 |
 |
 |
| |
Consensus Interferon: study results at AASLD
|
| |
| |
Reported by Jules Levin
AASLD
Nov 15, 2005, San Francisco
The efficacy and usefulness reported from studies of Consensus interferon are mounting, although small studies. Several studies were presented at AASLD Nov 2005 reporting impressive data. For several years running study data on the use of Consensus Interferon for peginterferon nonresponders has been presented at hepatitis conferences. The data has shown peginterferon/ribavirin nonresponders could achieve a sustained viral response with the use of high dose daily Consensus Interferon. The studies reported so far have been small nonrandomized & non-controlled & early studies. A large international phase III study is ongoing in nonresponders. But the data is becoming interesting & it appears worthy to consider Consensus Interferon therapy for peginterferon nonresponders. It does appear that the initial viral load decline with use of Consensus Interferon, also called phase I decline, which takes place within the first few days after starting therapy is significantly more potent. This may play a role in its efficacy. At this AASLD November 15, 2005 several studies were reported and again the results show Consensus Interferon performed well. Of note, one such study was conducted in genotype 1 treatment-naive patients and in combination with gamma interferon 71% and ribavirin achieved a sustained viral response. High dose daily Consensus Interferon may be associated with a higher degree of side effects so this should be considered. This is an initial report, I will followup this report with a more comprehensive one. I am in Dublin at the European AIDS Conference which limits my time, but I wanted readers to see this information on the utility of Consensus Interferon.
"Early Viral Response to Consensus Interferon plus Ribavirin Therapy In Patients who are Nonresponders or Relapsers to Prior Peg IFN Plus Ribavirin Therapy"
authors: Reem Ghalib, MD; Cheryl Levine, PhD, NP; Mariam Mouti, MD; Jeffrey Weinstein, MD; Armond Schwartz, MD; and Stephen Cheng, MD
The Liver Institute at Methodist Dallas, Texas
Study aim: To describe the early viral response to consensus interferon (CIFN) plus ribavirin combination therapy in patients who were nonresponders or relapsers to prior
treatment with PEG INF plus ribavirin therapy for chronic hepatitis C (HCV).
Study Design
Retrospective cohort analysis of chronic HCV patients in our clinic who are on treatment or have been treated with consensus interferon plus ribavirin therapy. This is an all-comers study including all patients of any genotype who are nonresponders or relapsers to prior Peg IFN plus RBV therapy and began treatment with consensus
interferon plus ribavirin.
Inclusion:
- Prior treatment with at least 12 weeks of an adequate
dose PEG IFN plus ribavirin therapy
- Began treatment with consensus interferon (15 mcg
per day) plus ribavirin therapy (weight based)
- Either discontinued treatment with consensus interferon plus ribavirin or completed at least 24 weeks therapy
Exclusion:
--Decompensated liver disease patients
The authors reported these RESULTS:
Conclusions
In nonresponders and relapsers to prior PEG IFN plus ribavirin therapy, the early virologic response rates (>2 log decrease) at week 12 with consensus interferon (CIFN) 15 mcg daily plus weight based ribavirin therapy are 67% and 95%, respectively.
The 24 week response rates (undetectable virus and >2 log decrease response rates, respectively) were 33% and 57% in the nonresponders and 79% and 89% in the relapsers. CIFN plus Ribavirin therapy was tolerated in this population with 71% continuing treatment beyond week 24 (60% in nonresponders and 89% in relapsers).
Further investigation of predictive factors of sustained viral response is needed in the nonresponder and relapser population to guide selection of appropriate treatment candidates and therapies.
Week 4 Evaluation (5 patients not tested):
HCV RNA negative (<5 IU/mL):
2 (6%) of prior nonresponders vs.
6 (32%) of prior relapsers (p = 0.045)
2 log reduction in HCV RNA:
9 (30%) of prior nonresponders vs.
14 (74%) of prior relapsers (p = 0.004)
Week 12 Evaluation:
HCV RNA negative (<5 IU/mL):
7 (23%) of prior nonresponders vs.
13 (67%) of prior relapsers (p = 0.005)
>2 log reduction in HCV RNA:
19 (63%) of prior nonresponders vs.
18 (95%) of prior relapsers (p = 0.033)
Week 24 Evaluation:
HCV RNA negative (<5 IU/mL):
10 (33%) of prior nonresponders vs.
15 (79%) of prior relapsers (p = 0.001)
>2 log reduction in HCV RNA:
17 (57%) of prior nonresponders vs.
17 (90%) of prior relapsers (p = 0.045)
Genotype 1 (ITT analysis)
After 24 weeks, 55% of nonresponders with genotype 1 had undetectable HCV RNA or >2 log decrease in viral load. About 25% had undetectable & about 30% had >2 log reductions.
DOSE MODIFICATIONS & DISCONTINUATIONS
16 patients (33%) made interferon dose modifications during weeks 2-24, 8% (4) for ANC & platelets, and 29% (14) for other side effects. 12 patients (24%) made ribavirin dose modifications for anemia. 14 patients (29%) discontinued treatment: 2 patients for viral rebound (4%), 5 patients (10%) for nonresponse at week 24, and 7 patients (14%) for adverse events.
35 patients (71%) continue treastment after week 24. 22 patients (45%) with undetectable HCV RNA (<10 IU/mL); 5 patients (10%) with HCV RNA <100 IU/mL; 4 patients (8%) with HCV RNA 100-150 IU/mL.
"Consensus Interferon in Combination with Gamma Interferon and / or Ribavirin leads to a significant Response in Treatment-naive Hepatitis C Genotype 1 Patients"
S. Kaiser, H. Hass and M. Gregor
University Hospital of Tuebingen, Germany
This study examined treatment of naive HCV patients with a bioengineered type 1 IFN (IFN-alfacon 1) and a recombinant type 2 IFN (IFN-g 1b).
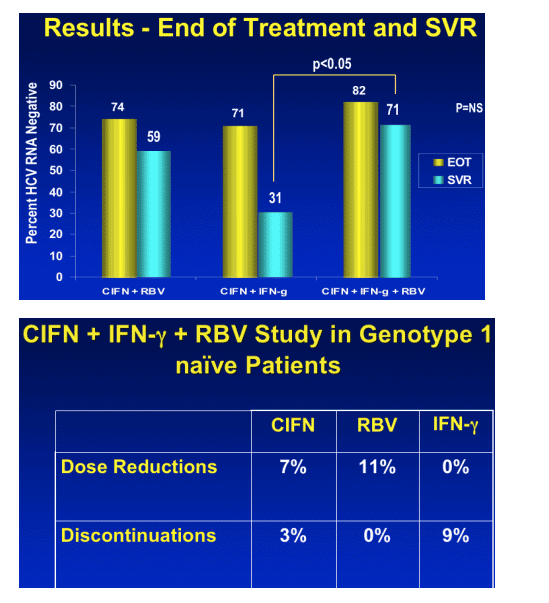
End of Treatment & SVR: CIFN+IFN-g + RBV, 82% EOT & 71% SVR
CIFN+RBV: 74% EOT, 59% SVR
CIFN+IFN-g: 71% EOT, 31% SVR
Safety Analysis
All patients have tolerated both drugs well with no discontinuations or dose reductions through week 12.
All patients had flu-like symptoms and fatigue typical of therapy with Interferons.
No patients had reductions in ANC that required growth factor support.
Patients with CIFN / Gamma IFN treatment had a significantly lower drop of Hemoglobin levels as compared to the other treatment arms.
All patients have tolerated both drugs well through week 12.
All patients had flu-like symptoms and fatigue typical of therapy with Interferons.
No patients had reductions in ANC that required growth factor support.
Patients with CIFN / IFN-g treatment had a significantly lower drop of Hemoglobin levels as compared to the other treatment arms.
CIFN + IFN-g + RBV Study in Genotype 1 naive Patients
Preliminary Conclusions by the study authors
Gamma Interferon has additive antiviral effects when being combined with interferon alfa and ribavirin therapy in HCV.
Triple therapy with alfa and gamma interefron together with ribavirin may be an emerging option for difficult-to-treat patients with HCV Genotype 1 and high viral load.
In special patients with intolerability to ribavirin combination therapy with alfa and gamma interferon may be an alternative therapeutic option.
|
|
| |
| |
|
 |
 |
|
|