| |
Hepatitis B in the Human Immunodeficiency Virus-Infected Patient: Epidemiology, Natural History, and Treatment
|
| |
| |
Chloe L. Thio, M.D.
Assistant Professor of Medicine, Johns Hopkins University, Baltimore, Maryland
Semin Liver Dis 23(2):125-136, 2003. © 2003 Thieme Medical Publishers
Posted 06/19/2003 to www.medscape.com
Abstract and Introduction
Abstract
Coinfection with hepatitis B virus (HBV) is common in the human immunodeficiency virus-1 (HIV)-infected patient because of shared modes of transmission. HBV does not appear to alter HIV disease progression; however, HBV infection is more frequent and more severe in the HIV-infected population, emphasizing the importance of preventing HBV infection. The goal of anti-HBV therapy is prevention of cirrhosis because therapy does not eradicate the hepatic reservoirs (cccDNA). The approved therapies -- interferon-alfa, lamivudine, and adefovir -- each have a niche in the treatment of chronic hepatitis B in the HIV-infected population, but none has been well-studied in this setting. As new drugs currently in clinical trials become available, therapy for chronic hepatitis B will enter the promising era of combination therapy.
Introduction
HBV is the leading cause of chronic liver disease worldwide, including hepatitis, cirrhosis, and hepatocellular carcinoma.[1] HIV and HBV share routes of transmission and thus up to 10% of HIV-infected individuals have chronic hepatitis B.[2,3] Until recently, clinical manifestations of chronic hepatitis B in persons infected with HIV were uncommon because of competing risks for mortality from opportunistic infections associated with immunosuppression. Over the past several years, effective antiretroviral medications have increased the life expectancy of HIV-infected persons, resulting in ease a changing spectrum of HIV-associated diseases. Chronic liver disease from hepatitis C virus (HCV) has been recognized as a HIV-associated opportunistic infection,[4] and hepatitis B is being included in the forthcoming Department of Health and Human Services Guidelines for the Therapy of HIV-Associated Opportunistic Infections. Of additional concern is the rising incidence of acute hepatitis B in high-risk groups because of poor compliance with hepatitis B vaccination[5]; thus, HIV-HBV coinfection may become a growing problem. The purpose of this article is to review the virology, epidemiology, natural history, diagnosis, and treatment of persons infected with both HIV and HBV.
Virology
HBV is a relaxed, circular, partially double-stranded, enveloped DNA virus of the family Hepadnaviridae. The viral genome is approximately 3200 bases and encodes for four proteins using overlapping open reading frames: envelope (S), core (C), polymerase (P), and X. HBV gains entry into the hepatocyte by attaching to an unknown receptor with subsequent translocation to the nucleus. Here, the relaxed circular DNA becomes fully double stranded by conversion to cccDNA. This cccDNA perpetuates the infection by serving as the template for production of pregenomic RNA and messenger RNA (mRNA), which is translated into viral envelope and X proteins. The pregenomic RNA is translated into core and polymerase proteins and is also capsidated into core particles and then reverse transcribed into an open circular DNA molecule to become progeny core molecules. These progeny cores can either enter the nucleus to repeat the cycle and increase the pool of cccDNA or acquire envelope protein and be secreted to productively infect other hepatocytes.
The eight genotypes of HBV (A-H), which are defined by sequence divergence in the entire genome of greater than 8%, are correlated with clinical outcome, and their geographical distribution may explain some variation in clinical disease throughout the world. [6]Genotypes A and D are most prevalent in Western countries and in India, whereas B and C are found in Asia. Genotype E is found in Africa, F is found in Central and South America, and G and H are found in North America. Genotype A is associated with better outcome, B with hepatocellular carcinoma (HCC) in younger patients, and C with more severe disease and HCC in older patients. [7,8]Within each genotype, the four major subtypes based on the allelic determinants of the hepatitis B surface antigen (HBsAg) are adr, adw, ayr, and ayw. The adw subtype is the most common in the United States.[1] A correlation between subtype and disease has been suggested, although further studies are needed. [9]
Epidemiology of HIV and HBV Coinfection
Approximately 400 million persons worldwide have chronic hepatitis B, which is defined as the presence of HBsAg for a minimum of 6 months, and between 500,000 and 1 million persons die annually of HBV-related dis-ease.[1] In the United States, there are approximately 335,000 incident cases annually,[10] and about 1 million persons are chronically infected.[11] Coinfection with HIV and HBV is common because of shared modes of transmission, but HBV is transmitted more efficiently than is HIV both in men who have sex with men[12] and in injection drug users (IDUs). In a large cohort of IDUs in Baltimore, Maryland, 84% had a serum marker of HBV infection, whereas 24% were anti-HIV positive. [13]The Centers for Disease Control estimates that 7% of IDUs and 6% of men who have sex with men have chronic hepatitis B.[14] In the United States, up to 10% of all HIV-infected persons have HIV-HBV coinfection.[2,3] This problem is likely to be even larger in areas of the world where HBV is endemic and transmission occurs at a younger age, resulting in a higher rate of chronicity.
Natural History of HBV Infection
After an acute HBV infection acquired in adulthood, 90 to 95% of persons develop a broad, multispecific cellular immune response that eliminates the virus and ultimately leads to the development of protective antibodies to HBsAg (anti-HBs).[1] In some of these persons, a low level of viremia is present[15]; however, the clinical significance of this is not known. The remaining 5 to 10% of people become chronically infected, and < 1% develop a fulminant hepatitis.
Chronic hepatitis B predisposes to cirrhosis and HCC. The annual probability of developing cirrhosis in persons with chronic hepatitis B varies between 0.1 and 10% per year and is highest in those with circulating hepatitis B e antigen (HBeAg), older age, and elevated alanine aminotransferase (ALT) levels. [16- 18]Cirrhosis, the presence of HBeAg, and genotypes B and C are associated with an increased risk for developing HCC.[6,19,20]
Most people with chronic hepatitis B eventually clear HBeAg and develop antibodies to the e antigen (anti-HBe). In a 10-year study of 1536 Alaskan natives with chronic hepatitis B, 70% and 7% cleared HBeAg and HBsAg, respectively.[16] HBeAg clearance was associated with older age and elevated ALT. Individuals who clear HBeAg after many years may continue to have circulating HBsAg particles with either low or undetectable levels of HBV DNA and are defined as being inactive carriers. Inactive carriers have a reduced risk of hepatic decompensation and a twofold-lower risk of death[21,22]; however, up to 30% of them will have repeated flares of ALT, which can lead to progressive fibrosis.[23]
During the process of clearance of HBeAg and anti-HBe seroconversion, some individuals develop an escape variant that is not well-controlled by the immune response. These precore and core mutant viruses develop under selective immune pressure and are able to retain high levels of HBV replication. They are clinically detected by the absence of HBeAg and the presence of anti-HBe and high HBV DNA levels. This particular pattern is seen most commonly in East Asia and Southern Europe because of the higher prevalence of non-A genotypes, which predispose to the development of this mutant. These individuals may have slower disease progression than those who remain HBeAg positive, with a calculated annual incidence of cirrhosis of 1.3% compared with 2.4% in those with HBeAg.[17]
There are also individuals with chronic hepatitis B who do not have circulating HBsAg but have low levels of HBV DNA by sensitive polymerase chain reaction (PCR) methods. These individuals with occult hepatitis B often have detectable antibodies to hepatitis B core antigen (anti-HBc) as the only serological indicator of prior hepatitis B infection; however, the significance of this condition is not known.[24]
Diagnosis
All HIV-infected persons should be evaluated for chronic hepatitis B. The optimal testing strategy has not been determined; however, the most conservative approach is to screen for HBsAg and anti-HBs. Those who are negative for both markers should receive three doses of the hepatitis B vaccine.[25] This strategy will not detect occult hepatitis B, but if this is suspected, testing for anti-HBc and HBV DNA is warranted. In HIV-infected patients with a positive serum marker only for anti-HBc, a high rate (20 to 50%) of detection of HBV DNA in blood and elevated ALT has been reported but not confirmed.[26] However, the interpretation of an isolated anti-HBc is difficult because false-positive tests for anti-HBc occur[27] and because the clinical significance of anti-HBc alone or with low levels of HBV DNA is not known. Although anti-HBc alone may signify occult hepatitis B, a small proportion of these people may have acute hepatitis B with HBsAg below the limit of detection, so repeating HBsAg and anti-HBs in 3 to 6 months may be useful.
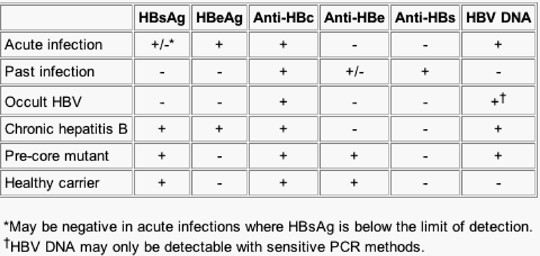
The serological pattern for viral clearance is negative HBsAg but positive anti-HBs and anti-HBc (if obtained) (Table 1). Individuals with chronic hepatitis B have HBsAg and anti-HBc without anti-HBs. Further evaluation of these individuals includes repeat HBsAg in 6 months, and, if still present, HBeAg, anti-HBe, and HBV DNA should be obtained to determine the state of their disease and to assess for treatment. At a recent National Institutes of Health (NIH) conference, an arbitrary HBV DNA level of 105copies/mL was proposed to indicate an active chronic hepatitis B infection.[28] As discussed earlier, those with the precore mutant will be HBeAg negative but usually have > 105copies/mL of HBV DNA.
Table 1. Serological Patterns Used in the Evaluation of Hepatitis B Virus Infection
|
|
| |
| |
 |
|
| |
| |
Impact of HIV Infection on HBV Disease Progression
The natural history of HBV disease progression is modified by HIV coinfection. A prospective study of 5293 gay men, 326 of whom had chronic hepatitis B, demonstrated that mortality attributable to liver disease was significantly increased in those who were coinfected with HIV and HBV (14.2/1000 person-years) compared with those with HBV alone (0.8/1000 person-years).[29] The investigators found that individuals with lower CD4 counts appeared to be at greatest risk. This finding also supports cross-sectional studies demonstrating increased cirrhosis in persons with HIV and HBV.[30]
In HIV-infected persons, clearance rates of HBsAg and HBeAg are decreased compared with those without HIV infection[31] and may be related to the degree of immunosuppression.[32] In a prospectively followed cohort, HIV-infected men had HBeAg loss of 12% in 5 years compared with 49% in the HIV-seronegative men.[31] HIV infection is also associated with reactivation of HBV,[33,34] accelerated loss of anti-HBs,[35] higher levels of HBV DNA,[30,31] and lower ALT levels.[30] The lower ALT levels suggest less hepatocyte destruction because of a depressed immune response and may contribute to the decreased effectiveness of anti-HBV therapy in coinfected individuals.
A severe, rapidly progressive form of liver disease described in liver transplant recipients, fibrosing cholestatic hepatitis, leads to liver graft rejection and is characterized by ballooning degeneration of hepatocytes and high HBV antigen expression in the liver.[36] This rare disease is thought to be due to a direct cytopathic effect of the virus and has also been described in patients with HIV infection.[37]
Impact of HBV on HIV Disease Progression
Although most evidence supports the idea that HIV accelerates the progression of HBV disease, the reciprocal relationship, the effect of HBV on HIV disease progression, is not clear. The available data suggest limited or no effect of HBV on progression to various HIV outcomes; however, the study populations were small and the time of HIV seroconversion was not documented.[3,38] Solomon et al[39] attempted to adjust for HIV serocon-version by CD4+ count stratification, and they also did not detect an association.
In contrast, two studies have noted associations between chronic hepatitis B and HIV progression. Eskild et al[40] studied 80 gay men and found that the 32 men who were anti-HBc-positive progressed to acquired immunodeficiency syndrome (AIDS) more rapidly. However, most of these anti-HBc-positive men had cleared the HBV infection. In a different study, HIV-HBV coin-fected patients with AIDS had decreased survival compared with patients with AIDS alone, but their analysis did not control for other factors that may have accounted for differences between the groups.[41]
HIV-HBV Coinfection and Highly Active Antiretroviral Therapy
Flares of liver enzymes in HIV-HBV coinfected patients receiving highly active antiretroviral therapy (HAART) can arise from one or more of the following causes, so a careful evaluation is required to discern the etiology prior to discontinuing or changing an effective HAART regimen. First, HBV infection clearly increases the risk for toxicity from antiretroviral medication[42,43]; however, only a few experience a severe, reversible hepatotoxicity (ALT > 5 x the upper limit of normal [ULN]) usually within the first 6 months of starting a regimen. Coin-fected patients are also at increased risk for the rare condition of hepatic steatosis and lactic acidosis from the nucleoside analogues, which can occur after years of therapy. Second, a flare may herald HBeAg seroconversion, so it is not unreasonable to continue HAART if this is a possibility. Third, immune reconstitution has been reported to lead to ALT elevations in patients with chronic hepatitis B.[44,45] Fourth, there have also been several case reports of reactivation or exacerbations of HBV after discontinuing lamivudine as part of a HAART regimen,[46,47] and it is well-described in the development of HBV resistance to lamivudine.[47] Fifth, reactivation of HBV replication has also been described in the setting of HAART independent of lamivudine withdrawal or resistance.[48] Last, superinfection with another hepatotropic virus must be considered.
Prevention of HBV Infection
Vaccination of persons at risk for acute HBV infection is the most effective means of prevention. After the standard vaccine dose, individuals with titers below 10 IU/L should be considered for revaccination.[49] HIV-HBV coinfected adults manifest decreased response to the hepatitis B vaccine. Their seroconversion rates to the inactivated plasma derived and the recombinant three-dose HBV vaccine are 50 to 56%[50,51] and 20 to 30%,[52,53] respectively, compared with the general population rate of 90%. This decreased antibody production rate appears to be independent of the mode of acquisition of HIV and tends to be more prominent with lower CD4+ counts.[52] A fourth immunization has had limited success in improving these responses.[53] Rey et al[54] demonstrated that three additional doses separated by 1 month each increased the response rate in coinfected individuals to 90%; however, after 1 year only 59% of them maintained protective anti-HBs titers.[54] If response to the vaccine does not occur, then other prevention methods, including barrier precautions to prevent sexual transmission; substance abuse counseling; and avoidance of tattooing, body piercing, and sharing personal care items, should be discussed.
The Centers for Disease Control recommends annual anti-HBs titers in hemodialysis patients with booster doses when levels fall below 10 IU/L because their immunologic memory may be less complete. Data do not exist regarding routine monitoring of anti-HBs titers and booster vaccinations in HIV-infected patients. However, it seems prudent to consider annual monitoring as has been recommended by the European Consensus Group on Hepatitis B Immunity[55] and to consider booster doses when the titer falls below 10 IU/L, especially in patients who have responded to HAART.
Preventive Care in Patients With Chronic Hepatitis B
Hepatitis A virus (HAV) vaccine should be administered to susceptible persons (negative immunoglobulin G [IgG] antibody). Although evidence does not exist for a more fulminant course of HAV in chronically HBV-infected individuals, vaccination is recommended because fulminant hepatitis A has been reported in HCV-infected individuals.[56]
Chronic hepatitis B predisposes to HCC, so routine interval screening for HCC is recommended. The American Association for the Study of Liver Disease (AASLD) recommends evaluation of HIV-negative patients every 6 to 12 months with alfa fetoprotein (AFP) or ultrasound of the liver, or both, especially if the patient is in a high-risk group (> 45 years of age, cirrhosis, or a family history of HCC).[28] In HIV-infected patients, the risk and natural history of HBV-related HCC have not been studied; thus, the optimal HCC screening method, interval, and cost-effective strategy are not known. Until more data become available, it seems reasonable to follow the recommendations for individuals uninfected by HIV.
Treatment of Chronic Hepatitis B In HIV-Infected Persons
The goal of chronic hepatitis B treatment is to prevent the development of cirrhosis rather than viral eradication because anti-HBV therapy does not directly affect the hepatic viral reservoirs (cccDNA). Because HBV integrates into the host genome, it is not known whether treatment will prevent the development of HCC, especially if cirrhosis exists at the time anti-HBV treatment is initiated. HCC has been described in persons with a remote history of HBV who did not have evidence of active viral replication,[57] and the HBV X gene can be detected in liver tissue from persons with HCC who are HBsAg-and HBV DNA-negative.[58]
The correlates of successful therapy are not clearly delineated, but markers of treatment success include improvement in liver histology, normalization of hepatic transaminases, substantial diminution in HBV viral load, and, in those with circulating HBeAg, the loss of e antigen with the development of anti-HBe ("HBeAg sero-conversion"). Sustained loss of HBsAg is considered by some to be a "complete response."[28] Although a decline in HBV viral load correlates with response, no target HBV viral load has been established, and HBV DNA assays are not standardized. The newer PCR-based methods have a lower sensitivity limit of 102 to 103 copies/mL compared with 105 for older assays.[59]
For persons without HIV, the AASLD recommends that persons with HBeAg-positive chronic hepatitis B with evidence of liver disease (ALT > two times normal or necroinflammation on biopsy) and those with HBeAg-negative chronic hepatitis B with HBV DNA > 105 copies/mL should be considered for treatment.[28]This value for the HBV DNA cutoff is arbitrary, but it is endorsed by the AASLD and seems reasonable based on the current state of knowledge. The same criteria can be applied to persons with HIV infection, with the caveat that the state of the HIV disease should receive priority in terms of evaluation and treatment because morbidity and mortality from HIV occurs more rapidly. Although the AASLD does not routinely recommend liver biopsy for the evaluation of chronic hepatitis B, a biopsy may support a decision to delay therapy in order to spare HAART toxicity or to prevent resistance in the HIVHBV coinfected person. The three currently approved therapies for chronic hepatitis B are interferon-alfa, lamivudine, and adefovir.
Interferon-alfa
Interferon-alfa, an agent with antiviral and immunomodulatory effects, is approved at a dose of 5 million units subcutaneous (SQ) daily or 10 million units SQ three times per week. For persons who have circulating HBeAg, 16 weeks of interferon-alfa are recommended, whereas those without circulating HBeAg require a minimum of 12 months.[28] Interferon-alfa is most effective in persons with high ALT (> 2 ULN), lower HBV DNA (< 2.8 x 107 copies/mL or 100 pg/mL), and circulating HBeAg.[60,61] Perrillo et al[61] found a 50% response rate in those with HBV DNA < 2.8 x 107 copies/mL and a 7% response with levels > 5.6 x 107 copies/mL. However, a reproducible, discriminatory level of HBV DNA above which response to interferon-alfa is unlikely has not been determined. The frequent side effects of interferon-alfa, including flulike symptoms, psychiatric effects, and bone marrow toxicity, have limited its use. Its advantages are higher HBeAg serocon-version rates and limited treatment duration (16 weeks) in persons who have detectable HBeAg. In HIV-infected individuals it has the additional advantage of not engendering HIV resistance. Interferon-alfa is contraindicated in decompensated liver disease because of an increased risk of death from liver failure or sepsis.[62]
A meta-analysis of randomized trials, which included 837 persons with an ALT > 2 ULN and circulating HBeAg, demonstrated a 33% HBeAg serocon-version rate in persons given 16 weeks of interferon-alfa compared with 12% in untreated controls.[63] A durable response of HBeAg clearance is seen in 80 to 90% of persons; however, HBV DNA and HBsAg often remain detectable.[64] Clearance of HBeAg and not HBV DNA has been correlated with improved clinical outcome.[65]
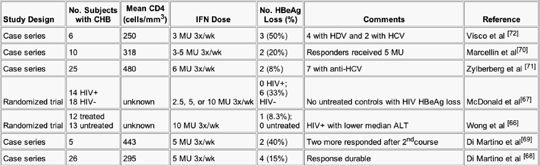
Studies of interferon-alfa therapy in HIV-HBV coinfected individuals are limited but suggest a decreased response compared with those without HIV infection. In the meta-analysis, 55 of the 837 individuals were coin-fected with HIV, and they showed a 38% decreased response compared with HIV uninfected persons (P=0.02).[63] However, none of these trials was randomized with respect to HIV status because the HIV status was determined retrospectively. In addition, the immune status (CD4 count) and the ALT values of these patients are not known. Two small, randomized studies and five case series have treated 98 HIV-HBV coinfected patients with interferon-alfa with a 14.3% HBeAg seroconversion rate (Table 2).[66-72] In the two randomized studies, none of the 14 untreated controls experienced HBeAg seroconversion.[66,67] Although data extrapolated from these studies collectively suggest a decreased response to interferon-alfa in HIV-infected patients, these studies did not consider the degree of immunosuppression, and interferon-alfa has not been evaluated in the era of HAART. Given that interferon-alfa is an immunomodulating agent, it seems reasonable that those with a more robust immune system (CD4 > 500) would have a greater likelihood of response. ALT levels, which correlate with response, were not considered, so coinfected individuals with high ALT values may have an equivalent response to the individuals not infected by HIV.
|
|
| |
| |
 |
|
| |
| |
A pegylated form of interferon-alfa that permits once weekly dosing and produces more constant levels is more efficacious than standard interferon-alfa is for treating chronic hepatitis C.[73] Preliminary observations suggest that this may also be true for the treatment of chronic hepatitis B in individuals not infected by HIV especially those with lower ALT values.[74]
In summary, although the response to standard interferon-alfa in HIV-HBV coinfected patients may be lower than it is in persons not infected by HIV, the data suggest that those most likely to benefit have an elevated ALT (> 2 ULN), low HBV DNA (< 2.8 107 copies/mL), and relatively preserved immune function (CD4 > 500). Thus, in the HIV-HBV coinfected individual with these characteristics and in whom HIV therapy is not indicated, pegylated interferon may be the preferred option because it spares both HAART toxicity and HIV resistance to antiretroviral medications.
Nucleoside and Nucleotide Analogues
Nucleoside and nucleotide analogues inhibit HBV replication by competing with the natural nucleoside triphosphates for incorporation into viral DNA. Lamivudine and adefovir are the approved nucleoside and nucleotide analogues, respectively, for the treatment of chronic hepatitis B.
Lamivudine, which also has activity against the HIV reverse transcriptase gene, was the first oral anti-HBV agent approved at a dose of 100 mg daily and has gained widespread use because of its ease of administration and its tolerability. In HIV-infected patients, lamivu-dine should be administered at 150 mg twice daily as a component of a HAART regimen in order to prevent the rapid emergence of HIV resistance. It is effective against HBeAg-positive and -negative disease; however, the treatment duration differs. For HBeAg-positive persons, treatment is for 6 months after HBeAg serocon-version or 1 year, whichever comes later. For HBeAgnegative persons, treatment is longer but the optimal duration of therapy is unknown. The HBeAg serocon-version rate in large studies is between 16 and 18% and correlates with ALT levels.[75,76] In the Asian trial, those with ALT > 5 x ULN had a 64% HBeAg seroconversion rate compared with 26 and 5% among those with an ALT > 2 x and < 2 x the ULN, respectively.[77] The durability of the response in HIV-uninfected patients varies between 63 and 80%75,78 and may be more likely if therapy is continued for more than 6 months after HBeAg seroconversion.[79] In those with HBeAg-negative disease, biochemical, histological, and virological responses occur, but the relapse rate is 90% on discontinuation of lamivudine.[80] Chronic dosing in this setting has not been adequately studied. Studies of HIV-HBV coinfected subjects show rates of HBeAg seroconversion of 22 to 28%81,82 that appear comparable to the HIV-uninfected patient.
As in the treatment of HIV, lamivudine therapy is limited by the selection of lamivudine-resistant HBV strains, which may develop more rapidly in subtype adw than in ayw.[83] As in HIV, the mutations usually occur in the YMDD motif of the catalytic domain of the polymerase gene and change the methionine at position 204 to either isoleucine or valine. [84]A recent report of primary infection with this mutant virus demonstrates the ability of it to be transmitted.[85] In persons uninfected by HIV, resistance increased from 14% at year 1 to 38, 49, and 66% at years 2, 3, and 4, respectively.[28] The development of resistance in HIV-HBV coinfected persons may be greater and is estimated at 20% per year.[86] Resistance may be predicted by the HBV DNA level 6 months after the initiation of treatment.[87]
The emergence of mutants is usually clinically detected by flares in liver disease, manifested by ALT elevations and reappearance of HBV DNA. If the mutation appears, lamivudine should be continued in the coinfected patient unless it is detrimental to effective HIV therapy because severe, even fatal, rebound hepatitis may occur on discontinuation of lamivudine in those who have detectable HBV DNA.[88] If lamivudine is discontinued, careful monitoring of ALT and HBV DNA is mandatory. Despite resistance, HBeAg seroconversion has been reported in 25% of patients who continue lamivudine after the appearance of the mutants,[89] but long-term lamivudine for isolated chronic hepatitis B after the development of resistance is a controversial area.
In summary, lamivudine is a potent anti-HBV drug, but it should not be used as a single agent in the coinfected patient because of the rapid emergence of HIV and HBV resistance mutations. Its use in the coinfected patient naive to therapy for both viruses will likely be in combination with either adefovir or tenofovir along with a potent antiretroviral regimen. Prior to starting a HIV-infected patient on lamivudine, one must be aware of the patient's HBV status in order to not limit future options for HBV therapy.
Adefovir is approved at a dose of 10 mg daily for chronic hepatitis B treatment and is active against lamivudine-resistant virus either alone or in combination with lamivudine.[90] After 48 weeks of therapy, HBeAg seroconversion occurred in 12% of patients[91] and seemed to be inversely correlated with hepatic levels of cccDNA.[92] Notably, adefovir appeared to decrease the hepatic cccDNA levels despite the general belief that nucleoside and nucleotide analogues do not decrease the pool of cccDNA.[92] HBV resistance mutations to adefovir are uncommon. A recent report demonstrated a novel mutation in the HBV polymerase (N236T) in 2 of 124 patients (1.6%) receiving 96 weeks of adefovir dipivoxil. This mutation reduced susceptibility to adefovir 5- to 23-fold in vitro, but there was no cross-resistance to lamivudine.[93] In a cohort of 32 HIV-HBV coinfected patients treated for 48 weeks with adefovir, the response rate is similar with an HBV DNA decline of -4 log10 copies/mL and HBeAg seroconversion in 2 patients.[93]
Tenofovir, a nucleotide analogue similar to adefovir, is not yet approved for the treatment of chronic hepatitis B, but data suggest that declines in HBV DNA are similar to those for adefovir in lamivudine-resistant and wild-type HBV.[95] For the HIV-HBV coinfected patient, its advantage is that it is also active against HIV.
In summary, adefovir and tenofovir are effective against both lamivudine-resistant and wild-type chronic hepatitis B, and resistance develops slowly. Thus, use of these agents as single agents should be done cautiously and is probably best reserved for the patient with lamivudine-resistant HBV who has evidence of moderate to severe fibrosis on liver biopsy. Adefovir could also be cautiously used in the patient with moderate to severe fibrosis who does not need HIV treatment and is not a candidate for interferon therapy because the dose of adefovir may be too low to exert mutation pressure on the HIV reverse transcriptase and thus may not select for adefovir-resistant HIV, but there are limited data to support this.[96]
Summary of Current Therapy Options
The treatment of chronic hepatitis B in the HIV-infected patient is complicated and needs to be individualized. Figure 1 should serve as a guide but is not intended to be exhaustive in its options. In considering the need for therapy, the HBeAg status is the first discriminatory point in the algorithm. Those who are inactive carriers (HBeAg negative with < 105 copies/mL of HBV DNA and normal ALTs) do not need treatment but should have periodic monitoring of ALT, aspartate aminotransferase, HBV DNA every 4 to 6 months and of AFP or ultra-sound, or both, for HCC. If ALTs become elevated for a sustained period of time, then a liver biopsy may be warranted. Those who do not have circulating HBeAg but have > 105 copies/mL of HBV DNA should be evaluated as if they are HBeAg positive.
The therapy options for the HBeAg positive can be grouped based on their need for HIV therapy. The most straightforward is the coinfected patient who needs treatment for both HIV and HBV and has not received therapy for either. Provided they have not been infected with a resistant virus, lamivudine and tenofovir should be active against both and provide a strong backbone to a HAART regimen. For the patient who does not need HIV treatment but has chronic hepatitis B, a liver biopsy is useful to assess the severity of the liver disease. If there is minimal liver disease, then close monitoring for liver disease and HCC is needed. If HBV therapy is needed, then pegylated interferon for 16 weeks is a good first option if the CD4 is > 500, the ALT is > 2 x ULN, and the HBV DNA is < 2.8 x 107 copies/mL. This option spares both HAART toxicity and selection of HIV-and HBV-resistant viruses. Those without these characteristics or who fail pegylated interferon can be treated with HAART, including lamivudine and tenofovir or potentially with adefovir alone. The most complicated situation is the HIV-infected patient who has been on lamivudine for years and has detectable HBV DNA signifying the presence of lamivudine-resistant HBV. If the patient is still on lamivudine, it should be continued, if possible, because discontinuation may be harmful. In this situation, a liver biopsy is preferable to assess the need for HBV therapy. If therapy is needed, adefovir or tenofovir are the favored options to add to the HAART regimen. Pegylated interferon is also an option if they have favorable characteristics; however, this has not been studied in lamivudine-resistant HBV. If therapy is not needed, then one should consider sparing adefovir and tenofovir for future combinations unless tenofovir is needed to treat the HIV disease.
Future Therapeutic Directions
Several other agents are in development but appear promising. Entecavir is active against the HBV polymerase and a 48-week course produces a -5.11 log HBV DNA decline against lamivudine-resistant HBV without evidence of resistance mutations.[97] (note from Jules Levin: phase III study results were reported at AASLD and reports are posted & archived in Conferences Section on NATAP website). Emtricitabine (FTC), a fluorinated derivative of lamivudine, has activity against HIV and HBV. A study of 96 weeks of FTC shows HBeAg loss of 51% and anti-HBe seroconversion in 29% of subjects. As expected, resistance develops but at a slower rate than to lamivudine.[98] Telbivudine (L-dT) is an L-nucleoside analogue that is very potent and appears to show greater efficacy in reducing HBV DNA levels than lamivudine does. [99]
Combination therapy for chronic hepatitis B is attractive because it has the potential for greater potency while preventing the development of resistance. The only combination that has been studied (although not extensively) is interferon-alfa and lamivudine, which does not appear to be substantially better than the individual medications alone.[100,101] However, it is anticipated that we are entering the era of combination nucleoside analogue therapy for the treatment of chronic hepatitis B. Those treating HIV-HBV coinfected patients may be at the forefront of this movement because the combination of tenofovir and lamivudine provides a potent combination against both viruses, especially in the patient who is naive to therapy.
Liver Transplantation
For end-stage liver disease, transplantation is an option. The HBV infection rate in the transplanted liver is very high but can be reduced by the posttransplant administration of hepatitis B immune globulin and lamivudine.[102] For the HIV-infected patient, liver transplantation is still experimental, so patients should be referred to centers with an active HIV transplant program.
Summary
Chronic hepatitis B is common in HIV-infected persons. Because HIV increases the morbidity and mortality from liver disease in persons with chronic hepatitis B, the hepatitis B status should be evaluated in all HIV-infected patients, and those at risk for acute hepatitis B should be vaccinated. The approved therapies, interferonalfa, lamivudine, and adefovir, have their advantages and disadvantages, so treatment of the coinfected patient involves consideration of both viruses and needs to be individualized. Because of the potential for lamivudine resistance, liver biopsy should be considered to support a decision to delay therapy. With several new antivirals active against HBV in clinical trials (some are also active against HIV), the next few years promise to be an exciting period for advances in therapy for the HIV-HBV coinfected patient. Randomized clinical trials for coin-fected patients with these new therapies in combination with existing therapies are needed to effectively treat this emerging opportunistic infection.
References
1. Mahoney FJ. Update on diagnosis, management, and prevention of hepatitis B virus infection. Clin Microbiol Rev 1999;12:351-366
2. Homann C, Krogsgaard K, Pedersen C, Andersson P, Nielsen JO. High incidence of hepatitis B infection and evolution of chronic hepatitis B infection in patients with advanced HIV infection. J Acquir Immune Defic Syndr 1991; 4:416-420
3. Rustgi VK, Hoofnagle JH, Gerin JL, et al. Hepatitis B virus infection in the acquired immunodeficiency syndrome. Ann Intern Med 1984;101:795-797
4. Centers for Disease Control and Prevention. 1999 USPHS/ IDSA guidelines for the prevention of opportunistic infections in persons infected with human immunodeficiency virus: disease-specific recommendations. USPHS/IDSA Prevention of Opportunistic Infections Working Group. US Public Health Services/Infectious Diseases Society of America. MMWR Morb Mortal Wkly Rept 1999;48:1-82
5. MacKellar DA, Valleroy LA, Secura GM, et al. Two decades after vaccine license: hepatitis B immunization and infection among young men who have sex with men. Am J Public Health 2001;91:965-971
6. Kao JH. Hepatitis B viral genotypes: clinical relevance and molecular characteristics. J Gastroenterol Hepatol 2002;17: 643-650
7. Kao JH, Chen PJ, Lai MY, Chen DS. Hepatitis B genotypes correlate with clinical outcomes in patients with chronic hepatitis B. Gastroenterology 2000;118:554-559
8. Hollinger FB. Hepatitis B virus. In: Knipe DM, Howley PM, et al, eds. Virology, 4th ed. Philadelphia: Lippincott Williams and Wilkins; 2001:2971-3036
9. Sakai T, Shiraki K, Inoue H, et al. HBV subtype as a marker of the clinical course of chronic HBV infection in Japanese patients. J Med Virol 2002;68:175-181
10. Coleman PJ, McQuillan GM, Moyer LA, Lambert SB, Margolis HS. Incidence of hepatitis B virus infection in the United States, 1976-1994: estimates from the National Health and Nutrition Examination Surveys. J Infect Dis 1998;178:954-959
11. McQuillan GM, Coleman PJ, Kruszon-Moran D, et al. Prevalence of hepatitis B virus infection in the United States: the National Health and Nutrition Examination Surveys, 1976 through 1994. Am J Public Health 1999;89:14-18
12. Kingsley LA, Rinaldo CR Jr, Lyter DW, et al. Sexual transmission efficiency of hepatitis B virus and human immunodeficiency virus among homosexual men. JAMA 1990;264: 230-234
13. Levine OS, Vlahov D, Koehler J, et al. Seroepidemiology of hepatitis B virus in a population of injecting drug users -- association with drug injection patterns. Am J Epidemiol 1995;142:331-341
14. Centers for Disease Control and Prevention. Recommendations for protection against viral hepatitis. MMWR Morb Mortal Wkly Rept 1985;34:313-335
15. Rehermann B, Ferrari C, Pasquinelli C, Chisari FV. The hepatitis B virus persists for decades after patients' recovery from acute viral hepatitis despite active maintenance of a cytotoxic T-lymphocyte response. Nat Med 1996;2:1104-1108
16. McMahon BJ, Holck P, Bulkow L, Snowball M. Serologic and clinical outcomes of 1536 Alaska natives chronically infected with hepatitis B virus. Ann Intern Med 2001;135: 759-768
17. Liaw YF, Tai DI, Chu CM, Chen TJ. The development of cirrhosis in patients with chronic type B hepatitis: a prospective study. Hepatology 1988;8:493-496
18. Fattovich G, Brollo L, Giustina G, et al. Natural history and prognostic factors for chronic hepatitis type B. Gut 1991;32: 294-298
19. Yang HI, Lu SN, Liaw YF, et al. Hepatitis B e antigen and the risk of hepatocellular carcinoma. N Engl J Med 2002; 347:168-174
20. Beasley RP, Hwang LY, Lin CC, Chien CS. Hepatocellular carcinoma and hepatitis B virus: a prospective study of 22707 men in Taiwan. Lancet 1981;2:1129-1133
21. Fattovich G, Rugge M, Brollo L, et al. Clinical, virologic and histologic outcome following seroconversion from HBeAg to anti-HBe in chronic hepatitis type B. Hepatology 1986;6:167-172
22. de Jongh FE, Janssen HL, De Man RA, et al. Survival and prognostic indicators in hepatitis B surface antigen-positive cirrhosis of the liver. Gastroenterology 1992;103:1630-1635
23. Davis GL, Hoofnagle JH, Waggoner JG. Spontaneous reactivation of chronic hepatitis B virus infection. Gastroenterology 1984;86:230-235
24. Torbenson M, Thomas DL. Occult hepatitis B. Lancet Infect.Dis. 2002;2:479-486
25. 2001 USPHS/IDSA guidelines for the prevention of opportunistic infections in persons infected with human immunodeficiency virus. HIV Clin Trials 2001;2:493-554
26. Hofer M, Joller-Jemelka HI, Grob PJ, Luthy R, Opravil M. Frequent chronic hepatitis B virus infection in HIV-infected patients positive for antibody to hepatitis B core antigen only. Swiss HIV Cohort Study. Eur J Clin Microbiol Infect Dis 1998;17:6-13
27. Silva AE, McMahon BJ, Parkinson AJ, et al. Hepatitis B virus DNA in persons with isolated antibody to hepatitis B core antigen who subsequently received hepatitis B vaccine. Clin Infect Dis 1998;26:895-897
28. Lok AS, McMahon BJ. Chronic hepatitis B. Hepatology 2001;34:1225-1241
29. Thio CL, Seaberg EC, Skolasky RL, et al. HIV-1, hepatitis B virus, and risk of liver-related mortality in the Multicenter AIDS Cohort Study (MACS). Lancet 2002;360:1921-1926
30. Colin JF, Cazals-Hatem D, Loriot MA, et al. Influence of human immunodeficiency virus infection on chronic hepatitis B in homosexual men. Hepatology 1999;29:1306- 1310
31. Gilson RJC, Hawkins AE, Beecham MR, et al. Interactions between HIV and hepatitis B virus in homosexual men: effects on the natural history of infection. AIDS 1997;11:597-606
32. Bodsworth NJ, Cooper DA, Donovan B. The influence of human immunodeficiency virus type 1 infection on the development of the hepatitis B carrier state. J Infect Dis 1991; 163:1138-1140
33. Vento S, Di Perri G, Garofano T, Concia E, Bassetti D. Re-activation of hepatitis B in AIDS. Lancet 1989;2:108-109
34. Lazizi Y, Grangeot-Keros L, Delfraissy JF, et al. Reappearance of hepatitis B virus in immune patients infected with the human immunodeficiency virus type 1. J Infect Dis 1988;158:666-667
35. Biggar RJ, Goedert JJ, Hoofnagle J. Accelerated loss of anti-body to hepatitis B surface antigen among immunodificient homosexual men infected with HIV. N Engl J Med 1987; 316:630
36. Davies SE, Portmann BC, O'Grady JG, et al. Hepatic histological findings after transplantation for chronic hepatitis B virus infection, including a unique pattern of fibrosing cholestatic hepatitis. Hepatology 1991;13:150-157
37. Fang JW, Wright TL, Lau JY. Fibrosing cholestatic hepatitis in patient with HIV and hepatitis B. Lancet 1993;342:1175
38. Scharschmidt BF, Held MJ, Hollander HH, et al. Hepatitis B in patients with HIV infection: relationship to AIDS and patient survival. Ann Intern Med 1992;117:837-838
39. Solomon RE, Van Raden M, Kaslow RA, et al. Association of hepatitis B surface antigen and core antibody with acquisition and manifestations of human immunodeficiency virus type 1 HIV-1) infection. Am J Public Health 1990;80:1475-1478
40. Eskild A, Magnus P, Petersen G, et al. Hepatitis B antibodies in HIV-infected homosexual men are associated with more rapid progression to AIDS. AIDS 1992;6:571-574
41. Ockenga J, Tillmann HL, Trautwein C, et al. Hepatitis B and C in HIV-infected patients. Prevalence and prognostic value. J Hepatol 1997;27:18-24
42. Saves M, Vandentorren S, Daucourt V, et al. Severe hepatic cytolysis: incidence and risk factors in patients treated by antiretroviral combinations. Aquitaine Cohort, France, 1996- 1998. Groupe d'Epidemiologie Clinique de Sida en Aquitaine (GECSA). AIDS 1999;13:F115-F121
43. Sulkowski MS, Thomas DL, Chaisson RE, Moore RD. Hepatotoxicity associated with antiretroviral therapy in adults infected with human immunodeficiency virus and the role of hepatitis C or B virus infection. JAMA 2000;283:74-80
44. Carr A, Cooper DA. Restoration of immunity to chronic hepatitis B infection in HIV-infected patient on protease inhibitor. Lancet 1997;349:995-996
45. Velasco M, Moran A, Tellez MJ. Resolution of chronic hepatitis B after ritonavir treatment in an HIV-infected patient. N Engl J Med 1999;340:1765-1766 (Letter)
46. Altfeld M, Rockstroh JK, Addo M, et al. Reactivation of hepatitis B in a long-term anti-HBs-positive patient with AIDS following lamivudine withdrawal. J Hepatol 1998;29: 306-309
47. Bessesen M, Ives D, Condreay L, Lawrence S, Sherman KE. Chronic active hepatitis B exacerbations in human immunodeficiency virus-infected patients following development of resistance to or withdrawal of lamivudine. Clin Infect Dis 1999;28:1032-1035
48. Manegold C, Hannoun C, Wywiol A, et al. Reactivation of hepatitis B virus replication accompanied by acute hepatitis in patients receiving highly active antiretroviral therapy. Clin Infect Dis 2001;32:144-148
49. Centers for Disease Control and Prevention. Sexually transmitted diseases treatment guidelines 2002. MMWR 2002; 51:63-66
50. Collier AC, Corey L, Murphy VL, Handsfield HH. Antibody to human immunodeficiency virus and suboptimal response to hepatitis B vaccination. Ann Intern Med 1988;109:101-105
51. Odaka N, Eldred L, Cohn S, et al. Comparative immunogenicity of plasma and recombinant hepatitis B virus vaccines in homosexual men. JAMA 1988;260:3635-3637
52. Bruguera M, Cremades M, Salinas R, et al. Impaired response to recombinant hepatitis B vaccine in HIV-infected persons. J Clin Gastroenterol 1992;14:27-30
53. Keet IP, van Doornum G, Safary A, Coutinho RA. Insufficient response to hepatitis B vaccination in HIV-positive homosexual men. AIDS 1992;6:509-510
54. Rey D, Krantz V, Partisani M, et al. Increasing the number of hepatitis B vaccine injections augments anti-HBs response rate in HIV-infected patients. Effects on HIV-1 viral load. Vaccine 2000;18:1161-1165
55. Are booster immunisations needed for lifelong hepatitis B immunity? European Consensus Group on Hepatitis B Immunity. Lancet 2000;355:561-565
56. Vento S, Garofano T, Renzini C, et al. Fulminant hepatitis associated with hepatitis A virus superinfection in patients with chronic hepatitis C. N Engl J Med 1998;338:286-290
57. Okoshi S, Igarashi M, Suda T, et al. Remote development of hepatocellular carcinoma in patients with liver cirrhosis type B serologically cured for HBs antigenemia with long-standing normalization of ALT values. Dig Dis Sci 2002;47:2002-2006
58. Higashi Y, Tada S, Miyase S, et al. Correlation of clinical characteristics with detection of hepatitis B virus X gene in liver tissue in HBsAg-negative, and HCV-negative hepato-cellular carcinoma patients. Liver 2002;22:374-379
59. Lopez VA, Bourne EJ, Lutz MW, Condreay LD. Assessment of the COBAS Amplicor HBV Monitor Test for quantitation of serum hepatitis B virus DNA levels. J Clin Microbiol 2002;40:1972-1976
60. Brook MG, Karayiannis P, Thomas HC. Which patients with chronic hepatitis B virus infection will respond to alpha-interferon therapy? A statistical analysis of predictive factors. Hepatology 1989;10:761-763
61. Perrillo RP, Schiff ER, Davis GL, et al. A randomized, controlled trial of interferon alfa-2b alone and after prednisone withdrawal for the treatment of chronic hepatitis B. N Engl J Med 1990;323:295-301
62. Perrillo R, Tamburro C, Regenstein F, et al. Low-dose, titratable interferon alfa in decompensated liver disease caused by chronic infection with hepatitis B virus. Gastroenterology 1995;109:908-916
63. Wong DK, Cheung AM, O'Rourke K, et al. Effect of alfainterferon treatment in patients with hepatitis B e antigen-positive chronic hepatitis B. Ann Intern Med 1993;119:312- 323
64. Krogsgaard K. The long-term effect of treatment with inter-feron-alpha 2a in chronic hepatitis B. The Long-Term Follow-up Investigator Group. The European Study Group on Viral Hepatitis (EUROHEP). Executive Team on Anti-Viral Treatment. J Viral Hepat 1998;5:389-397
65. Fattovich G, Giustina G, Realdi G, et al. Long-term out-come of hepatitis B e antigen-positive patients with compensated cirrhosis treated with interferon alfa. Hepatology 1997;26:1338-1342
66. Wong DK, Yim C, Naylor CD, et al. Interferon alfa treatment of chronic hepatitis B: randomized trial in a predominantly homosexual male population. Gastroenterology 1995; 108:165-171
67. McDonald JA, Caruso L, Karayiannis P, et al. Diminished responsiveness of male homosexual chronic hepatitis B virus carriers with HTLV-III antibodies to recombinant alpha-interferon. Hepatology 1987;7:719-723
68. Di Martino V, Thevenot T, Boyer N, Degos F, Marcellin P. Serum alanine transaminase level is a good predictor of response to interferon alfa therapy for chronic hepatitis B in human immunodeficiency virus-infected patients. Hepatology 2000;31:1030-1031
69. Di Martino V, Lunel F, Cadranel JF, et al. Long-term effects of interferon-alpha in five HIV-positive patients with chronic hepatitis B. J Viral Hepat 1996;3:253-260
70. Marcellin P, Boyer N, Colin JF, et al. Recombinant alpha interferon for chronic hepatitis B in anti-HIV positive patients receiving zidovudine. Gut 1993;34(Suppl 2):S106
71. Zylberberg H, Jiang J, Pialoux G, et al. Alpha-interferon for chronic active hepatitis B in human immunodeficiency virus-infected patients. Gastroenterol Clin Biol 1996;20: 968-971
72. Visco G, Alba L, Grisetti S, et al. Zidovudine plus inter-feron alfa-2b treatment in patients with HIV and chronic active viral hepatitis. Gut 1993;34(Suppl 2):S107-S108
73. Fried MW, Shiffman ML, Reddy KR, et al. Peginterferon alfa-2a plus ribavirin for chronic hepatitis C virus infection. N Engl J Med 2002;347:975-982
74. Cooksley WG, Piratvisuth T, Wang YJ, et al. 40 kDa peginterferon alfa-2A (PEGASYS): efficacy and safety results from a phase II, randomized actively controlled, multicenter study in the treatment of HBeAg positive chronic hepatitis B. Hepatology 2001;34:349A
75. Dienstag JL, Schiff ER, Wright TL, et al. Lamivudine as initial treatment for chronic hepatitis B in the United States. N Engl J Med 1999;341:1256-1263
76. Lai CL, Chien RN, Leung NWY, et al. A one-year trial of lamivudine for chronic hepatitis B. N Engl J Med 1998;339: 61-68
77. Chien RN, Liaw YF, Atkins M. Pretherapy alanine transaminase level as a determinant for hepatitis B e antigen sero-conversion during lamivudine therapy in patients with chronic hepatitis B. Asian Hepatitis Lamivudine Trial Group. Hepatology 1999;30:770-774
78. Song BC, Suh DJ, Lee HC, Chung YH, Lee YS. Hepatitis B e antigen seroconversion after lamivudine therapy is not durable in patients with chronic hepatitis B in Korea. Hepatology 2000;32:803-806
79. Pil HS, Kwang-Hyub H, Hoon AS, et al. Additional lamivudine therapy after HBeAg seroconversion would prolong the durability of response in HBV endemic area. Hepatology 2001;34:322A
80. Tassopoulos NC, Volpes R, Pastore G, et al. Efficacy of lamivu-dine in patients with hepatitis B e antigen-negative/hepatitis B virus DNA-positive (precore mutant) chronic hepatitis B. Lamivudine Precore Mutant Study Group. Hepatology 1999; 29:889-896
81. Benhamou Y, Katlama C, Lunel F, et al. Effects of lamivu-dine on replication of hepatitis B virus in HIV-infected men. Ann Intern Med 1996;125:705-712
82. Dore GJ, Cooper DA, Barrett C, et al. Dual efficacy of lamivudine treatment in human immunodeficiency virus/ hepatitis B virus-coinfected persons in a randomized, controlled study (CAESAR). The CAESAR Coordinating Committee. J Infect Dis 1999;180:607-613
83. Zollner B, Petersen J, Schroter M, et al. 20-fold increase in risk of lamivudine resistance in hepatitis B virus subtype adw. Lancet 2001;357:934-935
84. Stuyver LJ, Locarnini SA, Lok A, et al. Nomenclature for antiviral-resistant human hepatitis B virus mutations in the polymerase region. Hepatology 2001;33:751-757
85. Thibault V, Aubron-Olivier C, Agut H, Katlama C. Primary infection with a lamivudine-resistant hepatitis B virus. AIDS 2002;16:131-133
86. Benhamou Y, Bochet M, Thibault V, et al. Long-term incidence of hepatitis B virus resistance to lamivudine in human immunodeficiency virus-infected patients. Hepatology 1999; 30:1302-1306
87. Yuen MF, Sablon E, Hui CK, et al. Factors associated with hepatitis B virus DNA breakthrough in patients receiving prolonged lamivudine therapy. Hepatology 2001;34:785-791
88. Lim SG, Wai CT, Rajnakova A, Kajiji T, Guan R. Fatal hepatitis B reactivation following discontinuation of nucleoside analogues for chronic hepatitis B. Gut 2002;51:597-599
89. Liaw YF, Leung NW, Chang TT, et al. Effects of extended lamivudine therapy in Asian patients with chronic hepatitis B. Asia Hepatitis Lamivudine Study Group. Gastroenterology 2000;119:172-180
90. Peters M, Hann HW, Martin P, et al. Adefovir dipivoxil (ADV) alone and in combination with lamivudine (LAM) suppresses YMDD mutant hepatitis B virus replication: 48 week preliminary analysis. Hepatology 2002; 36:374A
91. Marcellin P, Chang TT, Lim SG, et al. Adefovir dipivoxil for the treatment of patients with HBe antigen-positive chronic hepatitis B. New Engl J Med 2003;348:808-816
92. Werle B, Wursthorn K, Petersen J, et al. Quantitative analyses of hepatic HBV cccDNA during the natural history of chronic hepatitis B and adefovir dipivoxil therapy: an international, multicenter study. Hepatology 2002;36:296A
93. Xiung S, Yang H, Westland C, et al. Resistance surveillance of HBeAg-chronic hepatitis B patients treated for two years with adefovir dipivoxil. 11 [th]International Symposium on Viral Hepatitis and Liver Disease, 2003, Abstract 779
94. Benhamou Y, Bochet M, Thibault V, et al. Safety and efficacy of adefovir dipivoxil in patients co-infected with HIV-1 and lamivudine-resistant hepatitis B virus: an open-label pilot study. Lancet 2001;358:718-723
95. Nunez M, Perez-Olmeda M, Diaz B, et al. Activity of tenofovir on hepatitis B virus replication in HIV-co-infected patients failing or partially responding to lamivudine. AIDS 2002;16:2352-2354
96. Delaugerre C, Marcelin AG, Thibault V, et al. Human immunodeficiency virus (HIV) Type 1 reverse transcriptase resistance mutations in hepatitis B virus (HBV)-HIV-coinfected patients treated for HBV chronic infection once daily with 10 milligrams of adefovir dipivoxil combined with lamivudine. Antimicrob Agents Chemother 2002;46:1586-1588
97. Chang TT, Hadziyannis S, Cianciara J, et al. Sustained viral load and ALT reduction following 48 weeks of entecavir treatment in subjects withchronic hepatitis B who have failed lamivudine. Hepatology 2002;36:300A
98. Gish R, Leung NW, Wang C, et al. Antiviral activity, safety, and incidence of resistance in chronically infected hepatitis B patients given once daily emtricitabine for 2 years. Hepatology. 2002;36: 372A
99. Lai CL, Leung NW, Teo EK, et al. International multicenter trial of LdT (telbividine) alone and in combination with lamivudine for chronic hepatitis B: an interim analysis. Hepatology 2002;36:301A
100. Barbaro G, Zechini F, Pellicelli AM, et al. Long-term efficacy of interferon alpha-2b and lamivudine in combination compared to lamivudine monotherapy in patients with chronic hepatitis B. An Italian multicenter, randomized trial. J Hepatol 2001;35:406-411
101. Schalm SW, Heathcote J, Cianciara J, et al. Lamivudine and alpha interferon combination treatment of patients with chronic hepatitis B infection: a randomised trial. Gut 2000; 46:562-568
102. Markowitz JS, Martin P, Conrad AJ, et al. Prophylaxis against hepatitis B recurrence following liver transplantation using combination lamivudine and hepatitis B immune globulin. Hepatology 1998;28:585-589
Reprint Address
Correspondence and reprint requests: Chloe L. Thio, M.D., 424 N Bond Street, Baltimore, MD 21231-1001. E-mail: cthio@jhmi.edu.
Abbreviation Notes
AASLD, American Association for the Study of Liver Diseases; AFP, alfa fetoprotein; ALT, alanine aminotransferase; anti-HBc, hepatitis B core antibody; anti-HBe, hepatitis B e antibody; anti-HBs, hepatitis B surface antibody; cccDNA, covalently closed circular DNA; HAART, highly active antiretroviral therapy; HBeAg, hepatitis B e antigen; HBsAg, hepatitis B surface antigen; HBV, hepatitis B virus; HCC, hepatocellular carcinoma; HCV, hepatitis C virus; IDU, injection drug use; ULN, upper limit of normal
|
|
| |
| |
|
|
|