 |
 |
 |
| |
Adefovir 3 Year Study Results in Presumed Precore Mutant Chronic Hepatitis B: safety and efficacy
|
| |
| |
Adefovir 3 Year Study Results in Presumed Precore Mutant Chronic Hepatitis B: safety and efficacy
"Three Year Results from a Double-Blind, Randomized, Placebo-Controlled, Study of Adefovir for Presumed Precore Mutant Chronic Hepatitis B"
Reported by Jules Levin
S Hadziyannis (Henry Dunant Hospital, Athens, Greece) reported results from this study at EASL Conference (April 14-18, 2004, Berlin, Germany). Here is his report.
Adefovir is a chain terminator of HBV DNA. It has activity against wild-type and lamivudine-resistant (LAM-R) HBV. The dose is one 10 mg tablet, once daily orally (dose interval modified for creatinine <50 ml/min).
The purpose of this study is to evaluate safety and efficacy of ADV over 96 and 144 weeks.
Brief summary: 3 years of ADV resulted in significant and sustained reductions in HBV DNA and ALT. Increasing proportions of patients achieved undetectable HBV DNA & ALT. Safety profile over 144 weeks consistent with first 48 weeks. Resistance to adefovir isdelayed & infrequent: In this study, 5.9% experienced ADV resistance after 3 yrs on ADV; across ADV studies over 144 weeks 3.9% had ADV resistance.
KEY INCLUSION CRITERIA
HBeAg-, HbeAb+, and HBsAg+
Compensated liver disease
HBV DNA >=105 copies/ml
ALT x ULN >= 1.5-15
Adequate renal function
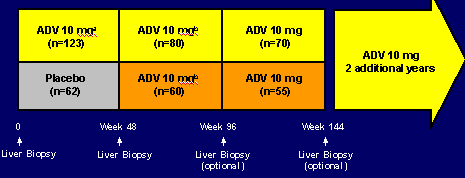
STUDY 438 (0-144 weeks)
STUDY DESIGN
185 patients randomized to ADV or placebo. After 48 weeks patients receiving placebo were offered ADV for 2 additional years. After 3 years of study patients are offered 2 additional years for a total of 5 years in this study/ Liver biopsies are at week 48, 96 (optional), and week 144 (optional).
|
|
| |
| |
 |
|
| |
| |
BASELINE CHARACTERISTICS
|
|
Weeks 0-144
N=70
|
|
Median age
|
47 yrs
|
|
Male
|
81%
|
|
Asian
|
26%
|
|
Caucasian
|
70%
|
|
Black
|
4%
|
|
Prior IFN
|
37%
|
|
Prior LAM*
|
7%
|
*less than 12 weeks exposure
BASELINE HBV DISEASE CHARACTERISTICS
| Weeks 0-144
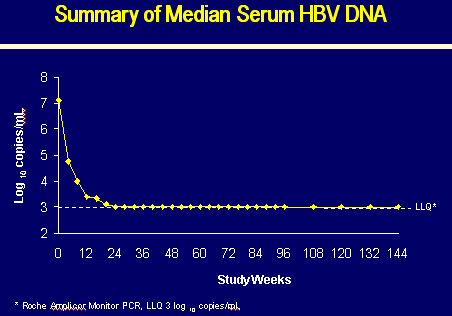
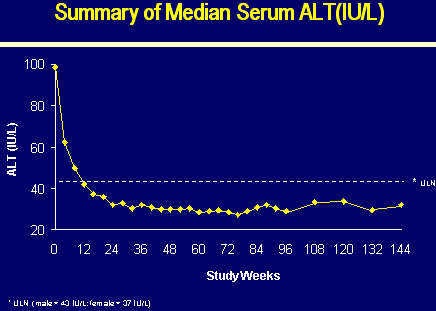
N=70 | | Median HBV DNA (log10 copies/ml*) | 7.08 | | Median ALT (x ULN) | 2.3 | | Median ALT (IU/L) | 99 | | Cirrhosis | 11% |
*Roche Amplicor Monitor PCR Assay, LLQ 3 log10 copies/ml
|
|
| |
| |
 |
|
| |
| |
|
|
| |
| |
 |
|
| |
| |
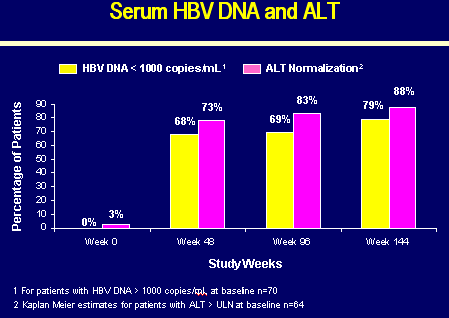
SERUM HBV DNA and ALT
Week 0:
0% HBV DNA <1000 copies/ml
3% ALT Normalization
Week 48:
68% HBV DNA <1000 copies/ml
73% ALT normalization
Week 96:
69% HBV DNA <1000 copies/ml
83% ALT normalization
Week 144:
79% HBV DNA <1000 copies/ml
88% ALT normalization
Patients with HBV DNA >1000 copies/ml at baseline, n=70
Kaplan Meier estimates for patients with ALT at baseline, n=64
|
|
| |
| |
 |
|
| |
| |
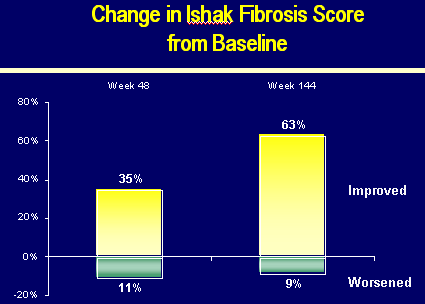
CHANGE IN ISHAK FIBROSIS SCORE from BASELINE
Week 48:
35% of patients improved
11% worsened
Week 144:
63% of patients improved
9% worsened
Improvement defined as >=1 point reduction
Worsened defined as >=1 point increase
Median Ishak Fibrosis at baseline=2
|
|
| |
| |
 |
|
| |
| |
CLINICAL ADVERSE EVENTS
| Week 0-96
N=79 | Week 0-144
n=70 | | % of patients w/any events | 85% | 86% | | % w/AE possibly/probably related to study drug | 47% | 49% | | Serious AE | 6% | 10% | | SAE possibly/probably related to study drug | 0% | 0% |
RENAL LAB PARAMETERS
| Wks 0-96
N=79 | Wks 0-144
n=70 | | Serum Creatinine | | | | increase from baseline >=0.5 mg/dL* | 2 (2.5%) | 3 (2.4%) | | median change from baseline (mg/dL) | 0.1 | 0 | | Serum phosphorous | | | | <1.5 mg/dL* | 0 | 0 | | median change from baseline (mg/dL) | -0.2 | -0.2 |
*confirmed (two consecutive lab abnormalities)
RESISTANCE EVALUATION
ADV resistance mutation N236T:
0 patients had mutation in 1st year (n=123), 2 in 2nd year (n=134*), and 1 in third year (n=70).
ADV-resistance mutation A181V:
0 patients got mutation in 1st yr, 2 in 2nd yr, and 1 in 3rd yr.
Reduced susceptibility to adefovir in vitro:
4-14 fold in patients with N236T;
2.5 to 3-fold in patients with A181V.
All samples with detectable serum HBV DNA by PCR (>1000 copies/ml) at weeks 48, 96, and 144 were analyzed.
The overall incidence of adefovir resistance in study 438 at weeks 48, 96, and 144 was 0%, 3.0%, and 5.9%, respectively.
*includes 55 and 144 samples from patients who received placebo during the first year.
Integrated Resistance Surveillance Across ADV Studies (0-144 weeks)
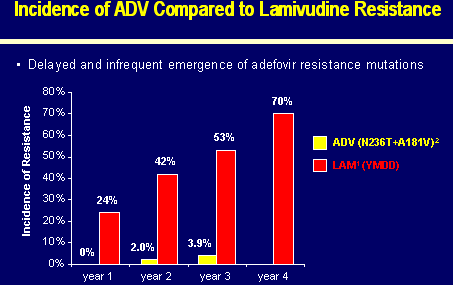
INCIDENCE OF ADV COMPARED TO LAMIVUDINE RESISTANCE
Year 1: 0% ADV; 24% lamivudine
Year 2: 2.0% ADV; 42% lamivudine
Year 3: 3.9% ADV; 53% lamivudine
Year 4: …ADV; 70% lamivudine
ADV (N236T+A181V)
LAM (YMDD)
|
|
| |
| |
 |
|
| |
| |
|
| |
|
 |
 |
|
|