 |
 |
 |
| |
Kaletra Drug levels in HCV/HIV Coinfection
|
| |
| |
Evaluation of the Multiple-Dose Pharmacokinetics of Lopinavir/Ritonavir (LPV/r) in HIV and HCV Co-Infected Patients with Mild or Moderate Hepatic Insufficiency
9th European AIDS Conference, Warsaw, Poland, 26–29 October 2003 Presentation #F2/6
J Arribas1*, F Pulido2, J Peng3, S Kemmis3, JL Li3, A Lorenzo1, C Cepeda2, T Reisch3,J Moseley3,K Grebner3, JA Cabanillas4, B DaSilva3, B Bernstein3, YL Chiu3, R Bertz3. 1 H. La Paz. HIV Unit, Madrid, Spain; 2 H. 12 de Octubre. HIV Unit, Madrid, Spain; 3 Abbott Laboratories, Abbott Park, Illinois, USA; 4 Abbott International, Spain
Summary
Kaletra (400 LPV/100 RTV) was given to study patients with liver (hepatic) impairment (defined by Child-Pugh Score) for 14 days. LPV Cmax and AUC are increased by 20-30% in both mild and moderate HI; Cmin by 79%. No significant differences in the PK parameters between mild and moderate HI were noted. Total plasma RTV concentrations are increased in patients with hepatic impairment relative to control. RTV concentrations are increased by a greater magnitude in moderate HI compared to mild HI. In general, Cmax and AUC of RTV increase by 1.6- and 1.4-fold in mild HI and by 3.2- and 2.8-fold in moderate HI. RTV plasma concentrations may increase more in moderate HI due to less efficient hepatic extraction relative to mild HI. The authors conclude: caution is warranted when LPV/r is administerd to patients with mild or moderate hepatic impairment. No recommendatins on dosing were provided by study authors.
Introduction
Lopinavir/ritonavir (LPV/r) is an HIV Protease Inhibitor (PI) approved worldwide for the treatment of HIV in combination with other antiretrovirals. Lopinavir (LPV) is eliminated by CYP3A-mediated metabolism and ritonavir (RTV) is a potent inhibitor of LPV metabolism. Metabolism of LPV is induced after multiple dosing of LPV/r. LPV clearance may be decreased further in hepatic impairment (HI).
Objective
To assess the effect of mild and moderate HI on the pharmacokinetics (PK) of LPV/r after multiple dosing in HIV-infected subjects.
Study Design
This was a phase 1, open-label, two-center study. 24 patients had stable HIV disease and were receiving LPV/r (Kaletra) at 400/100 mg twice daily (bid) with food for >6 weeks prior to study entry. All patients with hepatic impairment (HI) had evidence of hepatic fibrosis by history. Patients were receiving background NRTIs and other concurrent medications which were not known to affect LPV or RTV concentrations. The 14 day stydy period included adherence monitoring by MEMs caps. Subjects were not allowed to take NNRTIs, other protease inhibitors or enzyme inducers. 12 patients were in control group (4 female-8 male), 6 patients (2 female-4 male) had mild HI (Child-Pugh 5-6), and 6 patients (1 female-5 male) had moderate HI (Child-Pugh 7-9).
Trough plasma samples were collected on Study Day 7 and on one day between Study Days 10 and 12, inclusive, and Study Day 14.
Plasma samples were collected pre-dose and at 2, 4, 6, 8, 10 and 12 hours post dose for LPV and RTV and at pre-dose and 6 h for LPV protein binding. Breakfast did not include grapefruit juice. On day 14, 12 hr PK/protein binding was performed.
The Child-Pugh classification was determined by the sum of the scores for bilirubin, albumin, prothrombin time (PT), encephalopathy, and ascites.
90% confidence intervals for the bioavailability of HI vs. control were obtained for log-transformed Cmax , Cmin , Ctrough and AUC-12 using analysis of covariance (ANCOVA).
Patient Demographics
All 12 patients with mild and moderate HI had HCV. 1 patient with moderate HI also had HBsAg. No patients in control group had HBV or HCV. Average age: 39-41; weight: 65 kg in mild HI; 69 kg in moderate HI; 71 kg in control group.
RESULTS
Compliance and Tolerability
--Overall, subjects displayed very high adherence to study drug.
--One treatment emergent adverse event of mild diarrhea was reported; it occurred 22 days post study period and was attributed to tenofovir.
--Mean hepatic function values (AST, ALT, alkaline phosphatase, bilirubin) did not change over the time of the study period.
--The study regimen was well tolerated by all subjects.
Drug Levels
LOPINAVIR
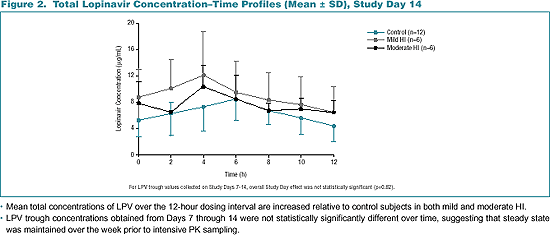
Mean total concentrations of LPV over the 12-hour dosing interval are increased relative to control subjects in both mild and moderate HI.
LPV trough concentrations obtained from Days 7 through 14 were not statistically significantly different over time, suggesting that steady state was maintained over the week prior to intensive PK sampling.
|
|
| |
| |
 |
|
| |
| |
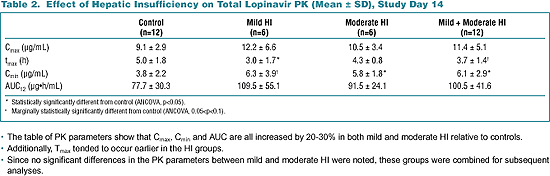
Table 2. Effect of Hepatic Insufficiency
The table of PK parameters show that Cmax, Cmin and AUC are all increased by 20-30% in both mild and moderate HI relative to controls.
Additionally, T max tended to occur earlier in the HI groups.
Since no significant differences in the PK parameters between mild and moderate HI were noted, these groups were combined for subsequent analyses.
|
|
| |
| |
 |
|
| |
| |
Protein Binding of LPV, Study day 14
--The unbound fraction of LPV in plasma was similar in mild and moderate HI.
--Relative to control subjects, the % unbound was about 30% higher in subjects with HI.
|
|
| |
| |
 |
|
| |
| |
LPV Total and Unbound AUC-12, day 14
--LPV AUC is approximately 30% higher in hepatically impaired subjects relative to control subjects (p=0.12).
--When accounting for protein binding differences, unbound LPV AUC is 68% higher in subjects with HI relative to controls (p<0.02).
--Total LPV C max is increased by 20% --AUC by 30% --Cmin by 79%. --Unbound LPV C max is increased by 56%, AUC by 68% and C min by 130%.
RITONAVIR LEVELS
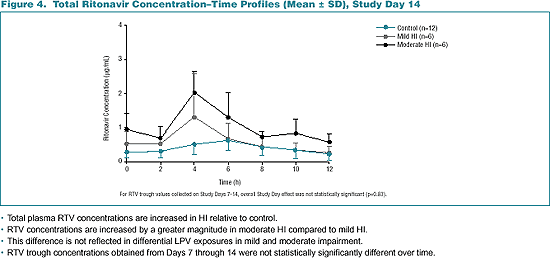
--Total plasma RTV concentrations are increased in HI relative to control. --RTV concentrations are increased by a greater magnitude in moderate HI compared to mild HI. --This difference is not reflected in differential LPV exposures in mild and moderate impairment. --RTV trough concentrations obtained from Days 7 through 14 were not statistically significantly different over time.
In general, Cmax and AUC of RTV increase by 1.6- and 1.4-fold in mild HI and by 3.2- and 2.8-fold in moderate HI.
RTV plasma concentrations may increase more in moderate HI due to less efficient hepatic extraction relative to mild HI.
|
|
| |
| |
 |
|
| |
| |
DISCUSSION and CONCLUSIONS
Mild and moderate HI showed similar effects on LPV PK, resulting in:
--30% increase in total AUC 12 and 20% increase in total C max .
--The % unbound LPV was ~30% higher in HI than in the control group.
--68% increase in unbound AUC-12 and 56% increase in unbound C-max after accounting for differences in LPV protein binding.
Hepatic impairment had a greater effect on low dose RTV in the moderate vs. mild HI group.
--The mean RTV AUC-12 observed in the subjects with moderate HI in this study was ~18% of that observed after clinical doses of RTV (600 mg BID).
--Caution is warranted when LPV/r is administered to patients with either mild or moderate hepatic impairment. |
|
| |
|
 |
 |
|
|