 |
 |
 |
| |
5 Years Adefovir in HBeAg-Negatives
|
| |
| |
"Long-Term Adefovir Dipivoxil Treatment Induces Regression of Liver Fibrosis in Patients
With HBeAg-Negative Chronic Hepatitis B: Results After 5 Years of Therapy"
Reported by Jules Levin
56th Annual Meeting of the American Association
for the Study of Liver Diseases
November 11-15, 2005
San Francisco, California, USA
Poster Number LB14
Summary:
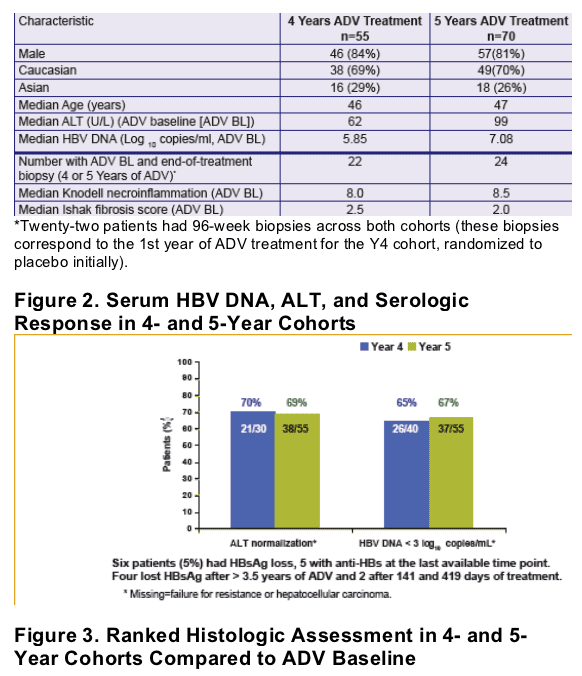
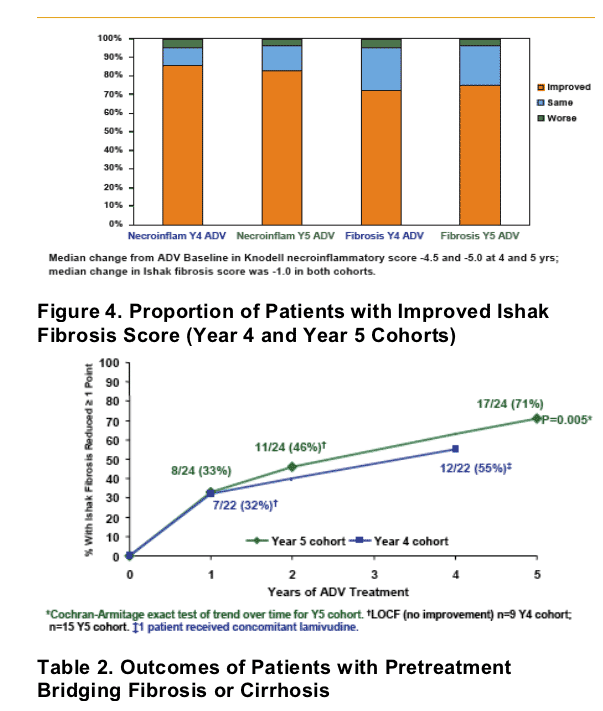
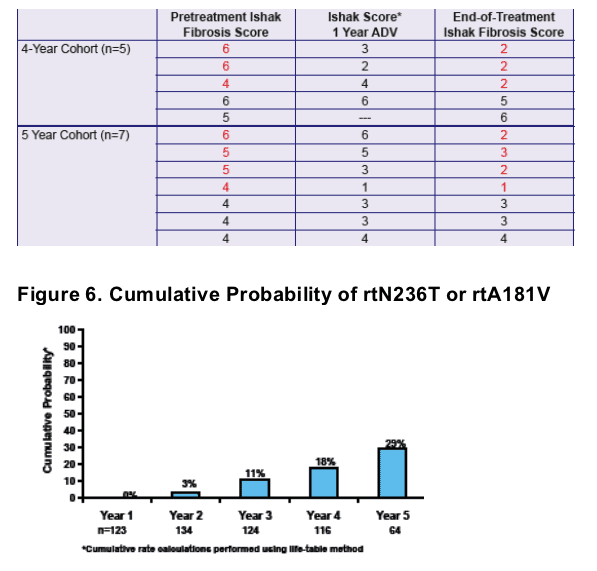
After 5 years of adefovir therapy, 71% of patients improved fibrosis, 65% reduced HBV-DNA <1,000 copies/ml, 69% normalized ALT, and the study reported 29% cumulative probability of drug resistance (rtN236T or rtA181V.
S Hadziyannis1, N Tassopoulos2, TT Chang3, J Heathcote4, G Kitis5, M Rizzetto6, P Marcellin7, SG Lim8, Z Goodman9,
S Arterburn10, J Ma10, S Xiong10, K Borroto-Esoda10, CL Brosgart10, and G Currie10
1Henry Dunant Hospital, Athens, Greece; 2Western Attica General Hospital, Athens, Greece; 3National Chen Kung Univ. Hospital, Tainan, Taiwan; 4Toronto Western Hospital, Toronto,
Canada; 5Georgios Papanikolaou District Hosp, Thessaloniki, Greece; 6Azienda Ospedaliera San Giovanni Battista, Torino, Italy; 7Hopital Beaujon, Clichy, France; 8National
University Hospital, Singapore; 9Armed Forces Institute of Pathology, Washington, DC; 10Gilead Sciences, Inc., Foster City, CA
Lamivudine is associated with histologic improvement and regression of
fibrosis in a majority of patients after 3 years of treatment.1 However, the
emergence of YMDD variants may limit or reverse histologic benefi t.2, 3
Adefovir dipivoxil produces sustained virologic suppression and histologic
improvement4 with delayed development of resistance during long-term
follow-up.
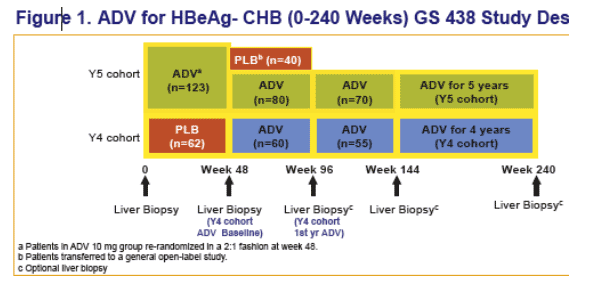
- This poster presents the first prospective, systematic set of biopsies from a trial of an oral antiviral following 5 years of treatment.
Background and Objectives
Adefovir dipivoxil (ADV) showed effi cacy and safety in HBeAg-negative
patients in a randomized, placebo-controlled trial over 96 weeks (GS-98-438)
Objectives for the 438 extension phase through 5 years were:
- To investigate the safety, tolerability, and durable effi cacy of 10 mg
ADV in patients with HBeAg- chronic hepatitis B (CHB)
- To fully characterize treatment-emergent resistance
Methods
Patients completing ADV in year 2 were offered 3 more years of open label ADV
- Effi cacy and safety of ADV were analyzed through end of treatment (Week 240) for both cohorts: Y4, n=55 enrolled; Y5, n=70 enrolled.
Assessments
- HBV DNA* and ALT every 12 weeks
- Liver biopsies at baseline (BL), 48 weeks, 96 weeks (optional), and end of study at Y4 or Y5 (optional).
- Annual genotypic analysis performed for all patients with HBV DNA > LLQ*
- All analyses were of observed data except histology (ITT last observation carried forward [LOCF] for missing data at 96 weeks only); 96-week biopsies were not performed in all patients and fi brosis was considered unimproved unless an actual fibrosis score was available.
* Roche Amplicor Monitor LLQ=1000 copies/mL performed through week 192 and Roche Cobas Taqman LLQ 169 copies/mL from week 192 through week 240.
Table 1. Pretreatment Characteristics for 4- and 5-Year ADV Cohorts
Safety Parameters
Renal Safety Parameters
- By Year 5, no patient had confi rmed serum phosphorus <2.0 mg/dL
during treatment
1 patient (<1%) had hypophosphatemia 3.4 months after discontinuing ADV
- Four patients (3%) had confi rmed increase in serum creatinine ≥0.5 mg/dL above Baseline
All absolute values were ≦ grade 1; maximum value 1.5 mg/dL, maximum increase 0.8 mg/dL (*unresolved)
- BL 0.5 mg/dL 1.1 mg/dL*
- BL 0.6 mg/dL 1.4 mg/dL*
- BL 0.7 mg/dL 1.3 mg/dL*
- BL 1.0 mg/dL 1.5 mg/dL (permanently discontinued with resolution)
Adverse events
- Three patients permanently discontinued due to an adverse event
- Serious adverse events occurred in 24 patients (19%)
- None were related to ADV
- 3 patients developed hepatocellular carcinoma
|
|
| |
| |
|
 |
 |
|
|