 |
 |
 |
| |
Entecavir Resistance
|
| |
| |
"Emergence of Entecavir Resistant Hepatitis B Virus After One year of Therapy in phase II & III studies is Only Observed in Lamivudine Refractory Patients"
presented by Richard Colonno, Bristol-Myers Squibb and others at AASLD (October 2004), poster 2464
ABSTRACT
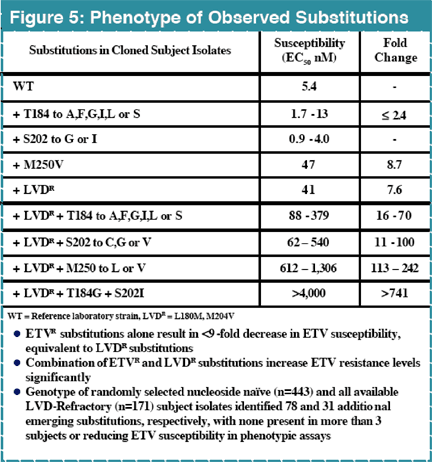
Background: Entecavir (ETV) is a potent and selective inhibitor of hepatitis B virus (HBV), with demonstrated efficacy in subjects with chronic HBV infection, including those who were lamivudine (LVD) refractory. Previous analyses of two HBV subjects experiencing a virologic rebound in Phase II trials identified emerging substitutions at residues rtT184, rtS202, and rtM250 within LVD-resistant (LDVR) HBV as critical for achievement of high ETV resistance (ETVR) levels1. The presence of either of these 3 substitutions or LVD R substitutions alone resulted in only modest decreases in ETV susceptibility in vitro1,2.
Methods: Clinical samples from ETV studies 022 and 027 (nucleoside naïve subjects) and studies 014 and 026 (LVD -refractory subjects) were monitored for the emergence of ETVR. Genotypic analysis compared HBV reverse transcriptase (RT) sequences amplified from patient serum DNA during therapy with sequences at study entry and with a consensus HBV sequence determined from 250 HBV genome sequences. HBV phenotypes were determined using antiviral assays in HepG2 cells measuring HBV DNA yields after transfection of recombinant plasmids containing patient RT sequences.
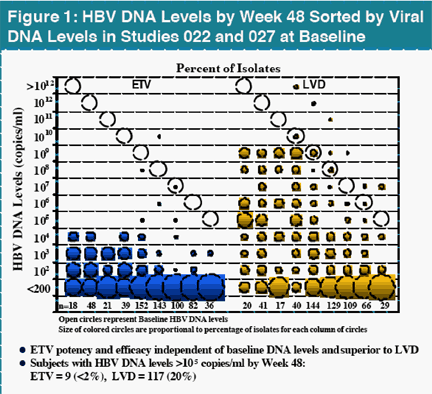
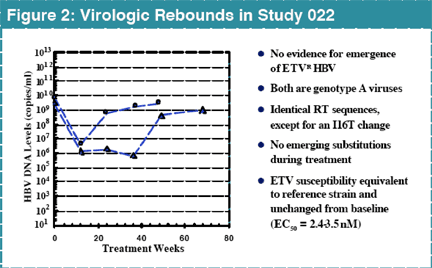
Results : There was no evidence of ETV R emergence in any nucleoside naïve subject by Week 48. Genotyping of 434 samples (223 HBeAg+, 211 HBeAg -) randomly selected from nucleoside treatment naïve subjects at Week 48 failed to identify any amino acid changes associated with decreased susceptibility to ETV (0.5 mg daily). This included the 2 subjects experiencing a virologic rebound on ETV in Study 022.
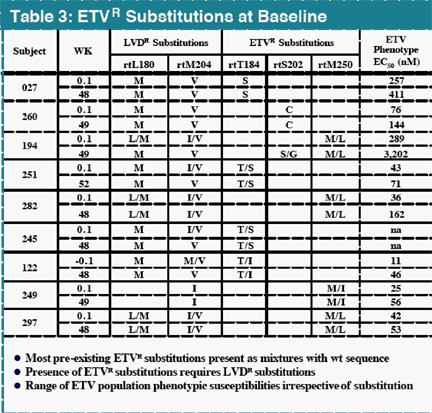
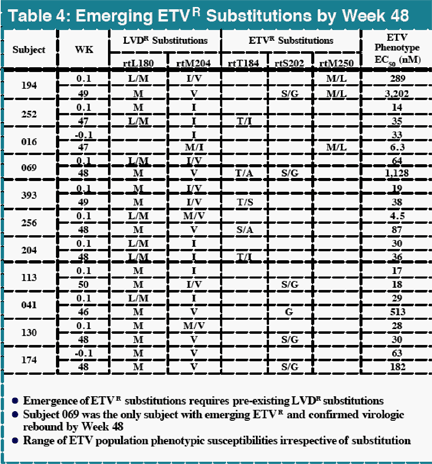
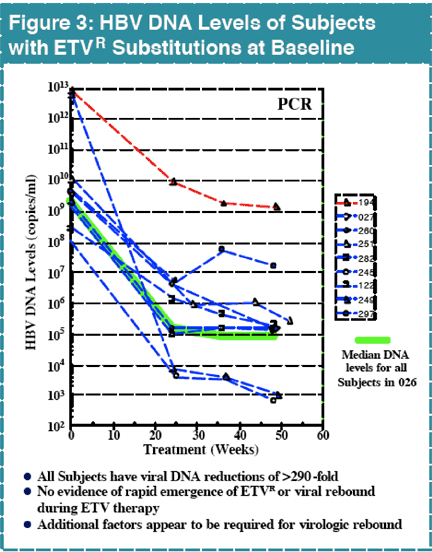
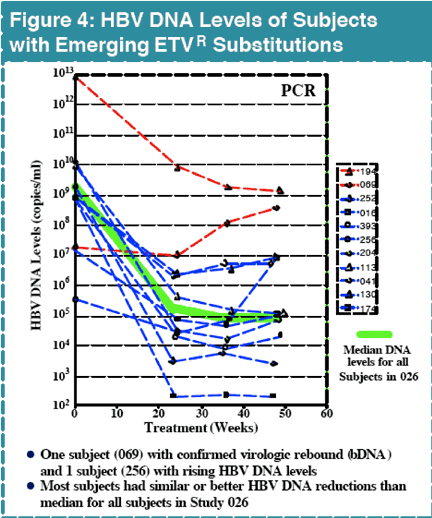
In contrast, there were 25 subjects with virologic rebound on LVD by Week 48, with 19 displaying evidence of LVDR substitutions at residues rtL180 and/or rtM204. Among the 181 LVD-refractory subjects treated with 1.0 mg daily ETV, one subject experienced a confirmed virologic rebound due to ETVR emergence by Week 48. Genotypic analysis of all subjects in studies 014 and 026 identified 11 (<7%) with evidence of emerging substitutions at residues rtT184, rtS202 or rtM250, and all of these substitutions emerged in a background of LVDR substitutions. ETVR substitutions were observed in 22 LVD refractory subjects at baseline, indicating that prior LVD use can select for these key substitutions. All subjects with ETVR substitutions in a LVDR background at baseline experienced at least a 290-fold reduction in HBV DNA levels.
Summary: There was no evidence of ETVR emergence in nucleoside treatment naïve subjects and 1% of LVD refractory subjects treated with ETV experienced a virologic rebound due to ETVR emergence by Week 48. Phenotypic ETVR required the presence of pre-existing LVDR substitutions. Coupled with superior clinical efficacy to LVD and a comparable safety profile, ETV represents a highly effective antiviral for primary treatmen t of chronic HBV infection.
AUTHOR CONCLUSIONS
No ETVR evident in nucleoside naïve subjects treated with ETV
Superior degree of viral suppression by ETV in nucleoside naïve subjects likely accounts for a lack of resistance emergence
Confirmed virologic rebound on ETV due to resistance was observed in 1% of LVD-refractory subjects by Week 48
High level resistance to ETV requires substitutions at residues rtT184, rtS202 and/or rtM250 (ETV R substitutions) in a background of pre-existing LVDR changes (rtL180 and/or rtM204)
ETVR substitutions emerged in 11 LVD-refractory subjects with HBV containing LVDR substitutions
ETVR substitutions were present at baseline in LVD-refractory subjects, suggesting emergence on prior LVD therapy
ETV did not select for LVDR substitutions or other novel substitutions that result in reduced susceptibility to ETV
Other factors beyond pre-existing ETVR substitutions appear to be needed to lead to a rapid development of ETV R and viral rebound during ETV therapy
|
|
| |
| |
 |
|
| |
| |
|
|
| |
| |
 |
|
| |
| |
|
|
| |
| |
 |
|
| |
| |
|
|
| |
| |
 |
|
| |
| |
|
|
| |
| |
 |
|
| |
| |
|
|
| |
| |
 |
|
| |
| |
|
|
| |
| |
 |
|
| |
| |
|
|
| |
| |
 |
|
| |
| |
|
|
| |
| |
 |
|
| |
| |
|
| |
|
 |
 |
|
|