 |
 |
 |
| |
In Vitro Cross-Resistance HBV Drugs
|
| |
| |
Reported by Jules Levin
"IN VITRO CROSS-RESISTANCE TESTING OF ADEFOVIR, LAMIVUDINE, TELBIVUDINE (L-DT), ENTECAVIR AND OTHER ANTI-HBV COMPOUNDS AGAINST FOUR MAJOR MUTATIONAL PATTERNS OF LAMIVUDINE-RESISTANT HBV"
William Delaney IV, Huiling Yang, Xiaoping Qi, A Sabogal, Michael Miller, Shelly Xiong, Gilead Sciences, Inc., Foster City, CA
Four major mutational patterns of lamivudine-resistant HBV are observed in patients who fail lamivudine therapy. The L180M+M204V mutant HBV is observed most frequently in ~ 60% of lamivudine-resistant patients. Three other patterns (V173L+L180M+M204V [17%], M204I [11%], and L180M + M204I [11%]) (Westland et al, JVH 2004;11:1-7)are also frequently observed. All four mutant HBV strains are highly resistant to lamivudine. However, other anti-HBV agents may display differential antiviral efficacy against different patterns of lamivudine-resistant HBV.
The aim of this study was to determine the cross-resistance profiles of clinical anti-HBV candidates against different patterns of lamivudine-resistant HBV.
Five stable cell lines expressing high levels of wild-type or lamivudine-resistant HBV (L180M+M204V, V173L+L180M+M204V, M204I, and L180M+M204I) were used for drug susceptibility testing. Eleven anti-HBV compounds (including entecavir, L-dT, and emtricitabine that are in phase 3 clinical trials) were tested in all five cell lines. For EC50 determination, cells were seeded in 96 well plates and treated with compounds for one week. HBV DNA was quantified in cytoplasmic extracts by real-time PCR as described previously (Weinberger. Virol. Meth. 85:75).
Results
L-nucleoside analogs lamivudine, emtricitabine, L-dT, L-dC, L-dA, and clevudine demonstrated >100-fold cross resistance in the four cell lines expressing the four different patterns of lamivudine-resistant HBV.
|
|
| |
| |
 |
|
| |
| |
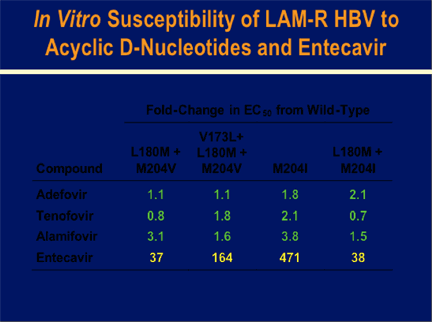
Acyclic phosphonate nucleotides adefovir, tenofovir, and alamifovir (MCC-478) retained near wild-type efficacy (0.7 to 3.8-fold increases in EC50) in cell lines expressing the four patterns of lamivudine-resistant HBV.
Entecavir demonstrated variable efficacy in the different cell lines, however all patterns of lamivudine-resistant HBV were significantly less susceptible to entecavir compared to wild-type HBV. While the M204I mutant HBV showed the greatest fold reduced susceptibility (471-fold) to entecavir, the other patterns showed 37- to 164-fold reduced susceptibility.
|
|
| |
| |
 |
|
| |
| |
DXG showed marginal anti-HBV activity with EC50 of 63 micromolar against wild-type HBV. The M204I (±L180M) mutant HBV displayed >5-fold resistance to DXG.
Delaney concluded that Adefovir, tenofovir, and alamifovir demonstrated consistent efficacy against wild-type and different patterns of lamivudine-resistant HBV. All patterns of lamivudine-resistant HBV were highly resistant to the L-nucleosides lamivudine, emtricitabine, L-dT, L-C, L-dA, and clevudine. Entecavir and DXG demonstrated varying levels of resistance to the four different patterns of lamivudine-resistant HBV.
|
| |
|
 |
 |
|
|