| |
Management of hepatitis B patients with antiviral resistance
|
| |
| |
Antiviral Therapy 9:1013--1026
Scott K Fung and Anna SF Lok*
Division of Gastroenterology, University of Michigan, Ann Arbor, Mich., USA
Drug resistance is an expected consequence of antiviral therapy for chronic hepatitis B because of the high rate of hepatitis B virus (HBV) replication, the lack of proofreading during reverse transcription of the pregenomic RNA and the low efficacy of available therapies in eliminating covalently closed circular HBV DNA. Mutations involving the YMDD motif of the catalytic domain of HBV reverse transcriptase have been reported in patients wh have received lamivudine, emtricitabine and telbivudine. Drug-resistant mutations affecting other regions of HBV polymerase have also been reported, but at much lower rates in patients who have received adefovir dipivoxil or entecavir. Antiviral resistance is initially manifested as virological breakthrough infection. In most patients, this is followed by biochemical breakthrough and, in some patients, hepatitis flares and hepatic decompensation. Monitoring drug resistance may improve the management of patients with antiviral-resistant HBV and can guide the selection of salvage therapy. The optimal management of patients with antiviral-resistant HBV continues to evolve. The ideal approach is to prevent antiviral resistance through judicious use of antiviral therapy and the use of more potent antiviral agents, possibly in combination.
The NATAP Hepatitis B section where you can numerous articles on HBV therapy including treatment management discussions including:
http://www.natap/hbv.htm
Review article: current management of chronic hepatitis B
http://www.natap.org/2004/HBV/102504_02.htm
HBV/HIV Coinfection Medical Management
http://www.natap.org/2004/HBV/092004_05.htm
INTRODUCTION
The development of oral antiviral agents that are effective in suppressing hepatitis B virus (HBV) replication has been a major advance in the treatment of chronic hepatitis B. Lamivudine was the first oral agent to be approved for the treatment of chronic hepatitis B. However, it soon became apparent that a major disadvantage was the high rate of drug resistance, reaching 70% after 5 years of therapy [1--3]. Adefovir dipivoxil, the second oral antiviral agent approved for the treatment of chronic hepatitis B, is associated with a much lower rate of drug resistance, approximately 6% after 3 years of therapy [4]. It is also effective in suppressing lamivudine-resistant HBV [5,6]. However, adefovir dipivoxil has the potential for nephrotoxicity, particularly with high dose or long-term use [6,7] (note from Jules Levin: to my knowledge at the standard adefovir HBV dose 10mg per day there have not been confirmation so far since adefovir has been on the market that nephrotoxicity has developed in patients that was not associated with underlying causes such as a predisposition towards kidney abnormalities. However, further follow-up will be useful). Drug resistance has also been reported in association with other new antiviral agents that are being evaluated in clinical trials. The optimal management of patients with antiviral-resistant hepatitis B is unclear. Unlike HIV infection, there is a limited choice of antiviral agents, lack of an optimal combination therapy and paucity of data on the incidence and clinical consequences of antiviral-resistant hepatitis B beyond the first year. In this article, we will review current knowledge on the incidence, risk factors and clinical consequences of antiviral-resistant hepatitis B and propose how it can be prevented and managed.
(note from Jules Levin: This article does not include discussion of pegylated interferon for treatment in HBV. Roche has submitted results from phase III study to the FDA in its application for approval of Pegasys for treatment of HBV. The data from this phase III shows usefulness of Pegasys in treatment HBV. Some clinicians prefer using peginterferon first in treating HBV before using an oral antiviral while other clinicians prefer use of oral drugs while other clinicians may use both types of treatment together. As mentioned in this article management for HVC treatment has yet to be well defined. In addition, this article was written before the AASLD liver conference in November 2004, where phase III study results were reported for entecavir. So although the authors discuss entecavir they do not report the phase III study results. Numerous entecavir studies were reported at AASLD and on the NATAP website you can find entecavir study results as well as a full report of coverage from the conference in the HBV section and in the Conference Resports section).
Biology of HBV
HBV is extremely prolific and its replication is errorprone resulting in a high rate of spontaneous mutations. The existence of an intracellular recycling pathway and the relative resistance of the covalently closed circular HBV DNA (cccDNA) to antiviral therapies make it difficult, if not impossible, to eradicate HBV. Thus, resistance to antiviral therapy of hepatitis B is to be expected.
High mutation rate during HBV replication
The replication cycle of HBV is depicted in Figure 1. The HBV genome is made of partly double-stranded DNA that replicates via reverse transcription of an RNA intermediate (pregenomic RNA). Because of the lack of proof-reading by reverse transcriptase, the spontaneous mutation rate during HBV replication is approximately 1.5×10--5--3.0×10--5 nucleotide substitutions per site per year [8]. Since HBV is extremely prolific, producing up to 1010--1012 virions on a daily basis, a large number of spontaneous mutations could potentially be generated at every site in the HBV genome [9,10]. Although the mutation rate of HBV is higher than that of other DNA viruses, the overlapping arrangement of open reading frames in the HBV genome limits the viability of spontaneous mutants. By comparison, the mutation rate of HIV is 100- to 1000- fold higher [11].
Persistence of cccDNA
After entry into a hepatocyte, the HBV genome repairs itself and resides in the nucleus as a mini-chromosome, the covalently closed circular DNA (cccDNA). The cccDNA serves as a transcription template for viral proteins and for the pregenomic RNA [12]. Treatment with currently available antiviral agents has very little effect on cccDNA. Thus, elimination of the pool of cccDNA ultimately depends on hepatocyte turnover [13]. This explains why viral eradication is difficult to achieve and accounts for the high relapse rate when treatment is stopped.
|
|
| |
| |
 |
|
| |
| |
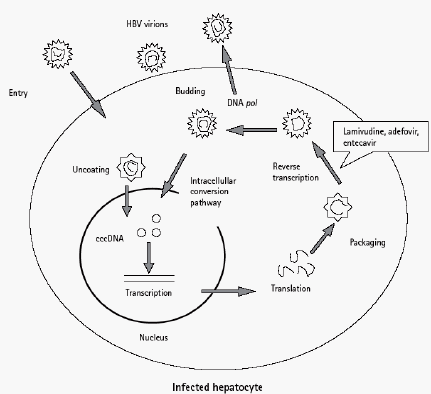
After entering a hepatocyte, the HBV genome repairs itself and resides in the nucleus as a mini-chromosome, the covalently closed circular DNA (cccDNA). cccDNA acts as a transcription template for viral proteins and for the pregenomic RNA. Nucleos(t)ide agents inhibit viral replication at the level of reverse transcription. DNA pol, DNA polymerase.
Mechanisms of antiviral-resistant HBV
Approved antiviral agents and antiviral agents in clinical trials target the HBV polymerase. Drug-resistant mutations have been reported with all nucleos(t)ide agents used for the treatment of chronic hepatitis B. However, the mechanisms by which the mutations confer resistance and the rates at which they emerge differ.
Mutations in the polymerase gene
The HBV polymerase gene is comprised of four different regions: primer, spacer, reverse transcriptase/ DNA polymerase and RNAse H [14]. The reverse transcriptase/DNA polymerase region is responsible for reverse transcription of the pregenomic RNA into the first (negative) strand of HBV DNA and synthesis of the second (positive) strand of HBV DNA. This region has a high degree of homology to other reverse transcriptases, including that of HIV [14].
It has been proposed that drug-resistant mutations may predate the start of antiviral treatment and become selected as the dominant viral species under antiviral pressure [15]. Given the large pool of HBV DNA and the myriad of spontaneous mutations, it would be impossible even with the most sensitive technology currently available to screen patients for drug-resistant mutations prior to treatment. Nevertheless, several investigators, including one group from Japan, have reported the detection of lamivudine resistant HBV in 5/18 (28%) treatment-naive patients [16]. These findings are potentially important but need to be validated.
Lamivudine resistance
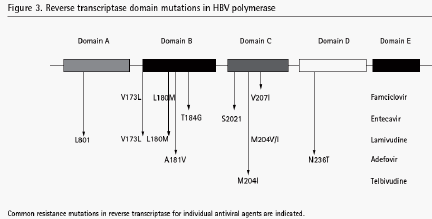
Lamivudine-resistant HBV mutations have been studied extensively because lamivudine has been available for a longer period of time than other antivirals and the rate of lamivudine-resistant mutation is relatively high. Long-term follow-up data from Phase III clinical trials have shown that the rate of drug resistance progressively increases with the duration of treatment from 15--30% after 1 year, to 40--50% after 3 years and to 70% after 5 years of treatment [1--3]. The most important mutations involve the YMDD (tyrosine-methionine-aspartate-aspartate) motif, a catalytic region in the C domain of the reverse transcriptase in the HBV polymerase, in which methionine is substituted by valine, isoleucine or, very occasionally, serine (rtM204V/I/S) (Figure 3) [17]. The analogous mutation in HIV is rtM184V, which appears within weeks of exposure to lamivudine monotherapy [18]. Steric hindrance caused by amino acid substitution at the YMDD motif prevents the binding of lamivudine in the dNTP pocket and incorporation into viral DNA [19]. In vitro data indicate that IC50 increases by as much as 1000 to 10,000-fold [20]. Thus, an escalation in lamivudine dose cannot overcome the resistance conferred by these mutations. Initial studies found that rtM204V was more common than rtM204I, but recent data suggest that the selection of different mutations may be genotype-dependent: rtM204I is more commonly associated with HBV genotype D while rtM204V is more frequently found in association with genotype A [21]. In patients with virological breakthrough during lamivudine therapy, rtM204I has been found in isolation, while rtM204V is invariably associated with an upstream mutation rtL180M, a leucine to methionine substitution in the B domain of HBV reverse transcriptase [22].
In vitro studies have shown that rtM204V/I decreases the replication fitness of lamivudine-resistant HBV [23--25]. This explains why patients with lamivudine resistance initially have low HBV DNA and ALT levels. Once lamivudine therapy is withdrawn, the re-emergence of wild-type virus occurs within a matter of weeks to months, since drug resistant mutants do not confer any survival advantage in the absence of the drug [23]. However, lamivudine resistant mutants may remain detectable for many months after stopping treatment and may be rapidly selected upon re-exposure to the drug [26]. Additional mutations may be selected during continued treatment. The most common mutations are V173L, L80I and L180M; L180M and V173L have been shown to partially restore viral replication fitness in in vitro studies [27]. Case reports suggest that V173L may be associated with aggressive liver disease but our experience with non-transplant patients who have not been exposed to multiple antiviral agents did not substantiate this finding [28]. Lamivudine-resistant P gene mutations can lead to amino acid substitutions in the overlapping S gene and may reduce the protective efficacy of HBV vaccine or hepatitis B immune globulin [29]. Mutations at the YMDD motif, rtM204V and rtM204I result in substitutions in codon 195 and 196 of the surface protein (sI195M and sW196S), while the corresponding change for rtL180M is silent in the S protein [30].
|
|
| |
| |
 |
|
| |
| |
Emtricitabine resistance
Emtricitabine (FTC) is a nucleoside analogue of dCTP and is structurally similar to lamivudine. The same drug-resistant mutations (rtM204V/I +/- L180M) were found in 9--16% patients after 1 year and in 19--37% patients after 2 years of treatment with emtricitabine 200 mg [31]. The high rate of drug resistance and cross-resistance with lamivudine will limit the role of emtricitabine in the treatment of chronic hepatitis B.
Telbivudine resistance
Telbivudine (LdT) belongs to a new class of agents, the β-L-nucleosides, which are active against HBV but not HIV. In vitro studies showed that it is active against rtM204V. Data from a Phase IIb trial, in which 104 patients were randomized to receive LdT, lamivudine or a combination of the two drugs, showed that rtM204I mutants were detected in 5% of LdT-treated patients compared with 10% of patients in the combination group at the end of year 1 [32]. These data confirmed that LdT selects for mutation in the YMDD motif but so far only rtM204I has been detected, and at a lower rate compared with lamivudine, possibly because of its greater potency (6 log10 vs 4.5 log10 decrease in serum HBV DNA). However, the combination of LdT and lamivudine was associated with a higher rate of antiviral resistance and a lower rate of hepatitis B e antigen (HBeAg) seroconversion compared with LdT alone, indicating that the combination may have antagonistic effects.
Famciclovir resistance
Famciclovir, a nucleoside analogue of dGTP, is less potent than lamivudine in suppressing HBV replication. Resistance to famciclovir has been reported during long-term administration, the most common mutation being L180M with or without V173L and V207I [33,34]. Development of famciclovir as an antiviral agent for chronic hepatitis B has been abandoned because of the need for three-times daily dosing, weak antiviral effects and cross-resistance with lamivudine.
Adefovir dipivoxil resistance
Resistance to adefovir dipivoxil is less frequent and occurs later than with lamivudine [35,36]. Various mechanisms have been postulated to explain the low rate of resistance to adefovir [35]. The adefovir molecule is structurally very similar to dATP, which limits steric discrimination by HBV polymerase. The phosphonate bond in adefovir may be less susceptible to chain terminator removal once the molecule is incorporated into viral DNA. Also, its flexible molecular structure allows binding to HBV polymerase, despite minor changes in the dNTP pocket. The adefovir resistant HIV mutation, K70E (lysine to glutamate change), also occurs at a very low rate (<1% after 1 year of treatment). The equivalent of this site in HBV is at codon 38, and is usually represented by T, A, E or S but mutations to a K have been reported [37].
Adefovir-resistant HBV mutation was not detected after 1 year of treatment despite extensive testing of 498 patients [35]. However, adefovir-resistant mutations have been reported in patients who received longer durations of therapy. These mutations are different from those that confer resistance to lamivudine. The most important mutation is a substitution of asparagine for threonine, rtN236T, in the D domain of HBV polymerase (Figure 3) [38]. This mutation decreases sensitivity to adefovir by four- to 13-fold and it has been suggested that rtN236T favours binding to its natural substrate (dATP) rather than to adefovir [39]. A second mutation, an alanine to valine substitution, rtA181V, decreases sensitivity to adefovir by two to threefold. In vitro studies show that adefovir resistant mutants remain sensitive to lamivudine, emtricitabine, telbivudine and entecavir [39].
Serial testing of 167 patients who received adefovir for 144 weeks found that rtN236T was detected in six (3.6%) patients and rtA181V in three (1.8%) patients [4]. Some of the patients in this series had prior resistance to lamivudine and were receiving a combination of lamivudine plus adefovir, which may have reduced the rate of adefovir resistance. In a subset of 67 treatmen tnaive patients with HBeAg-negative chronic hepatitis B, adefovir-resistant mutations were detected in four (6%) patients after 3 years of adefovir monotherapy [4]. Further studies are needed to determine the exact incidence of adefovir resistance as well as the risk factors and clinical consequences.
Entecavir resistance
Entecavir is a deoxyguanosine analogue that selectively inhibits HBV polymerase as well as the priming step of HBV replication. Phase II clinical trials showed that entecavir is more potent than lamivudine in suppressing HBV replication [40]. Results of Phase III clinical trials have not been released. To date, there has been no report of entecavir resistance among treatment-naïve patients. Resistance to entecavir was recently reported in two patients with lamivudine resistance who had received entecavir in combination with lamivudine for 80--100 weeks [8]. Several novel mutations were detected: rtI169T, rtM250V, rtT184G and rtS202I; the latter three mutations have been confirmed to confer resistance to entecavir in in vitro studies, but only in the presence of lamivudine-resistant mutations. Entecavirresistant mutants are sensitive to adefovir dipivoxil but resistant to lamivudine. Our laboratory recently identified another HBV polymerase mutation, rtT184L, in a patient who developed breakthrough infection after more than 2 years of treatment with entecavir and lamivudine for lamivudine-resistant HBV.
Continued surveillance is necessary to determine if entecavir-resistant HBV mutants will be detected in patients with no prior antiviral therapy. It is likely that over time more patients with entecavir resistance will be diagnosed. Further studies are needed to determine the rate of entecavir resistance in patients who have prior exposure to lamivudine and other antiviral agents used for treating hepatitis B, and to unravel the mechanisms by which lamivudine-resistant mutations increase the risk of entecavir resistance.
Resistance to combination therapy
One advantage of combination therapy is the potential to reduce the risk of drug resistance compared with monotherapy. Several studies have reported that patients who received combination therapy of interferon and lamivudine had lower rates of lamivudine resistance compared with those who received lamivudine only [41--43]. However, in most studies the duration of lamivudine treatment was shorter in the combination therapy group: 16--24 weeks versus 52 weeks in the lamivudine monotherapy group. Thus, additional studies in which the duration of lamivudine treatment is identical in the groups that receive lamivudine alone or in combination with interferon are needed to determine if the addition of interferon reduces the risk of lamivudine resistance.
In an ongoing study of 115 treatment-naive patients who received lamivudine alone or in combination with adefovir, lamivudine-resistant mutation was detected in 20% of patients in the monotherapy group compared with 2% in the combination therapy group after 1 year of treatment [44]. These data indicate that the combination of two nucleoside or nucleotide analogues with activity against HBV can reduce the rate of drug resistance. However, viral suppression and HBeAg seroconversion rates were similar in the two groups. Follow-up data in year 2 will determine if the lower rate of resistance in the combination group will translate into a higher rate of maintained virological response. A key question that will not be addressed in this study is whether the combination of lamivudine and adefovir as de novo therapy will reduce the risk of antiviral resistance and increase the rate of HBeAg seroconversion compared with treatment with adefovir alone.
The potential benefits of combination therapy were not demonstrated in another study where the rate of drug-resistant mutations was higher in the group that received combination therapy of telbivudine plus lamivudine compared with that which received telbivudine only, 10% versus 5%, respectively [32]. In this study, the rate of HBeAg loss was also lower in the combination therapy group (17%), compared with the telbivudine monotherapy group (33%). These findings indicate that caution needs to be exercised in selecting appropriate agents for use in combination therapy. In general, drugs that select for the same resistant mutations should not be used together.
(note from Jules Levin: at the AASLD liver meeting in November 2004 Lau et al reported results from a study of adefovir plis FTC combination therapy versus adefovir alone in HBeAg+ chronic hepatitis B. Lau found combination therapy demonstrated greater antiviral activity using ADV+FTC than monotherapy using ADV (-5.44 log vs -3.40 log using Roche PCR, with lower limit of detection of 200 copies/ml). You can read detailed results from this study on the NATAP website in the HBV section and in the Conference Reports section).
Diagnosis of antiviral resistance
Clinically, antiviral resistance is suspected when biochemical or virological breakthrough occurs in medication-compliant patients. Monitoring for genotypic resistance may detect the development of resistance earlier and can guide the management of patients with antiviral resistance.
Breakthrough infection
Biochemical breakthrough is usually defined as alanine aminotransferase (ALT) levels >1.5 times the upper limit of normal (ULN) during continued treatment in patients who initially had normalization of ALT [45,46]. Virological breakthrough is usually defined as >1 log10 copies/ml increase in serum HBV DNA levels during continued treatment in patients who initially had adequate viral suppression [45,46]. Virological breakthrough is usually followed by biochemical breakthrough but the time lapse may vary from weeks to years [47]. Not all breakthrough infection is due to drug resistance. Among patients who were closely followed in clinical trials, only two out of three of patients with virological breakthrough had detectable drug-resistant mutations [48,49]. Thus, compliance should be checked in all patients with breakthrough infection and when possible, confirmatory testing should be performed. Testing for genotypic resistance will be increasingly important as more patients are exposed to multiple antiviral agents and additional treatment options become available. Screening for drug resistance prior to initiation of treatment and regular surveillance during treatment is an integral part of management of patients with HIV infection [50].
Confirmation of antiviral resistance
For new antiviral agents, initial surveillance for drug resistant mutations requires direct sequencing of the HBV polymerase gene. This method is labour-intensive and requires serial samples from the same patient or large cohorts of patients to determine if changes from a prototype sequence are associated with breakthrough infection. Novel mutations associated with breakthrough infection are replicated in a prototype strain by site-directed mutagenesis to confirm that the mutation does confer resistance. Phenotypic assays are then performed to determine the degree of resistance to the drug of interest and the activity of other antiviral agents against the mutant.
Unlike HIV treatment, testing for drug-resistant HBV mutants is not routinely available. However, several high-throughput assays have been developed to test for known antiviral resistance mutations. These tests include restriction fragment length polymorphism (RFLP), reverse hybridization/line probe assays (InnoLiPA; Innogenetics, Ghent, Belgium) and realtime PCR, as well as in situ sequencing (Trugene; Visible Genetics Inc., Toronto, Ont., Canada). The advantages of these tests include rapid turnaround time and ease of use. Some of these tests, such as the line probe assay, are more sensitive in detecting minor viral species and are able to detect drug-resistant mutants earlier than direct sequencing [26,51,52]. They also demonstrate that drug-resistant mutants remain persistently detectable for many months after drug withdrawal. Monitoring for antiviral resistance should be considered in patients who may decompensate during breakthrough infection such as patients with underlying cirrhosis, transplant recipients and other immunosuppressed patients.
Clinical consequences of antiviral resistance
Compared with pre-treatment values, most patients with lamivudine resistance initially have low HBV DNA and ALT levels, but over time these values may increase [1]. The potential adverse consequences of lamivudine resistance are listed in Table 1, but the rate at which they occur is uncertain as most studies involved case reports or small case series. Little is known about the clinical consequences of resistance to other antiviral agents for hepatitis B, but similar scenarios are likely to be true.
Table 1. Clinical consequences of drug-resistant HBV
Flares in ALT
Reduced HBeAg seroconversion
Histological progression of disease
Increased recurrence post-transplant
Changes in HBsAg antigenicity
Transmission of drug-resistant HBV
Decreased rate of HBeAg seroconversion
The rate of HBeAg seroconversion is decreased but not abolished in patients with lamivudine resistance. In a study of 55 patients who received lamivudine for 2 years, HBeAg seroconversion was observed in 8/27 (26%) patients with lamivudine resistance and in 12/23 (52%) patients without lamivudine resistance [53]. HBeAg seroconversion occurred more often among patients with lamivudine resistance who had ALT flares compared with those who did not have ALT flares, 8/12 (75%) versus 0/19 (0%), respectively, suggesting that this may be a result of immune clearance.
Deterioration in liver histology
Improvement in liver histology is maintained in most patients who have had lamivudine resistance for 1--2 years, but over time this benefit may be negated. In a study of 63 patients who had received lamivudine for 3 years, patients who had lamivudine-resistant mutants for >2 years were more likely to have worse necroinflammatory scores compared with the baseline biopsy: 3/22 (14%) versus 1/22 (5%) in those who had wild-type HBV [54]. Eleven (50%) patients with lamivudine resistance had unchanged necroinflammatory scores and 8/22 (36%) had improved scores, indicating that histological benefit may be maintained in most patients who had lamivudine resistance for 1--3 years. A more marked impact of lamivudine resistance on liver histology was reported in a study of 51 Asian patients receiving long-term lamivudine: six out of nine (67%) patients with lamivudine resistance had evidence of deterioration in necroinflammatory score when the year 3 biopsies were compared with the year 1 biopsies [2].
Hepatitis flares
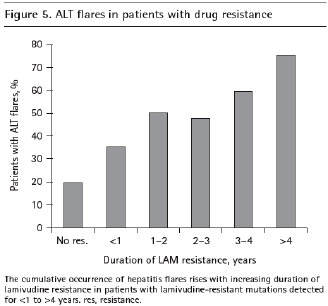
Hepatitis flares have been frequently reported in patients with lamivudine resistance but the incidence and severity of these flares are not known. A study of 998 patients treated with lamivudine for a median of 4 years found that hepatitis flares (>3 × ULN after initial normalization) were common in patients with lamivudine resistance (21--29% per year), but severe hepatitis flares (ALT >10 × ULN) were infrequent, with annual rates of 3--5% in those with lamivudine resistance and 1--3% in those without lamivudine resistance [55]. The cumulative occurrence of hepatitis flares rose with increasing duration of lamivudine resistance from 35% to 75% in patients with lamivudine- resistant mutations detected for <1 to >4 years (Figure 5). While most of the flares were related to increasing HBV DNA levels, a small proportion was associated with HBeAg seroconversion.
Hepatic decompensation
Hepatic decompensation and death from liver failure have been reported in patients with lamivudine resistance, particularly among transplant patients [28,56]. In most instances, hepatic decompensation was triggered by a severe hepatitis flare. The incidence of hepatic decompensation among patients with lamivudine resistance was assessed in a study of 998 patients who received lamivudine for a median of 4 years and was found to be very low among patients with lamivudine resistance for <4 years (0--2%), but increased to 6% in patients who had lamivudine resistance for >4 years [55]. These data suggest that most patients with lamivudine resistance initially remain stable but liver disease may worsen over time. It should also be emphasized that the generally favourable outcome in this study may be related to the young age of the patients (median of 32 years) and the low percentage (10%) of patients with cirrhosis.
Increased risk of HBV recurrence post-transplant
High serum HBV DNA level pre-transplant is the strongest predictor of HBV recurrence post-transplant [57]. Patients who develop lamivudine resistance pretransplant have an increased risk of HBV recurrence
The cumulative occurrence of hepatitis flares rises with increasing duration of lamivudine resistance in patients with lamivudine-resistant mutations detected for <1 to >4 years. res, resistance. post-transplant because HBV DNA levels may not be adequately suppressed at the time of transplant and the prophylactic therapy post-transplant may be ineffective unless additional antiviral therapy such as adefovir is administered.
|
|
| |
| |
 |
|
| |
| |
Public health concerns
Lamivudine-resistant HBV mutants have been detected in hepatitis B patients who had no prior exposure to the drug [16]. Transmission of lamivudine-resistant HBV mutants to seronegative individuals has also been reported [58]. Some investigators have suggested that antiviral drug-resistant HBV mutants may have mutations in the overlapping S gene that diminish the protective efficacy of HBV vaccine [30]. These concerns need to be confirmed. Nevertheless, vigilance and surveillance are needed in light of increasing reports of transmission of antiretroviral drug-resistant HIV [59,60]. De novo infection with antiviral-resistant HBV is a potentially serious public health problem as it limits treatment options.
Risk factors for antiviral resistance
Selection of resistant mutations is most likely to occur with antiviral agents that do not provide rapid and potent viral suppression (Table 2).
Lamivudine resistance
Pretreatment factors that have been found to be associated with an increased risk of lamivudine resistance include high ALT, high serum HBV DNA levels, male gender and increased body mass index (BMI) [61,62]. High serum HBV DNA levels reflect a greater pool of virus and a higher rate of virus replication, thereby increasing the likelihood that drug-resistant mutations will be selected. It is not clear why patients with high ALT, a predictor for treatment-related HBeAg seroconversion, would have an increased risk of lamivudine resistance. One potential explanation is that patients with high ALT have a higher rate of hepatocyte turnover, which facilitates creation of new replication space for drug-resistant mutants [63]. The association between high BMI and lamivudine resistance suggests that the approved dose of 100 mg daily may be suboptimal for overweight patients.
Table 2. Risk factors for lamivudine resistance
Elevated pretreatment HBV DNA
Elevated pretreatment ALT
Male gender
High body mass index
Previous exposure to lamivudine
Previous exposure to famciclovir
HBV DNA >103 copies/ml after 6 months on lamivudine
Lack of an early virological response has also been found to predict lamivudine resistance. A study of 159 patients treated with lamivudine for up to 30 months showed that 63% of patients with serum HBV DNA >103 copies/ml after 6 months of treatment had lamivudine resistance compared with 13% of those with <103 copies/ml (OR=11.5; 95% CI 3.2--40.5) [64]. Previous exposure to lamivudine or other antiviral drugs that select for cross-resistant mutants will also increase the risk of lamivudine resistance [65]. Resistant mutants may have been selected and archived even in patients who did not have breakthrough infection during previous therapy.
Resistance to other antiviral agents
Preliminary data suggest that patients with lamivudine resistance who received a combination of lamivudine and adefovir, are less likely to develop adefovir resistance than those who received adefovir monotherapy. This may be related to the antiviral activity of lamivudine against adefovir-resistant HBV. In contrast, preliminary data suggest that continuation of lamivudine treatment in patients with lamivudine resistance may increase the risk of entecavir resistance [66]. As more data emerge, guidelines on appropriate management of patients with lamivudine resistance will need to be updated [67].
Management of hepatitis B patients with antiviral resistance
The optimal management of hepatitis B patients with antiviral resistance has not been determined. Strategies include prevention of resistance through judicious use of antiviral agents and avoidance of sequential monotherapy. For patients with antiviral resistance, the options include treatment withdrawal with or without the addition of salvage therapy.
Judicious use of antiviral therapy
As with HIV infection, the most important approach in the management of antiviral resistance is to prevent or minimize the risk of resistance by limiting exposure to antiviral agents if treatment is deemed unnecessary or futile. Thus, treatment should be deferred in hepatitis B patients (outlined below) with minimal liver disease who have a low likelihood of sustained benefit [45,68]. A similar recommendation is made for patients with asymptomatic HIV infection who have CD4 T cells >350/ml and HIV-RNA level <20000 copies/ml [69].
HBeAg-positive patients with normal ALT. These are usually young Asian patients with perinatally acquired HBV infection who are still in the immune tolerant phase. Neither interferon nor lamivudine resulted in higher rates of HBeAg seroconversion compared with placebo [70--73].
HBeAg-positive patients with very high ALT. Data from placebo-treated patients in lamivudine trials demonstrated that 14--15% patients with ALT >5×ULN will undergo spontaneous HBeAg seroconversion within 1 year of presentation [70,74]. Thus, patients who are anicteric and have no evidence of hepatic decompensation may be monitored and treatment initiated if spontaneous HBeAg seroconversion does not occur in the ensuing 6 months.
HBeAg-negative patients with normal ALT and low or undetectable HBV DNA. HBV reactivation may occur in some inactive carriers but there is no evidence that any of the approved antiviral therapies can eradicate HBV or prevent reactivation.
Hazards of sequential monotherapy
Sequential monotherapy promotes the emergence of multi-drug resistance [65]. Studies in patients with HIV infection clearly demonstrated that combination therapy with three or more highly active antiretroviral agents is more effective in suppressing HIV viral load and in delaying the emergence of resistance compared with monotherapy [75,76]. However, combination therapy is only an option when several drugs, preferably with different mechanisms of action, are available. For HIV infection, at least 15 drugs representing three different classes have been approved by the FDA. In contrast, only three agents (interferon-α, lamivudine and adefovir dipivoxil) have been approved for patients with chronic HBV infection. As discussed earlier, clinical trials have not identified the appropriate combination that will lead to higher rates of sustained viral suppression and lower rates of drug resistance. Thus, until an effective combination is identified, physicians will continue to use monotherapy and add a second agent when resistance occurs. Fortunately, drug-resistant HBV mutations are less common and take longer to emerge compared with drug-resistant HIV [18].
Treatment withdrawal
Withdrawal of therapy is an option that has been studied in patients with lamivudine-resistant HBV. Combined data from Phase III trials indicate that significant post-withdrawal ALT flares (>2 × baseline level) occurred in 6/33 (18%) patients with lamivudine- resistant HBV but none showed signs of hepatic decompensation [61]. In another study of 66 Asian patients with lamivudine-resistant HBV, ALT flares were observed in 67% of patients who continued lamivudine treatment compared with 54% patients who discontinued treatment [77]. Therefore, treatment withdrawal may be considered in patients who do not have underlying cirrhosis and are not immunosuppressed, but these patients must be monitored closely so that salvage therapy can be initiated promptly if patients develop moderate or severe flares.
Treatment continuation
Patients who have normal or minimally elevated ALT despite antiviral resistance may continue to derive benefit from the treatment. In one study, necroinflammatory scores improved or did not change in 19/22 (86%) of patients with lamivudine resistance for >2 years [54]. These patients require close monitoring so that salvage therapy can be started if moderate or severe hepatitis flares occur. Adding antiviral agents with activity against drugresistant mutants
Addition of antiviral agents with activity against antiviral-resistant mutants is recommended particularly for patients with underlying cirrhosis, immunosuppressed patients and patients who develop worsening liver disease or moderate/severe hepatitis flares. Three antiviral agents (adefovir, tenofovir and entecavir) have demonstrated in vivo activity against lamivudine resistant HBV (Table 3) [6,78--80]. Lamivudine and entecavir have in vivo activity against adefovir-resistant HBV and adefovir has in vivo activity against entecavir- resistant HBV [38].
Adefovir dipivoxil. In an open-label study, 128 pre- and 196 post-liver transplant patients with breakthrough infection during lamivudine therapy received adefovir 10 mg daily for a mean duration of 19 and 56 weeks, respectively [6]. Most patients continued to receive lamivudine. Undetectable HBV DNA by PCR assay and normal ALT levels were achieved in 81% and 76% pre-transplant patients and in 34% and 49%
|
|
| |
| |
 |
|
| |
| |
post-transplant patients, respectively. Child--Turcotte--Pugh (CTP) scores improved or stabilized in over 90% patients in both groups. Nephrotoxicity (reproducible increase in serum creatinine >0.5 mg/dl) occurred in 15 (12%) pre- and 26 (13%) post-transplant patients, but it was unclear if the renal effects were related to adefovir, underlying liver failure or concomitant nephrotoxic medications.
Another study found that viral suppression was comparable in patients with compensated liver disease and lamivudine resistance who received adefovir monotherapy or a combination of lamivudine and adefovir [5]. These data suggest that lamivudine may be discontinued when adefovir is initiated. However, only 19 patients were included in the adefovir monotherapy group. Furthermore, Grade 3 ALT flares (5--10 × ULN) occurred in one-third of the patients who stopped lamivudine. These findings led to the recommendation of a 4--12 week overlap period, particularly for patients with underlying cirrhosis or immunosuppression. Whether lamivudine should be continued beyond the transition period depends on further data clarifying the role of continued lamivudine treatment in reducing the risk of adefovir resistance.
Tenofovir. Tenofovir disoproxil fumarate is a nucleotide analogue that is active against wild-type and lamivudine resistant HBV. On a mole for mole basis, tenofovir and adefovir have comparable antiviral activity in vitro. However, the approved dose of tenofovir (300 mg daily) is 30 times that of adefovir (10 mg daily). This may account for the clinical observation that tenofovir is more potent in decreasing serum HBV DNA levels.
In a study of 55 patients with lamivudine-resistant HBV, patients were treated with tenofovir or adefovir for at least 3 months [81]. At month 6, serum HBV DNA decreased by a mean of 5--7 log10 copies/ml in the tenofovir group and 2 log10 copies/ml in the adefovir group. Similar results were reported in a case series of nine patients with lamivudine-resistant HBV who received tenofovir and ongoing lamivudine for 12 months [79]. These data indicate that tenofovir is an effective and more potent alternative to adefovir in patients with lamivudine resistance.
Entecavir. In vitro data showed that entecavir is less effective against lamivudine-resistant HBV than wildtype HBV [82]. Because entecavir is 100-fold more potent than lamivudine in suppressing HBV replication, in vivo activity against lamivudine-resistant HBV can be achieved using higher doses of entecavir 1.0 mg versus 0.1--0.5 mg for wild-type HBV. Preliminary data from a Phase II trial in 181 lamivudine-refractory patients confirmed that entecavir in doses of 0.5 or 1.0 mg daily resulted in 4 log10 decrease in serum HBV DNA after 52 weeks of therapy [78]. This study also suggested that lamivudine-resistant patients can be safely switched to entecavir without ongoing lamivudine therapy as ALT flares were observed in only four (2%) patients receiving 0.5 or 1.0 mg entecavir. Further data are needed to determine the role of entecavir in the management of patients with lamivudine resistance, especially in light of recent findings indicating that pre-existing lamivudine-resistant mutations increase the risk of entecavir resistance.
A practical approach
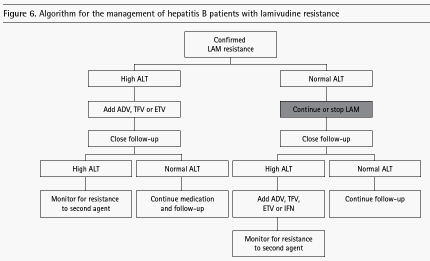
An algorithm for the management of hepatitis B patients with antiviral resistance is shown in Figure 6. Confirmation of genotypic resistance will help in identifying medication-noncompliant patients who will benefit from continuation of current treatment.
Patients who are confirmed as having antiviral resistance but have normal ALT with no cirrhosis or immunosuppression may continue current treatment but close monitoring is required so that salvage therapy can be added if liver disease worsens. Some investigators recommend withdrawal of lamivudine in this setting. The advantage of continuing treatment is that lamivudine may continue to exert a suppressive effect on residual wild-type HBV. The disadvantage is that continued treatment will select for additional compensatory mutations, which may restore replication fitness to the mutant HBV. Patients with worsening liver disease or moderate/severe hepatitis flares and those with cirrhosis or immunosuppression should receive additional antiviral therapy that is effective against the drug-resistant mutant [67]. Whether the initial therapy should be continued remains unclear and may vary depending on the choice of the second antiviral agent. Although the optimal duration of treatment for hepatitis B patients with antiviral resistance is unknown, it is likely that most patients will require life-long treatment, which raises the concern that resistance to multiple drugs may develop over time.
|
|
| |
| |
 |
|
| |
| |
Patients with confirmed lamivudine resistance who have normal ALT and no cirrhosis or immunosuppression may continue current treatment but require close monitoring in case of worsening liver disease. However, some investigators recommend discontinuing treatment in these patients to avoid selection of compensatory mutations. Patients with lamivudine resistance who have worsening liver disease or moderate/severe hepatitis flares and those with cirrhosis or immunosuppression should receive additional antiviral therapy effective against the drug-resistant mutant. Patients should be also monitored for resistance to salvage therapy. ALT, alanine aminotransferase; LAM, lamivudine; ADV, adefovir; TFV, tenofovir; ETV, entecavir; IFN, interferon.
Summary
Drug-resistant mutations have been reported with all antiviral agents used in the treatment of hepatitis B. Resistance to lamivudine occurs at a higher rate, while resistance to adefovir and entecavir is infrequent and slow to emerge. The rate of drug resistance increases with the duration of treatment and antiviral resistance may result in hepatitis flares and hepatic decompensation. The management of hepatitis B patients with antiviral resistance is evolving. Adefovir, tenofovir and entecavir have demonstrated in vivo activity against lamivudine-resistant HBV. However, further studies are needed to clarify the role of each of these agents and the need to continue lamivudine in patients with lamivudine-resistant HBV. The most effective way to prevent drug resistance is to avoid indiscriminate use of antiviral therapy. Sequential monotherapy should be discouraged, but until effective combination regimens become available, judicious use of single antiviral agents in patients who are likely to derive benefit from treatment may remain a common practice.
Author disclosures
Dr Fung is supported by grants from the Canadian Association for the Study of the Liver, GlaxoSmithKline Canada and the Canadian Institutes of Health Research. Dr Lok received research support from GlaxoSmithKline, Bristol-Myers Squibb, Gilead, Idenix, Roche, Valeant and Innogenetics and serves on advisory boards for Idenix, GlaxoSmithKline, Gilead, Roche, Innogenetics, Nabi, Bristol-Myers Squibb, Anadys and XTL Pharmaceuticals.
REFERENCES
1. Liaw YF, Leung NW, Chang TT, Guan R, Tai DI, Ng KY, Chien RN, Dent J, Roman L, Edmundson S & Lai CL. Effects of extended lamivudine therapy in Asian patients with chronic hepatitis B. Gastroenterology 2000; 119:172--180.
2. Leung NW, Lai CL, Chang TT, Guan R, Lee CM, Ng KY, Wu PC, Dent JC, Edmundson S, Condreay LD & Chien RN; on behalf of the Asia Hepatitis Lamivudine Study Group. Extended lamivudine treatment in patients with chronic hepatitis B enhances hepatitis B e antigen seroconversion rates: results after 3 years of therapy. Hepatology 2001; 33:1527--1532.
3. Chang TT, Lai CL, Chien RN, Guan R, Lim SG, Lee CM, Ng KY, Nicholls GJ, Dent JC & Leung NW. Four years of lamivudine treatment in Chinese patients with chronic hepatitis B. Journal of Gastroenterology & Hepatology 2004; 19:1276--1282.
4. Qi X, Snow A, Thibault V, Zhu Y, Curtis M, Hadziyannis SJ, Brosgart CL, Currie G, Arterburn S, Gibbs CS, Miller MD & Xiong S. Long-term incidence of adefovir dipivoxil (ADV) resistance in chronic hepatitis B (CHB) patients after 144 weeks of therapy (abstract). Journal of Hepatology 2004; 40:A57.
5. Peters M, Hann HW, Martin P, Heathcote EJ, Buggisch P, Rubin R, Bourliere M, Kowdley K, Trepo C, Gray Df D, Sullivan M, Kleber K, Ebrahimi R, Xiong S & Brosgart CL. Adefovir dipivoxil alone or in combination with lamivudine in patients with lamivudine-resistant chronic hepatitis B. Gastroenterology 2004; 126:91--101.
6. Schiff E, Lai CL, Hadziyannis S, Neuhaus P, Terrault N, Colombo M, Tillmann HL, Samuel D, Zeuzem S, Lilly L, Rendina M, Villeneuve JP, Lama N, James C, Wulfsohn MS, Namini H, Westland C, Xiong S, Choy GS, Van Doren S, Fry J & Brosgart CL; Behalf of the Adefovir Dipovoxil Study 435 International Investigators Group. Adefovir dipivoxil therapy for lamivudine-resistant hepatitis B in pre- and post-liver transplantation patients. Hepatology 2003; 38:1419--1427.
7. Marcellin P, Chang TT, Lim SG, Tong M, Sievert W, Schiffman M, Jeffers L, Goodman Z, Wulfsohn MS, Xiong S, Fry J & Brosgart CL; Adefovir Dipivoxil 437 Study Group. Adefovir dipivoxil for the treatment of hepatitis B e-antigen-positive chronic hepatitis B. New England Journal of Medicine 2003; 348:808--816.
8. Locarnini S. Molecular virology of hepatitis B virus. Seminars in Liver Disease 2004; 24:3--10.
9. Nowak MA, Bonhoeffer S, Hill AM, Boehme R, Thomas HC & McDade H. Viral dynamics in hepatitis B virus infection. Proceedings of the National Academy of Sciences, USA 1996; 93:4398--4402.
10. Zeuzem S, de Man RA, Honkoop P, Roth WK, Schalm SW & Schmidt JM. Dynamics of hepatitis B virus infection in vivo. Journal of Hepatology 1997; 27:431--436.
11. Roberts JD, Bebenek K & Kunkel TA. The accuracy of reverse transcriptase from HIV-1. Science 1988; 242:1171--1173.
12. Summers J & Mason WS. Replication of the genome of a hepatitis B-like virus by reverse transcription of an RNA intermediate. Cell 1982; 29:403--415.
13. Fourel I, Cullen JM, Saputelli J, Aldrich CF, Schaffer P, Averett DR, Pugh J & Mason WS. Evidence that hepatocyte turnover is required for rapid clearance of duck hepatitis B virus during antiviral therapy of chronically infected ducks. Journal of Virology 1994; 68:8321--8330.
14. Poch O, Sauvaget I, Delarue M & Tordo N. Identification of four conserved motifs among the RNA-dependent polymerase encoding elements. EMBO Journal 2004; 8:3867--3874.
15. Najera I, Holguin A, Quinones-Mateu ME, Munoz- Fernandez MA, Najera R, Lopez-Galindez C & Domingo E. Pol gene quasispecies of human immunodeficiency virus: mutations associated with drug resistance in virus from patients undergoing no drug therapy. Journal of Virology 1995; 69:23--31.
16. Kobayashi S, Ide T & Sata M. Detection of YMDD motif mutations in some lamivudine-untreated asymptomatic hepatitis B virus carriers. Journal of Hepatology 2001; 34:584--586.
17. Stuyver L, Locarnini S, Lok A, Richman DD, Carman WF, Dienstag JL & Schinazi RF. Nomenclature for antiviralresistant human hepatitis B virus mutations in the polymerase region. Hepatology 2001; 33:751--757.
18. Schuurman R, Nijhuis M, van Leeuwen R, Schipper P, de Jong D, Collis P, Danner SA, Mulder J, Loveday C & Christopherson C. Rapid changes in human immunodeficiency virus type 1 RNA load and appearance of drug-resistant virus populations in persons treated with lamivudine (3TC). Journal of Infectious Diseases 1995; 171:1411--1419.
19. Gao HQ, Boyer P, Sarafianos S, Arnold E & Hughes S. The role of steric hindrance in 3TC resistance of human immunodeficiency virus type-1 reverse transcriptase. Journal of Molecular Biology 2000; 300:403--418.
20. Allen MI, Deslauriers M, Andrews CW, Tipples GA, Walters KA, Tyrrell DL, Brown N & Condreay LD. Identification and characterization of mutations in hepatitis B virus resistant to lamivudine. Hepatology 1998; 27:1670--1677.
21. Hadziyannis S, Papatheodoridis GV, Dimou E, Laras A & Papaioannou C. Efficacy of long-term lamivudine monotherapy in patients with hepatitis B e antigen-negative chronic hepatitis B. Hepatology 2000; 32:847--851.
22. Locarnini S. Hepatitis B viral resistance: mechanisms and diagnosis. Journal of Hepatology 2003; 39:S124--S132.
23. Chayama K, Suzuki Y, Kobayashi M, Tsubota A, Hashimoto M, Miyano Y, Koike H, Kobayashi M, Koida I, Arase Y, Saitoh S, Murashima N, Ikeda K & Kumada H. Emergence and takeover of YMDD motif mutant hepatitis B virus during long-term lamivudine therapy and retakeover by wild type after cessation of therapy. Hepatology 1998; 27:1711--1716.
24. Ono-Nita SK, Kato N, Shiratori Y, Lan KH, Yohida H, Carrilho FJ & Omata M. Susceptibility of lamivudine resistant hepatitis B virus to other reverse transciptase inhibitors. Journal of Clinical Investigation 1999; 103:1635--1640.
25. Fu L & Cheng YC. Role of additional mutations outside the YMDD motif of hepatitis B virus polymerase in L(-)SddC (3TC) resistance. Biochemical Pharmacology 1998; 55:1567--1572.
26. Lok AS, Zoulim F, Locarnini S, Mangia A, Niro G, Decraemer H, Maertens G, Hulstaert F, De Vreese K & Sablon E. Monitoring drug resistance in chronic hepatitis B virus (HBV)-infected patients during lamivudine therapy: evaluation of performance of INNO-LiPA HBV DR assay. Journal of Clinical Microbiology 2002; 40:3729--3734.
27. Ogata N, Fujii K, Takigawa S, Nomoto M, Ichida T & Asakura H. Novel patterns of amino acid mutations in the hepatitis B virus polymerase in association with resistance to lamivudine therapy in Japanese patients with chronic hepatitis B. Journal of Medical Virology 1999; 59:270--276.
28. Bock C, Tillmann H, Torresi J, Klempnauer J, Locarnini S, Manns M & Trautwein C. Selection of hepatitis B virus polymerase mutants with enhanced replication by lamivudine treatment after liver transplantation. Gastroenterology 2002; 122:264--273.
29. Torresi J. The virological and clinical significance of mutations in the overlapping envelope and polymerase genes of hepatitis B virus. Journal of Clinical Virology 2002; 25:97--106.
30. Torresi J, Earnest-Silveira L, Deliyannis G, Edgtton K, Zhuang H, Locarnini S, Fyfe J, Sozzi T & Jackson DC. Vaccine-induced anti-HBs escape mutants of hepatitis B virus arising as a consequence of sequence changes in the overlapping polymerase gene that are selected by lamivudine therapy. Virology 2002; 293:305--313.
31. Gish R, Leung N, Wang C, Corey L, Sacks L, Fried M, Wright T, Huy T, Chan F, Rousseau F, Mommeja-Marin H, Snow A, Anderson J, Rigney A & Mondou E. Antiviral activity, safety and incidence of resistance in chronically infected hepatitis B (CHB) patients given once daily emtricitabine for 2 years. Hepatology 2003; 38:A372.
32. Lai CL, Leung N, Teo EK, Tong M, Wong F, Hann W & Han S. Results of a one-year international phase IIB comparative trial of telbivudine, lamivudine, and the combination, in patients with chronic hepatitis B (abstract). Hepatology 2003; 38:A219.
33. Zoulim F, Pichoud C, Wang Z, Aguesse-Germonb S & Trepo C. Hepatitis B virus genome variability during famciclovir therapy. Hepatology 1997; 26:A1200.
34. Tillmann H, Trautwein C, Bock C, Glowenka M, Kruger M, Boker K, Jackel E, Pichlmayr R, Condreay L, Bruns I, Deslaurias M, Gauthier J & Manns M. Response and mutations in patients sequentially treated with lamivudine and famciclovir for recurrent hepatitis B after liver transplantation. Hepatology 1997; 26:A1202.
35. Westland CE, Yang H, Delaney WE, Gibbs CS, Miller MD, Wulfsohn M, Fry J, Brosgart C, Xiong S & 437 and 438 Study Teams. Week 48 resistance surveillance in two phase 3 clinical studies of adefovir dipivoxil for chronic hepatitis B. Hepatology 2003; 38:96--103.
36. Xiong S, Yang H, Westland CE, Delaney WE, Colledge D, Bartholomeusz A, Thibault V, Benhamou Y, Angus P, Wulfsohn M, Gibbs CS, Fry J, Brosgart C & Locarnini S. Resistance surveillance of HBeAg- chronic hepatitis B (CHB) patients treated for two years with adefovir dipivoxil (ADV). Journal of Hepatology 2003; 38:182.
37. Bartholomeusz A, Tehan BG & Chalmers DK. Comparisons of the HBV and HIV polymerase, and antiviral resistance mutations. Antiviral Therapy 2004; 9:149--160.
38. Angus P, Vaughan R, Xiong S, Yang H, Delaney WE, Gibbs CS, Brosgart C, Colledge D, Edwards R, Ayres A, Bartholomeusz A & Locarnini S. Resistance to adefovir dipivoxil therapy associated with the selection of a novel mutation in the HBV polymerase. Gastroenterology 2003; 125:292--297.
39. Yang H, Qi X, Das K, Arnold E, Westland CE, Delaney WE. Brosgart CL, Gibbs CS, Miller MD & Xiong S. In vitro characterization and molecular modeling analysis of a novel adefovir resistance mutation rtN236T in the HBV polymerase. Journal of Hepatology 2004; 40:A383.
40. Lai CL, Rosmawati M, Lao J, Van Vlierberghe H, Anderson F, Thomas N & Dehertogh D. Entecavir is superior to lamivudine in reducing hepatitis B virus DNA in patients with chronic hepatitis B infection. Gastroenterology 2002; 123:1831--1838.
41. Barbaro G, Zechini F, Pellicelli A, Francavilla R, Scotto G, Bacca D, Bruno M, Babudieri S, Annese M, Matarazzo F, Di Stefano G & Barbarini G; Lamivudine Italian Study Group Investigators. Long-term efficacy of interferon alpha-2b and lamivudine in combination compared to lamivudine monotherapy in patients with chronic hepatitis B. An Italian multicenter, randomized trial. Journal of Hepatology 2001; 35:406.
42. Sanantonio T, Niro GA, Sinisi E, Leandro G, Insalata M, Guastadisegni A, Facciorusso D, Gravinese E, Andriulli A & Pastore G. Lamivudine/interferon combination therapy in anti-HBe positive chronic hepatitis B patients: a controlled pilot study. Journal of Hepatology 2002; 36:799.
43. Schalm SW, Heathcote J, Cianciara J, Farrell G, Sherman M, Willems B, Dhillon A, Moorat A, Barber J & Gray DF. Lamivudine and alpha interferon combination treatment of patients with chronic hepatitis B infection: a randomised trial. Gut 2000; 46:562-568.
44. Sung JJY, Lai JY, Zeuzem S, Chow WC, Heathcote E, Perrillo R, Brosgart C, Woessner M, Scott SA & Campbell FM. A randomised double-blind phase ll study of lamivudine (LAM) compared to lamivudine plus adefovir dipivoxil (ADV) for treatment naive patients with chronic hepatitis B: week 52 analysis (abstract). Journal of Hepatology 2003; 38:25A.
45. Lok A & McMahon B. Chronic hepatitis B. Hepatology 2001; 34:1225--1241.
46. Lok ASF & McMahon B. AASLD Practice Guidelines: chronic hepatitis B. Hepatology 2004.
47. Papatheodoridis G, Dimou E, Laras A, Papadimitropoulos V & Hadziyannis S. Course of virologic breakthroughs under long-term lamivudine in HBeAg-negative precore mutant HBV liver disease. Hepatology 2002; 32:219--226.
48. Dienstag JL, Schiff ER, Wright TL, Perrillo RP, Hann HW, Goodman Z, Crowther L, Condreay LD, Woessner M, Rubin M & Brown NA. Lamivudine as initial treatment for chronic hepatitis B in the United States. New England Journal of Medicine 1999; 341:1256--1263.
49. Lai CL, Chien RN, Leung N, Chang TT, Guan R, Tai DI, Ng KY, Wu PC, Dent JC, Barber J, Stephenson SL & Gray DF. A one-year trial of lamivudine for chronic hepatitis B. New England Journal of Medicine 1998; 339:61--68.
50. Hirsch MS, Brun-Vézinet F, Clotet B, Conway B, Kuritzkes DR, D'Aquila RT, Demeter LM, Hammer SM, Johnson VA, Loveday C, Mellors JW, Jacobsen DM & Richman DD. Antiretroviral drug resistance testing in adults infected with human immunodeficiency virus type 1: 2003 recommendations of an International AIDS Society-USA Panel. Clinical Infectious Diseases 2003; 37:113--128.
51. Pas SD, de Man RA, Fries E, van Nunen AB, Osterhaus AD & Niesters HGM. The dynamics of mutations in the polymerase gene of hepatitis B virus during and after lamivudine therapy. Journal of Clinical Virology 2002; 25:63--71.
52. Hussain M, Chu CJ, Sablon E & Lok ASF. Rapid and sensitive assays for the determination of hepatitis B virus (HBV) genotypes and detection of HBV precore and core promoter variants. Journal of Clinical Microbiology 2003; 41:3699--3705.
53. Liaw YF, Chien RN, Yeh CT, Tsai SL, Chu CM. Acute exacerbation and hepatitis B virus clearance after emergence of YMDD motif mutation during lamivudine therapy. Hepatology 1999; 30:567--572.
54. Dienstag J, Goldin R, Heathcote EJ, Hann H, Woessner M, Stephenson SL, Gardner S, Gray DF, Schiff ER. Histological outcome during long-term lamivudine therapy. Gastroenterology 2003; 124:105--117.
55. Lok AS, Lai CL, Leung N, Yao GB, Cui ZY, Schiff E, Dienstag J, Heathcote EJ, Little NR, Griffiths DA, Gardner SD & Castiglia M. Long-term safety of lamivudine treatment in patients with chronic hepatitis B. Gastroenterology 2003; 125:1714--1722.
56. Rosenau J, Bahr MJ, Tillmann HL, Trautwein C, Klempnauer J, Manns MP & Boker KHW. Lamivudine and low dose hepatitis B immune globulin as prophylaxis of hepatitis B reinfection after liver transplantation: possible role of mutations in the YMDD motif prior to transplantation as a risk factor for reinfection. Journal of Hepatology 2001; 34:895--902.
57. Mutimer D, Pillay D, Dragon E, Tang H, Ahmed M, O'Donnell K, Shaw J, Burroughs N, Rand D, Cane P, Martin B, Buchan S, Boxall E, Barmat S, Gutekunst K, McMaster P & Elias E. High pretreatment serum hepatitis B virus titer predicts failure of lamivudine prophylaxis and graft re-infection after liver transplantation. Journal of Hepatology 1999; 30:715--721.
58. Thibault V, Aubron-Olivier C, Agut H & Katlama C. Primary infection with a lamivudine-resistant hepatitis B virus. AIDS 2002; 16:131--133.
59. Little SJ, Holte S, Routy JP, Daar ES, Markowitz M, Collier AC, Koup RA, Mellors JW, Connick E, Conway B, Kilby M, Wang L, Whitcomb JM, Hellmann NS & Richman DD. Antiretroviral drug-resistance among patients recently infected with HIV. New England Journal of Medicine 2002; 347:385--394.
60. Yerly S, Kaiser L, Race E, Bru JP, Clavel F & Perrin L. Transmission of antiretroviral drug-resistant HIV-1 variants. Lancet 1999; 354:729--733.
61. Lai CL, Dienstag J, Schiff E, Leung N, Atkins M, Hunt C, Brown N, Woessner M, Boehme R & Condreay L. Prevalence and clinical correlates of YMDD variants during lamivudine therapy for patients with chronic hepatitis B. Clinical Infectious Diseases 2003; 36:687--696.
62. Nafa S, Ahmed S, Tavan D, Pichoud C, Berby F, Stuyver L, Johnson M, Merle P, Abidi H, Trepo C & Zoulim F. Early detection of viral resistance by determination of hepatitis B virus polymerase mutations in patients treated by lamivudine for chronic hepatitis B. Hepatology 2000; 32:1078--1088.
63. Zhou T, Saputelli J, Aldrich CF, Deslauriers M, Condreay L & Mason WS. Emergence of drug-resistant populations of woodchuck hepatitis virus in woodchucks treated with the antiviral nucleoside lamivudine. Antimicrobial Agents & Chemotherapy 1999; 43:1947--1954.
64. Yuen MF, Sablon E, Hui CK, Yuan HJ, Decraemer H & Lai CL. Factors associated with hepatitis B virus DNA breakthrough in patients receiving prolonged lamivudine therapy. Hepatology 2001; 34:785--791.
65. Mutimer D, Pillay D, Cook P, Ratcliffe D, O'Donnell K, Dowling D, Shaw J, Elias E & Cane PA. Selection of multiresistant hepatitis B virus during sequential nucleosideanalogue therapy. Journal of Infectious Diseases 2000; 181:713--716.
66. Locarnini S. Antiviral-resistant hepatitis B: current state of knowledge. AASLD Hepatitis Single Topic Conference: New Directions in the Treatment of Hepatitis B. 12--13 March 2004, Chicago, Ill., USA.
67. Lok ASF & McMahon BJ. Chronic hepatitis B: update of recommendations. Hepatology 2004; 39:857--861.
68. Lok A, Heathcote J & Hoofnagle J. Management of hepatitis B 2000, summary of a workshop. Gastroenterology 2001; 120:1828--1853.
69. NIH. Guidelines for the use of antiretroviral therapy in HIV-infected adults and adolescents. NIH website 2004.
70. Chien RN, Liaw YF, Atkins M & Asian Hepatitis Lamivudine Trial Group. Pretherapy alanine transaminase level as a determinant for hepatitis B e antigen seroconversion during lamivudine therapy in patients with chronic hepatitis B. Hepatology 1999; 30:770--774.
71. Lai CL, Lin HJ, Lau JN, Lok AS, Wu PC, Chung HT, Wong LK, Leung MP & Yeung CY. Effect of recombinant alpha 2 interferon with or without prednisone in Chinese HBsAg carrier children. Quarterly Journal of Medicine 1991; 78:155--163.
72. Lok AS, Lai CL, Wu PC & Leung EK. Long term followup in a randomised controlled trial of recombinant alpha 2-interferon in Chinese patients with chronic hepatitis B infection. Lancet 1988; 2:298--302.
73. Lok AS, Wu PC, Lai CL, Lau JY, Leung EK, Wong LS, Ma OC, Lauder IJ, Ng CP & Chung HT. A controlled trial of interferon with or without prednisone priming for chronic hepatitis B. Gastroenterology 1992; 102:2091--2097.
74. Perrillo RP, Lai CL, Liaw YF, Dienstag JL, Schiff ER, Schalm SW, Heathcote EJ, Brown NA, Atkins M, Woessner M & Gardner SD. Predictors of HBeAg loss after lamivudine treatment for chronic hepatitis B. Hepatology 2002; 36:186--194.
75. Gulick RM, Mellors JW, Havlir D, Eron JJ, Gonzalez C, McMahon D, Richman DD, Valentine FT, Jonas L, Meibohm A, Emini EA & Chodakewitz JA. Treatment with indinavir, zidovudine and lamivudine in adults with human immunodeficiency virus infection and prior antiretroviral therapy. New England Journal of Medicine 1997; 337:734--739.
76. Hammer SM, Squires KE, Hughes MD, Grimes JM, Demeter LM, Currier JS, Eron JJ Jr, Feinberg JE, Balfour HH Jr, Deyton LR, Chodakewitz JA & Fischl MA. A controlled trial of two nucleoside analogues plus indinavir in persons with human immunodeficiency virus infection and CD4 counts of 200 per cubic millimeter or less. New England Journal of Medicine 1997; 337:725--733.
77. Liaw YF, Chien RN & Yeh CT. No benefit to continue lamivudine after the emergence of YMDD mutations. Antiviral Therapy 2004; 9:145--148.
78. Gish R, Chang T, Hadziyannis S, Cianciara J, Rizzetto M, Schiff E, Pastore G, Jackson R, Thiry A & Hindes R. Lamivudine-refractory hepatitis B patients can be safely switched directly to entecavir 1 mg daily therapy (abstract). Journal of Hepatology 2004; 40:A428.
79. Kuo A, Dienstag J & Chung RT. Tenofovir disoproxil fumarate for the treatment of lamivudine-resistant hepatitis B. Clinical Gastroenterology & Hepatology 2004; 2:266--272.
80. Schiff E, Chang T. Gish R, Hadziyannis S, Cianciara J, Rizzetto M, Pastore G, Cross A & Hindes R. Entecavir 1.0 mg is consistently superior to lamivudine at 48 weeks across multiple baseline HBV disease characteristics in lamivudine-refractory patients. Journal of Hepatology 2004; 40:A442.
81. van Bommel F, Wunsche T, Schnurmann D, Bergk A, Wiedenmann B, Hopf U & Berg T. A comparison of adefovir and tenofovir in the treatment of lamivudineresistant hepatitis B virus (HBV) infection. Hepatology 2003; 38:A246.
82. Levine S, Hernandez D, Yamanaka G, Zhang S, Rose R, Weinheimer S & Colonno RJ. Efficacies of entecavir against lamivudine-resistant hepatitis B virus replication and recombinant polymerases in vitro. Antimicrobial Agents & Chemotherapy 2002; 46:2525--2532.
|
|
| |
| |
|
|
|