 |
 |
 |
| |
Evidence Suggesting Mitochondrial Toxicity in HIV/HCV Co-infected Patients Receiving Ribavirin and Didanosine
|
| |
| |
FDA Changes Ribavirin & DDI Labels: "Co-Adminstration of Ribavirin and DDI Not Recommended"
This study was reported at the Retrovirus Conference by the FDA and Ken Sherman, MD (University of Cinncinati). As a result of searching the FDA database for reported toxicities associated with taking ribavirin and ddI together or any other NRTI, 31 cases of mitochondrial toxicity were found. The FDA reporting system is voluntary, so the study authors said there is a potential for under-reporting. The authors report finding in this study a significant increase in risk of MT among patients who received concomitant RBV and ddI (with and without d4T) compared to use of RBV with other NRTIs. They report that in this data set concomittent use of ddI and ribavirin appeared to be associated with an approximately 5-fold increased likelihood of events suggestive of mitochondrial toxicity compared to the use of ribavirin with other NRTIs. They recommend ribavirin and ddI should be co-administered with caution, and ddI should be suspended if signs or symptoms suggestive of mitochondrial toxicity develop. The FDA will inclide in FDA labels for ribavirin and ddI: "Co-administration of [ribavirin] and didanosine is not recommended. Reports of fatal hepatic failure, as well as peripheral neuropathy, pancreatitis, and symptomatic hyperlactatemia/lactic acidosis have been reported in clinical trials."
Several cases suggestive of Mitochondrial Toxicity (MT) (e.g., pancreatitis, lactic acidosis with liver failure and/or hepatic steatosis) in HIV/HCV co-infected patients (pts) treated with ribavirin (RBV) and didanosine (ddI) have been reported. At conferences over the past year or two there have been reports of findings of toxicities for patients taking ddI, d4T and other nukes who add ribavirin and interferon therapy and experience the toxicities discussed in this abstract presented by the FDA at the Retrovirus 2003 Conference. ICAAC 2001, Barcelona, and others conferences have had such reports. The possibility of experiencing these toxicities has been low in each study presented that report these findings, But it is important to bear in mind that some individuals can experience this toxicity and for doctors to monitor closely for it. It may also be appropriate to consider changing NRTI therapy before starting interferon/ribavirin treatment. A liver friendly HAART regimen ought to be considered at all times for patients with hepatitis C and B. There was a time we did not consider this but now we realize this.
Two reports from clinical trial databases estimated the risk of mitochondrial toxicity (MT) to be 4-23 fold higher amongst patients who received concomitant RBV and ddI compared to patients treated with other NRTIs. Individual cases suggestive of fatal and non-fatal MT in HIV/HCV co-infected patients receiving RBV and NRTIs, most commonly ddI and d4T, have been reported in the literature.
Mitochondrial Toxicity [MT] (e.g., pancreatitis, lactic acidosis with liver failure and/or hepatic steatosis) has been described in patients treated with anti-HIV nucleoside reverse transcriptase inhibitors (NRTIs). In vitro data demonstrate that IMPDH inhibitors, such as ribavirin (RBV), either antagonize (zidovudine [ZDV] and stavudine [d4T]) or promote (didanosine [ddI] and abacavir [ABC]) conversion of NRTIs to their active metabolite.
The FDA's Adverse Event Reporting System (AERS) was searched on 21 May 2002 and updated 21 November 2002 for all reports of events in patients taking ribavirin and any of the seven approved NRTIs:
Didanosine (ddI)
Stavudine (D4T)
Lamivudine (3TC)
Tenofovir (TDF)
Zidovudine (AZT)
Abacavir (ABC)
Zalcitabine (DDC)
RESULTS
There were 31 cases reported of mitochondrial toxicity.
The reported cases of mitochondrial toxicity events were:
--pancreatitis/increased lipase (lipase is a blood test): 21 cases
--lactic acidosis/increased lactate (lactate is a blood test): 20 cases
--increased LFTS: 8 cases
--hepatic steatosis (fat accumulation in the liver): 6 cases
--increased CK: 1 case
--neuropathy: 1 case
--multiorgan failure: 1 case
Average age of patients for whom MT was reported was 40 years; 22 were male (71%) & 7 female (23%); average weight was 66 kg (145 lbs). There were 5 deaths, all due to lactic acidosis: 1 with concurrent pancreatitis, 1 with concurrent liver failure.
NRTI USE
Of the 31 patients reporting mitochondrial toxicity:
--ddI: 27 patients (87%), 20/27 were also taking d4T
--d4T: 22 (71%)
--3TC: 9 (29%)
--AZT: 1
--Abacavir: 5
--Tenofovir: 3
--DDC: 0
The average duration of ddI therapy for patients with reported MT was 18 months; 19 months for d4T; 20 months for 3TC; but for ribavirin it was about 3 months. So apparently toxicity may occur within a couple of months after adding ribavirin. But according to data from this study it appeared that toxicities occurred as early as 1-2 months after adding RBV and as late as 10 months after adding RBV.
DEATHS
16% (5/31) of the patients with reported cases of MT died. All deaths were due to lactic acidosis: 1 with concurrent pancreatitis, 1 with concurrent liver failure.
All 5 patients were taking ddI: 3/5 also taking d4T in combination; 2/5 taking TDF along with ddI.
|
|
 |
| |
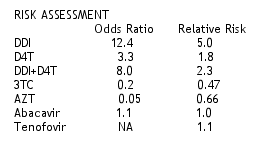
The authors concluded that in this data set, concomitant use of ddI and RBV appeared to be associated with an approximately 5-fold increased likelihood of events suggestive of MT compared to use of RBV with other NRTIs. RBV and ddI should be co-administered with caution, and ddI should be suspended if signs or symptoms suggestive of MT develop. There was a 1.8-fold increased risk of events suggestive of MT compared to use of RBV with d4T; 2.3-fold increased risk when ddI/d4T was used. But the risk of an event suggestive of MT was lower when using 3TC, AZT, abacavir and Tenofovir compared to using the other NRTIs. Based on these odds ratio the it is unlikely that 3TC and AZT would result in an event, and the risk associated with abacavir was also low.
The authors also reported:
--No association was found between sex and body weight, and the development of MT.
--No association was found between protease inhibitors or non-nucleoside reverse transcriptase inhibitors.
--d4T can cause or worsen MT and appears to pose a risk when co-administered with RBV. However, in vitro RBV antagonizes phosphorylation of d4T. Thus, the contribution of d4T to development of MT is uncertain.
Limitations of the AERS database:
--This is a voluntary reporting system
--Pancreatitis, lactic acidosis, and hepatic steatosis are labeled events for nucleoside analogues, resulting in potential under-reporting
--The incidence of an adverse event cannot be calculated with confidence due to under-reporting and uncertainty regarding the total number of patients taking ribavirin and didanosine.
AUTHOR CONCLUSION/RISK MANAGEMENT
(1) We found a significant increase in risk of MT among patients who
received concomitant RBV and ddI (with and without d4T) compared
to use of RBV with other NRTIs, which may be related to RBV's
promotion of ddI's conversion to its active metabolite.
(2) Change to FDA Product Labels:
The RBV and ddI product labeling will include the following precautionary language:
"Co-administration of [ribavirin] and didanosine is not recommended. Reports of fatal hepatic failure, as well as peripheral neuropathy, pancreatitis, and symptomatic hyperlactatemia/lactic acidosis have been reported in clinical trials."
Poster #763, Retrovirus Conference 2003.
|
|
| |
|
 |
 |
|
|