 |
 |
 |
| |
UPDATE on SCHERING'S PLOUGH'S ENTRY INHIBITOR (CCR5 Inhibitor) IN PHASE II CLINICAL DEVELOPMENT
|
| |
| |
From Jules Levin. I just arrived at the Tokyo Airport after a 10 hour flight on my way to Bangkok. After a 3 hour layover here & 6 hour flight to Bangkok this will make a total trip of 19 hours from SF, not including trip to SF. It's a big trip. Surprisingly, so far I feel ok. This is because since finishing 18 months peginterferon/ribavirin 2 years ago with a sustained viral response I feel much better—more energy and mental clarity. HCV definitely caused me low energy & cognitive impairment and this has increasingly improved over the past two years since I finished therapy, the improvement did not plateau. Saw two good movies on the 10 hour flight, the meals in economy were not good but the service on flight to Tokyo is better than on US flights. The United Club in Tokyo airport is great with US power connections for computer & great AOL connections. Its like being in the Big Apple for me. There are lots of US advocates and researchers here all heading to Bangkok. ACTG and academic researchers and US & NIH folks are here also. I hear it will be very hot (>100 degrees & humid) in Bangkok and conference center is 1-2 hours from hotels. Hopefully there will be some interesting new data to make the trip fruitful. Meanwhile I have time & I'm sending reports to you. Jules
Schering-Plough presented Phase I clinical data on SCH-D at the Conference on Retroviruses and Opportunistic Infections (CROI) in San Francisco in February. Below is background review on this Phase I data. Following is an outline antiviral effects seen in studies for SCH-D, phase II study plans, and commitment to Expanded Access Program.
SCH-D, which we now refer to as SCH-417690 or "690", has progressed to Phase II clinical development in two separate trials: a U.S. study to be conducted by the ACTG in treatment-experienced patients and an international study (Europe and Canada) to be conducted by Schering-Plough Research Institute (SPRI) in treatment-naive patients. The respective study protocols are currently undergoing local IRB (institutional review board) review by the individual study centers.
THE FOLLOWING IS A PAPER RELEASED BY SCHERING-PLOUGH and forwarded to you for your information.
SCHERING-PLOUGH CCR5 RECEPTOR ANTAGONISTS: A NOVEL THERAPEUTIC APPROACH IN THE TREATMENT OF HIV INFECTION
Recent progress in the field of antiretroviral therapy has been dramatic. However, the prospect of drug resistance to antiviral agents, the toxicity of current therapies, the challenge of patient compliance to complex regimens and the uncertainty of the long-term outcome of current treatments support the development of new agents to treat HIV infection.
Background
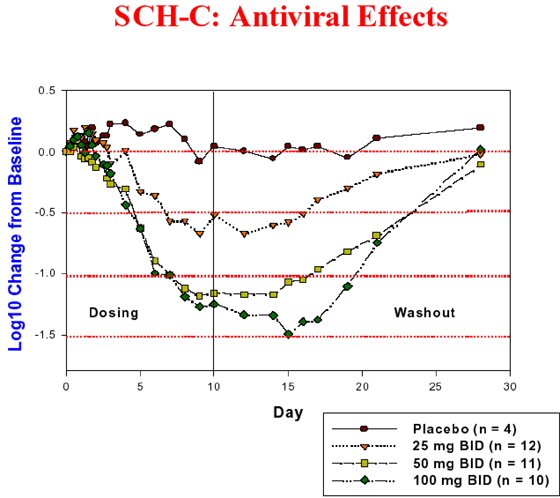
Schering-Plough Research Institute (SPRI) is developing orally active CCR5 receptor antagonists, offering a novel therapeutic approach to treating HIV infection. Two clinical candidates, SCH-C and SCH-D, have emerged from this program. SCH-C and SCH-D efficiently block cell entry of a wide range of primary HIV-1 isolates that use the CCR5 receptor for infection (R5 viruses). These compounds do not inhibit HIV isolates that gain entry into cells via the CXCR4 receptor (X4 viruses). SPRI has reported Phase I results for SCH-C and SCH-D in HIV- infected patients. SCH-D has shown greater in vitro potency than SCH-C and has favorable pharmacokinetic and metabolism profiles in several animal species. Phase I clinical results with SCH-D continue to support the CCR5 receptor as a viable target for antiretroviral therapy. Based on these Phase I results, SPRI expects to advance SCH-D to Phase II clinical studies in the first half of 2004.
Background on CCR5 Receptor Antagonists
Chemokine receptors expressed on the surface of immune cells are known to play a critical role in virus infection and transmission. One such chemokine receptor, CCR5, acts in tandem with another receptor (CD4) to facilitate entry of HIV into cells. Targeting CCR5 as anti-HIV therapy was initially suggested by a finding that individuals who lack a functional CCR5 receptor (i.e., those who have a 32 bp CCR5 mutation) are largely resistant to HIV infection. Preclinical studies showing that natural ligands and antibodies to CCR5 can inhibit HIV-1 infection provided proof-of-principle validation of the development of CCR5 antagonists as novel antiviral agents.
CCR5 receptor antagonists belong to a new, investigational class of antiretrovirals known as HIV entry inhibitors. This new class includes various experimental compounds designed to block cell surface receptors, such as CCR5 or CXCR4, as well as other novel compounds that block HIV fusion with the cell surface. Unlike existing HIV drugs that work inside the cell and target viral enzymes involved in the replication of the virus, entry inhibitors work by blocking HIV before the virus enters the cell and begins its replication process.
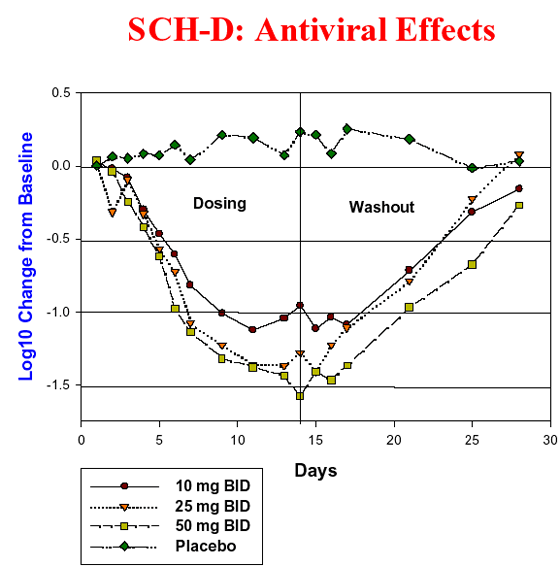
SCH-D Phase-I Study and Results
Forty eight patients chronically infected with HIV were enrolled into a sequential rising-dose study of SCH-D 10 mg, 25 mg and 50 mg BID for 14 days. Within each of the three cohorts (N = 16), 12 patients received SCH-D and four patients received placebo in a randomized, blinded design.
Patients were required to have been on no antiretroviral treatment for at least eight weeks prior to enrollment and to have a CD4+ cell count greater than 200/mm3. Overall results showed that SCH-D was well tolerated at all dose levels. The study did not identify any drug-specific toxicity in humans. The study showed a dose-related decrease in viral load, with mean Log10 reductions from baseline of 1.08, -1.56 and -1.62 in the 10, 25 and 50 mg BID dose groups, respectively. The percentage of patients with a >1.0 Log10 reduction in viral load was 55%, 69% and 81% in the 10, 25 and 50 mg BID dose groups, respectively. The percentage of patients with a >1.5 Log10 reduction in viral load was 27%, 46% and 45, respectively.
|
|
| |
| |
 |
|
| |
| |
|
|
| |
| |
 |
|
| |
| |
SCH-417690
(Formerly SCH-D)
CLINICAL DEVELOPMENT
Phase II Program Overview
Drug Characteristics:
• Small molecule inhibitor of CCR5 receptor
-- Prevents entry of HIV into T-cells
• Orally bioavailable (~ 80%)
• Long T1/2 means once daily dosing (Q Day)
• Exposure boosted by ritonavir
• Anti-HIV activity with good safety margin based on blood concentrations
-- 1.62 log10 suppression of HIV RNA over 14 days (Phase I results)
Program Overview:
• Multicenter, multinational program
• Two pivotal, complementary studies
-- Treatment-experienced subjects -- U.S.
-- Treatment-naïve subjects -- Europe & Canada
• Supplemental studies in HCV co- infection and salvage population
• Prepare for seamless transition to Phase III
PHASE II TRIAL OF SCH-417690 IN TREATMENT-EXPERIENCED SUBJECTS
Adult ACTG 5211:
• Randomized, placebo-controlled "add-on" design
-- 3 dose levels of 690 vs. placebo plus continued 3-drug HAART regimen for 14 days
-- 46 additional weeks with new "optimized" HAART regimen
• New HAART regimen based on HIV resistance pattern
• Must contain ritonavir = 100 mg/day
• Doses of 690: 5, 10, 15 mg Q Day
-- Chosen to achieve range of exposure known to exhibit antiviral effect
Patient Population:
• Male and female adults = 18 years
• On stable ritonavir-containing HAART = 8 weeks; must have failed at least one prior 3-drug
regimen
• CD4 = 50; HIV RNA = 5000
• Only CCR5 HIV detected on phenotypic screen
• No prior CNS pathology
• No HCV or HBV co-infection
Endpoints:
• Change from baseline HIV RNA
• % subjects with HIV suppression by 1 log10, < 400, <50 copies
• Emergence of resistance to 690
• Change in HIV "tropism" from CCR5 to CXCR4
• Safety and tolerability
• Change in CD4 counts
• Occurrence of AIDS-defining events
Logistic Considerations:
• Sample size: 120
-- Randomized 1:1:1:1
• Study to be conducted at all AACTG sites
-- Satellite sites eligible to participate
• Study information available on www.ClinicalTrials.gov
• Powered to select Phase III dose
PHASE II TRIAL OF SCH-417690 IN TREATMENT-NAIVE SUBJECTS
P03802 -- Sponsored by Schering-Plough Research Institute (SPRI)
• Randomized, placebo-controlled
-- 3 dose levels of 690 vs. placebo for 14 days
-- 46 additional weeks in combination with Combivir®
• Placebo-recipients also receive efavirenz with Combivir®
• Doses of 690: 25, 50, 75 mg Q Day
-- Chosen to achieve range of exposure known to exhibit antiviral effect
Patient Population:
• Male and female adults = 18 years
• No prior antiretroviral therapy
• CD4 = 50; HIV RNA = 5000
• Only CCR5 HIV detected on phenotypic screen
• No prior CNS pathology
• No HCV or HBV co-infection
Endpoints:
• Change from baseline HIV RNA
• % subjects with HIV suppression by 1 log10, < 400, <50 copies
• Emergence of resistance to 690
• Change in HIV "tropism" from CCR5 to CXCR4
• Safety and tolerability
Logistic Considerations:
• Sample size: 80
-- Randomized 1:1:1:1
• Study to be conducted at 20 sites in EU and Canada
• Primary analysis powered to select Phase III dose
Schering-Plough's Commitments:
• Subjects who respond to 690 in Phase II may receive it until commercialization
• Placebo recipients in P03802 may receive 690 at the selected dose after study completion
• An Expanded Access program for patient s with no other options will be initiated, where permitted, during Phase III
|
| |
|
 |
 |
|
|