 |
 |
 |
| |
Clevudine, new HBV Drug: 24 week study results
|
| |
| |
Reported by Jules Levin
AASLD Nov 2005, San Francisco
"A 24-week Clevudine monotherapy produced profound on-treatment viral suppression as well as sustained viral suppression and normalization of aminotransferase levels for 24 weeks off-treatment in HBeAg(+) chronic hepatitis B patients"
B.C. Yoo, Samsung Medical Center, Seoul, Korea; Pharmasset Inc, et al.
Author Summary
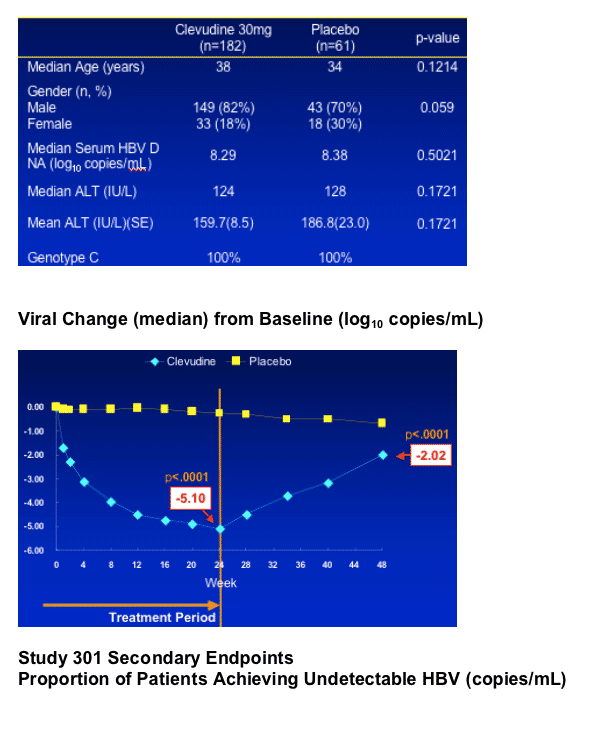
--potent anti-HBV activity: -5.10 log decline from baseline at week 24
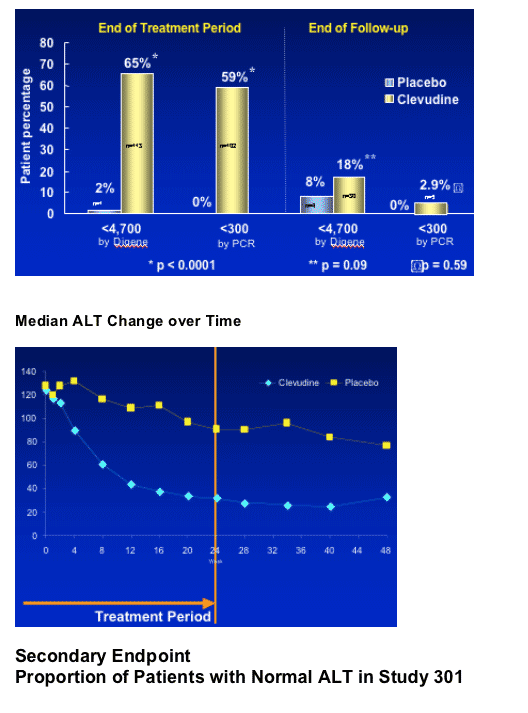
--good ALT normalization rate: 68% at week 24
--sustained viral suppression off therapy
--appears safe & tolerable
--suppression of cccDNA
--suppression of viral antigens
Of note there was a presentation of a study of clevudine in HBeAg negatives, which I will report & there was a poster studying the combination of FTC plus clevudine BUT a they used a low dose of clevudine (10mg/day) in that study. The dose is 30 mg once daily in this study.
Clevudine (L-FMAU)
- Pyrimidine nucleoside analogue, L- enantiomer
- Potent inhibitor of hepatitis B virus in vitro
(EC50 = 0.02 to 0.84 mM)
- Demonstrated antiviral activity in duck and woodchuck models of chronic hepatitis B infection
Pharmacologic Characteristics
1. Potent inhibition of HBV and EBV replication
-Inhibition of synthesis of ds-DNA from ss-DNA
-Suppression of CCC-DNA
2. No interference on human DNA polymerases
3. No cytotoxicity or mitochondrial toxicity
4. Rapid absorption, high bioavailability, long half life
5. Major route of elimination: renal excretion
A 12-Week Clevudine Therapy Showed Prolonged Antiviral Activities and Normalization of ALT Levels for 6-Months after Discontinuation of Treatment in Patients with Chronic Hepatitis B
L-FMAU-201
Hepatology 2004;40(S):652A
--Clevudine showed potent antiviral activities during therapy for 12 weeks.
--Clevudine characteristically induced sustained post-treatment antiviral effect after a 12-week treatment period.
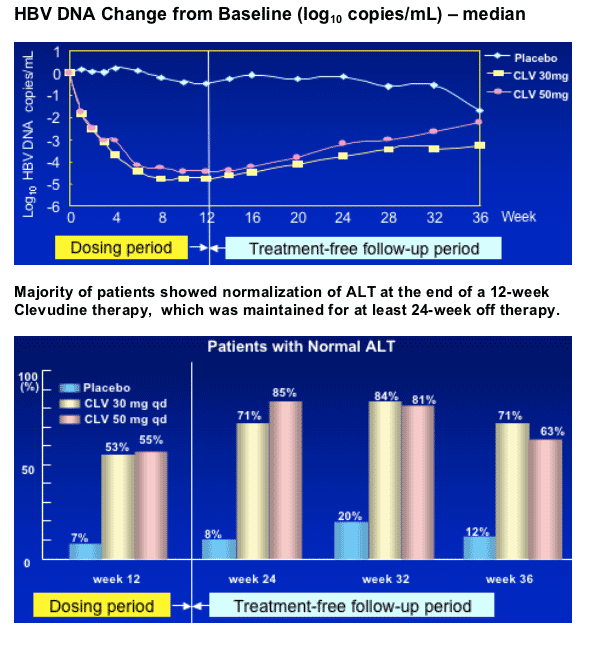
--A double-blind, randomized, parallel, placebo-controlled study in HBeAg(+) CHB patients
--Treatment with clevudine 30mg QD or Placebo for 24 weeks Clevudine 30 mg : Placebo = 3 : 1
--Follow-up for 24 weeks after cessation of dosing
--Multicenter study at 33 sites in Korea
Inclusion and Exclusion Criteria
Inclusion Criteria
- HBsAg positive for > 6 months
- HBeAg positive and anti-HBe negative
- HBV DNA ≥1 x 106 copies/mL
- 1.2 < ALT values < 15 x ULN at screening
Exclusion Criteria
- HIV- and HCV positive
- evidence of cirrhosis or hepatocellular carcinoma
- treatment with interferon within 6 months of screening
- previous treatment with any nucleoside analogue
Study Endpoints
Primary Endpoints: Antiviral activity - median change in log10 HBV DNA from baseline
Secondary Endpoints
Antiviral activity - proportion of patients with HBV DNA levels below
LOD of the assays
Digene HC II assay : <4,700 copies/mL
Amplicor PCR assay : <300 copies/mL
Biochemical improvement - normal ALT
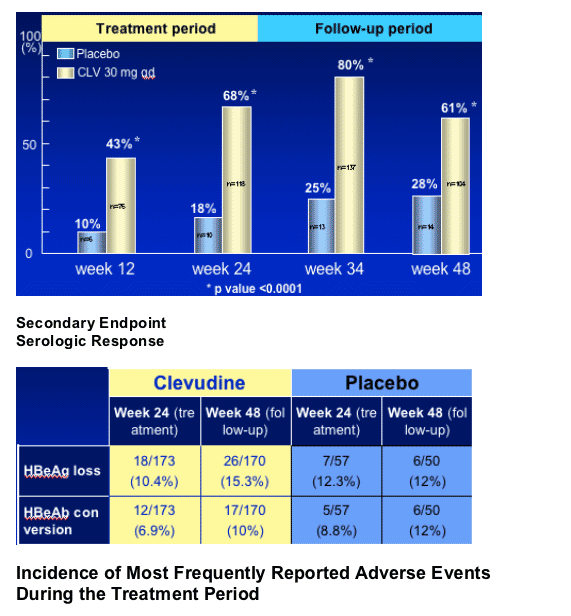
Serology - HBeAg loss and/or seroconversion
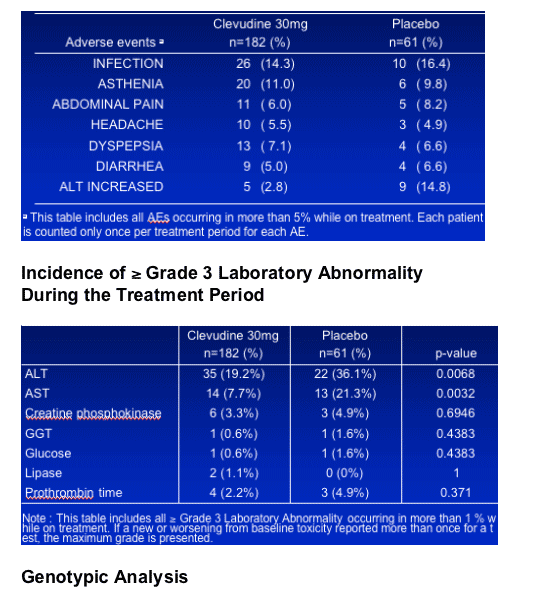
Nucleotide substitutions at 24 weeks
clevudine group : rtA181A/T (n=5), rtV191V/I (n=1) in the 5 patients
placebo group : rtL132M (n=1), rtA181S (n=1), rtV191V/I (n=2) in the 4 patients
Overall, none of the observed mutations was associated with a rebound in HBV DNA levels during the treatment period.
Author Summary
Clevudine 30mg QD for 24 weeks was well tolerated and showed potent antiviral activity in HBeAg(+) CHB patients.
Clevudine characteristically induced sustained viral suppression and normalization of ALT for 6 months after cessation of dosing
No emergence of resistant mutants during therapy
|
|
| |
| |
|
 |
 |
|
|