ribavirin reported at the European liver meeting in April 2002 found that genotype 2/3 were successfully treated with only 24 weeks therapy, with slide shows: http://www.natap.org/2002/easl/day1.htm
Don't forget these studies are in HCV momoinfected patients, not HIV/HCV coinfected patients where response rates appear to be less.
Several adverse events typically associated with the use of interferon (including influenza-like symptoms and depression) occurred less frequently with peginterferon alfa-2a, alone or in combination with ribavirin, than with interferon alfa-2b plus ribavirin. Monotherapy with peginterferon alfa-2a was generally better tolerated than the ribavirin-containing regimens. As is usual with interferon-based therapy, there were reductions in neutrophil and platelet counts with all treatments. Although these decreases were greater in patients treated with peginterferon alfa-2a plus ribavirin than in those treated with interferon alfa-2b plus ribavirin, they did not appear to be
associated with serious sequelae and were effectively managed by dose modifications. Interestingly, for patients treated with peginterferon alfa-2a plus ribavirin who had an early virologic response, completion of therapy with dose reduction was not associated with a substantial decrease in efficacy.
Virologic Response
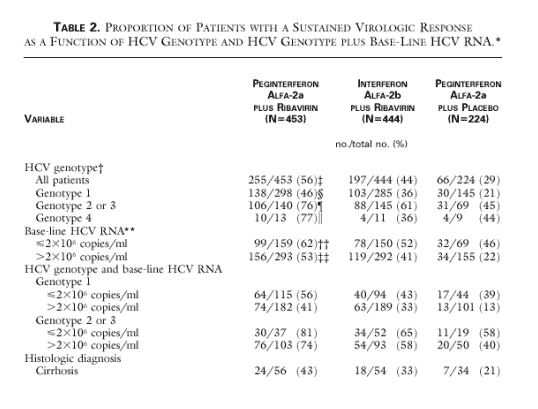
Significantly more patients treated with peginterferon alfa-2a plus ribavirin had end-of-treatment virologic responses than patients treated with interferon alfa-2b plus ribavirin (69 percent vs. 52 percent, P<0.001) or peginterferon alfa-2a plus placebo (69 percent vs. 59 percent, P=0.01) (Figure 1). Significantly more patients treated with peginterferon alfa-2a plus ribavirin had a sustained virologic response (i.e., no detectable HCV RNA 24 weeks after cessation of therapy) than those treated with interferon alfa-2b plus ribavirin (56 percent vs. 44 percent, P<0.001) or peginterferon alfa-2a plus placebo (56 percent vs. 29 percent, P<0.001).
Forty-six percent of patients with HCV genotype 1 who received peginterferon alfa-2a plus ribavirin had a sustained virologic response, as compared with 36 percent of those who received interferon alfa-2b plus ribavirin (P=0.01) and 21 percent of those who received peginterferon alfa-2a plus placebo (P<0.001). Among the patients with HCV genotype 2 or 3, significantly more of those treated with peginterferon alfa-2a plus ribavirin had a sustained virologic response than of those treated with interferon alfa-2b plus ribavirin (76 percent vs. 61 percent, P=0.005). Among patients with HCV genotype 1 and high base-line viral RNA levels (more than 2 million copies per milliliter), 41 percent of those receiving peginterferon alfa-2a plus ribavirin had a sustained virologic response, as compared with 33 percent of those receiving interferon alfa-2b plus ribavirin. Among patients with low viral load (<2 million copies) and genotype 1 Pegasys+ribavirin had better Sustained response than interferon+ribavirin (56% vs 43%). Among patients with cirrhosis, 43 percent of those treated with peginterferon alfa-2a plus ribavirin and 33 percent of those treated with interferon alfa-2b plus ribavirin had a sustained virologic response. See table 2 of viral responses at end of this report.
Independent Factors Associated with a Sustained Virologic Response
In multivariable analyses to identify predictors of sustained virologic response among patients who received peginterferon alfa-2a plus ribavirin, our final multiple logistic-regression model, including the following factors, was entered in the final stepwise regression analysis: sex, race (white vs. nonwhite), age (40 years vs. >40 years), body weight (75 kg vs. >75 kg), pretreatment viral load (2 million copies per milliliter vs. >2 million copies per milliliter), pretreatment alanine aminotransferase quotient (>3 vs. 3), pretreatment histologic diagnosis (cirrhosis vs. noncirrhosis), and HCV genotype (1 vs. non-1).
Three factors independently and significantly increased the odds of achieving a sustained virologic response: an HCV genotype other than 1 (odds ratio, 3.25; p<.001), an age of 40 years or less (odds ratio, 2.60;p<.001), and a body weight of 75 kg or less (odds ratio, 1.91; P=0.002).
Predictive Value of Early Virologic Response
By week 12, 86 percent of patients (390 of 453) treated with peginterferon alfa-2a plus ribavirin had had a virologic response, defined as a 2-log decrease from baseline HCV RNA levels (97 patients) or no detectable serum HCV RNA (293 patients). The absence of an early virologic response was not associated
with early treatment discontinuation (before week 12) or dose modification (data not shown). Of those with early virologic responses, 65 percent subsequently had a sustained virologic response. Those with no detectable HCV RNA by week 12 were more likely to have a sustained virologic response than those who had only a 2-log decrease in HCV RNA (221 of 293 vs. 32 of 97). In contrast, among the 63 patients who did not have an early virologic response, 61 (97 percent) did not have a sustained virologic response.
Safety
The proportions of patients withdrawn from treatment because of laboratory abnormalities or other adverse events were similar in the groups receiving peginterferon alfa-2a plus ribavirin (3 percent for laboratory abnormalities and 7 percent for other adverse events), peginterferon alfa-2a plus placebo (1 percent and 6 percent, respectively), and interferon alfa-2b plus ribavirin (1 percent and 10 percent)
The median hemoglobin values decreased between weeks 1 and 8 in all treatment groups, then stabilized, and then returned to near base-line values after treatment was completed. The maximal decrease was greater in patients treated with peginterferon alfa-2a plus ribavirin (3.7 g per deciliter) or interferon alfa-2b plus ribavirin (3.6 g per deciliter) than in patients treated with peginterferon alfa-2a plus placebo (2.2 g per deciliter). There was no association between the incidence of serious cardiovascular events and the incidence of anemia.
The median neutrophil counts decreased from base line in all treatment groups, particularly during the first two weeks of treatment, and then stabilized for the remainder of the treatment period, increasing rapidly to base-line values after the completion of treatment. Four patients (three receiving peginterferon alfa-2a plus ribavirin and one receiving interferon alfa-2b plus ribavirin) discontinued treatment because the neutrophil count was below 500 per cubic millimeter. The median platelet counts remained close to base-line values throughout treatment with interferon alfa-2b plus ribavirin but decreased progressively during the first eight weeks of treatment with
peginterferon alfa-2a plus ribavirin or placebo, before stabilizing. After treatment was completed, the median platelet counts returned to normal within four weeks. Two patients with thrombocytopenia (one receiving peginterferon alfa-2a plus placebo and the other receiving interferon alfa-2b plus ribavirin) had serious bleeding. Five patients (four receiving peginterferon alfa-2a plus ribavirin and one receiving peginterferon alfa-2a plus placebo) discontinued treatment because of thrombocytopenia.
Most adverse events in all study groups were those commonly associated with interferon-based treatment. Patients treated with peginterferon alfa-2a plus
ribavirin or placebo had a lower incidence of influenza-like symptoms, such as pyrexia, myalgia, and rigors, than those treated with interferon alfa-2b plus ribavirin. Although few patients had a history of depression or active depression at base line (1 to 2 percent and 3 to 5 percent, respectively), a substantial minority reported depression during the study. Patients treated with peginterferon alfa-2a plus ribavirin or placebo had a lower incidence of depression than those treated with interferon alfa-2b plus ribavirin (22 percent and 20 percent vs. 30 percent). Three patients died after the end of treatment. One patient who had received interferon alfa-2b plus
ribavirin died of hypertensive heart disease, and two who had received peginterferon alfa-2a plus placebo died, one from drowning and the other from liver cancer. None of the deaths were considered related to treatment.
| |
 |
 |
| |
More patients withdrew for insufficient response in IFN/RBV group than Pegasys-ribavirin patients (43 vs 32). Less patients withdrew from Pegasys-ribavirin group than IFN/RBV group (34 vs 59). There were less adverse events for patients receiving Pegasys alone (13 vs 32 Peg/RBV & 43 IFN/RBV).
DOSE MODIFICATION
There were more dose modifications in Pegasys/RBV patients than IFN/RBV patients due to neuropenia. The dose changes were in the interferon: 20% modified Pegasys dose vs 5% modified IFN dose. As well, thrombocytopenia (reduced platelets) led to more dose changes in patients receiving Pegasys than in patients receiving IFN (4% Peg/RBV, 6% in Peg monotherapy arm vs <1% in IFN/RBV group).
ADVERSE EVENTS
Although side effects and adverse events are common with HCV therapy, the patients receiving Pegasys/RBV had significantly less incidence of certain side effects than patients receiving IFN/RBV: depression (22% vs 30%), myalgia (body aches: 42% vs 50%), pyrexia (43% vs 56%), rigors (shakes: 24% vs 35%).
| |
 |
 |
|
|
|