 |
 |
 |
| |
LdT, new HBV Drug: week 52 phase II results; Viral Dynamics
|
| |
| |
Reported by Jules Levin
"HBV Viral Dynamics in a Comparative Clinical Trial of Telbivudine (LdT), Lamivudine, and the Combination in Patients with Chronic Hepatitis B"
XIAO-JIAN ZHOU1, RICHARD E BOEHME1, NATHANIEL A BROWN1, GEORGE CHAO1, CHING-LUNG LAI2
1Idenix Pharmaceuticals, Inc., Cambridge, MA, USA;
2University of Hong Kong
Zhou reported this background on LdT (telbivudine).
--Specific inhibitor of HBV polymerase; not active against HIV or other viruses
--Inhibits primarily 2nd strand DNA synthesis
--Favorable preclinical safety profile
--Once daily oral dosing indicated by PK
Phase I/II dose escalation trial results:
(Lai et al, Hepatology 40(3): 719-26, 2004)
--Marked HBV suppression after 4 weeks: 3.4 -- 3.8 log10 with 400 -- 800 mg/day
--Excellent safety: no dose-related or dose-limiting toxicities
TELBIVUDINE 1-YEAR PHASE IIB CLINICAL TRIAL
104 patients who were treatment-naïve & HBeAg+ were randomized blindly in a 52 week trial to 1 of these 5 once daily treatment regimens:
lamivudine 100 mg
telbivudine 400 mg
telbivudine 600 mg
telbivudine 400 + lamivudine 100
telbivudine 600 + lamivudine 100
There were 15 sites in Hong Kong, Singapore, US, Canada & France. Endpoints were antiviral efficacy (serum HBV DNA reduction) and clinical efficacy (HBeAg seroconversion & ALT normalization).
BASELINE CHARACTERISTICS
There were 19-22 patients in each arm.
Median serum HBV DNA (log copies/ml) was 8.96 to 9.28.
Median serum ALT was 122 to 152 IU/L (range from 35 to 1600).
All patients were HBeAg+ & HBsAg+ at entry.
Mean age was 37 yrs (18-68).
81% male.
86% Asian.
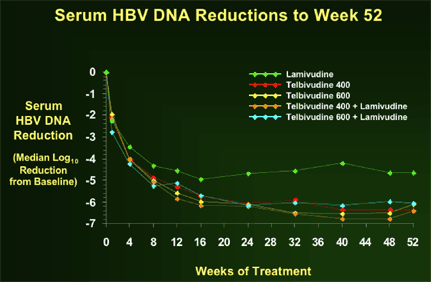
Key Clinical Results at Week 52: Phase IIb trial
There was little difference between the 4 telbivudine arms in terms of HBV DNA reduction. LdT 400 & 600 mg doses had similar viral load reductions and both of the LdT+LAM combinations arms provided no added benefit in terms of HBV DNA reductions. But all 4 LdT arms showed better HBV DNA reductions compared to lamivudine.
| Telbivudine | Lamivudine | Combo | | n=44 | (n=19) | n=41 | | Median reduction (Log10)* | -6.43 | -4.66 | -6.06 | | % PCR Negativity | 61% (27)* | 32% (6) | 49% (20) | | % ALT normalization | 86% (38)* | 63% (12) | 78% (32) | | % HBeAg seroconversion | 31% (13) | 21% (4) | 17% (7) | | % Viral breakthroughs | 4% (2) | 21% (4) | 12% (5) |
|
|
| |
| |
 |
|
| |
| |
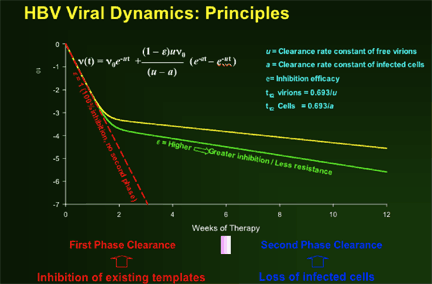
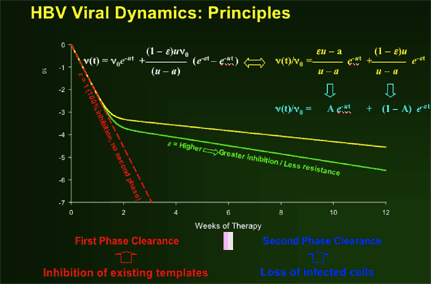
HBV Viral Dynamics Study: A retrospective analysis
The purpose of the viral dynamics study:
--To determine the comparative HBV viral dynamics in patients receiving lamivudine, telbivudine and the two drugs in combination
--To investigate relationship of HBV early viral dynamics under potent antiviral therapy to efficacy outcomes at Week 52
The authors concluded:
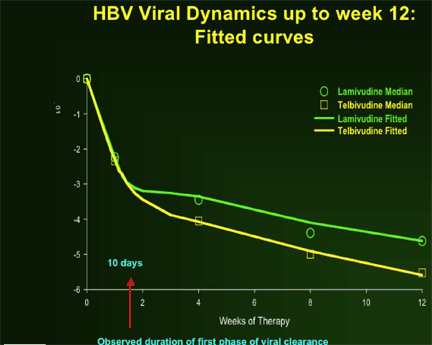
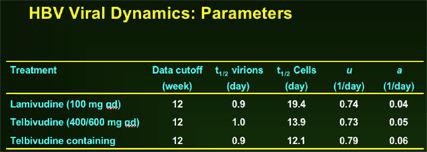
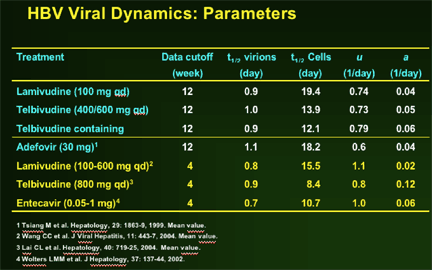
--Greater anti-HBV activity of telbivudine, compared to lamivudine, during first 2 weeks demonstrated by extended 1st phase viral clearance. Difference maintained through 2nd phase to Week 52.
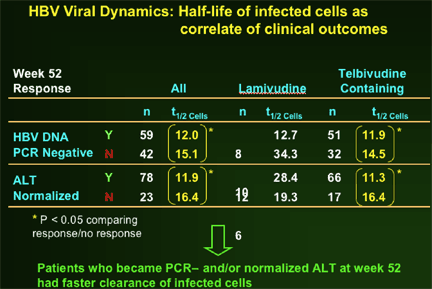
--Greater second phase viral clearance quantitatively related to estimated t1/2 of infected cells by viral dynamic modelling: Telbivudine data → shortest estimate of t1/2 of infected cells (ca. 8 days, vs. previous reports of 10-100 days)
--Viral kinetic analysis confirms that earlier and more profound HBV suppression is associated with greater efficacy on later clinical endpoints (HBeAg response, ALT normalization), and with viral clearance (by PCR assays) vs viral breakthrough
|
|
| |
| |
 |
|
| |
| |
|
|
| |
| |
 |
|
| |
| |
|
|
| |
| |
 |
|
| |
| |
|
|
| |
| |
 |
|
| |
| |
|
|
| |
| |
 |
|
| |
| |
|
|
| |
| |
 |
|
| |
| |
|
| |
|
 |
 |
|
|