 |
 |
 |
| |
24 Week Pegasys + RBV for Genotype 1: SVR predicted by week 4 response
|
| |
| |
"Is shorter treatment with peginterferon alfa-2a (40KD) (PEGASYS) plus ribavirin (COPEGUS) possible in HCV genotype 1 'super-responders'?"
Preliminary results of a prospective randomized clinical trial
Reported by Jules Levin (see AASLD Coverage at http://www.natap.org)
Oral presentation at 56th AASLD Nov 2005, San Francisco
Peter Ferenci, MD
Medical University of Vienna, Vienna, Austria
On behalf of the Austrian Hepatitis Study Group
AUTHOR'S CONCLUSIONS
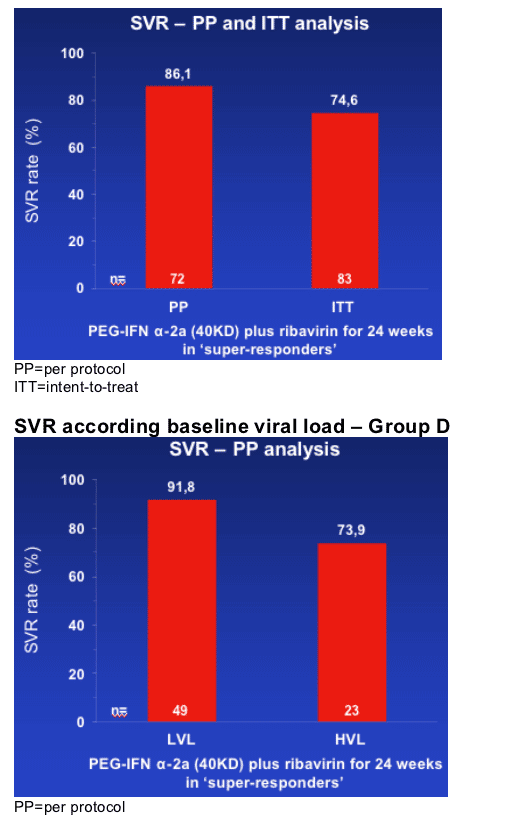
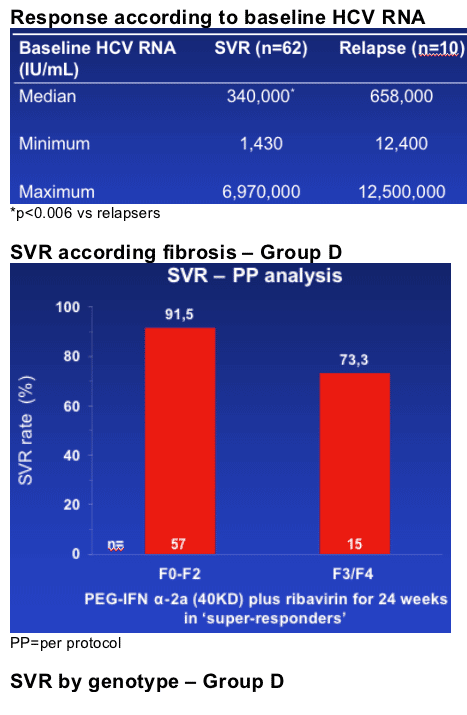
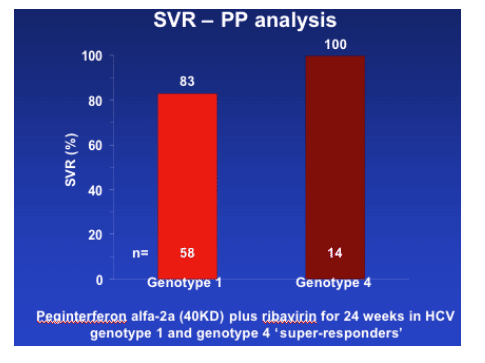
By ITT analysis, 75% of genotype 1 and 4 patients who had an RVR with peginterferon alfa-2a (40KD) (PEGASYS) plus ribavirin (COPEGUS) achieved an SVR following only 24 weeks' treatment
--SVR rates in genotype 1 and genotype 4 patients were 82% and 100% (PP analysis)
The 82% SVR rate in genotype 1 patients is greater than the highest SVR rate (52%; ITT) observed in genotype 1 patients in a multinational, randomized, phase III trial1 treated with the same regimen for 48 weeks, the current standard of care.
This analysis indicates that a treatment duration of only 24 weeks is effective in the 30% of genotype 1 and 4 patients who achieve an RVR. Thus, treatment duration might be best customized based on HCV RNA level at week 4.
Final results of prolonged therapy are awaited with interest
Background provided by Ferenci
The standard treatment of patients infected with difficult to treat HCV genotypes (1and 4) is a pegylated interferon plus ribavirin 1000/1200 mg/day for 48 weeks.
However, some genotype 1 patients may require only 24 weeks of treatment to achieve an SVR.
Patients who fail to achieve an EVR (early viral response) after 12 weeks of treatment with a pegylated interferon plus ribavirin are highly unlikely to achieve an SVR.
--Negative predictive value of 98% in genotype 1 patients treated with peginterferon alfa-2a (40KD) (PEGASYS) plus ribavirin (COPEGUS)
An RVR after 4 weeks of treatment is a strong predictor of response 88% of genotype 1 patients with an RVR achieved an SVR following 24 weeks' treatment with peginterferon alfa-2a (40KD) plus ribavirin 1000/1200 mg/day in a randomized, multinational phase III study (Jensen D et al, AASLD 2005, see poster report from me).
EVR = early virological response; HCV RNA negative or ≥2–log10 decrease at week 12
RVR = rapid virological response; HCV RNA negative at week 4
Main objective
Prospectively investigate the customization of combination therapy in patients infected with HCV genotypes 1 and 4 based on HCV RNA level at weeks 4 and 12
--In this interim analysis, SVR rates (PP, ITT) in patients treated for 24 weeks are presented
PP = per protocol
ITT = intent-to-treat
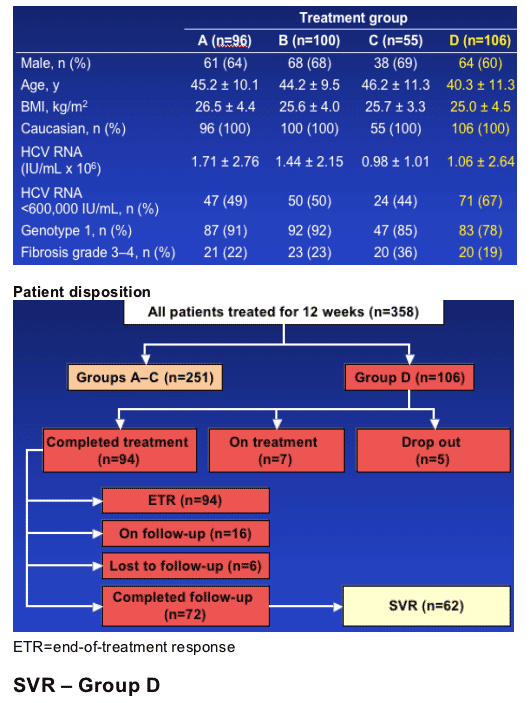
Patients
--Treatment naive
--Age 18–65 years
--Quantifiable HCV RNA (>600 IU/mL) in serum
--HCV genotype 1 or 4 infection
--Elevated serum ALT levels
--Liver biopsy within 6 months consistent with a diagnosis of chronic hepatitis C
--Compensated liver disease
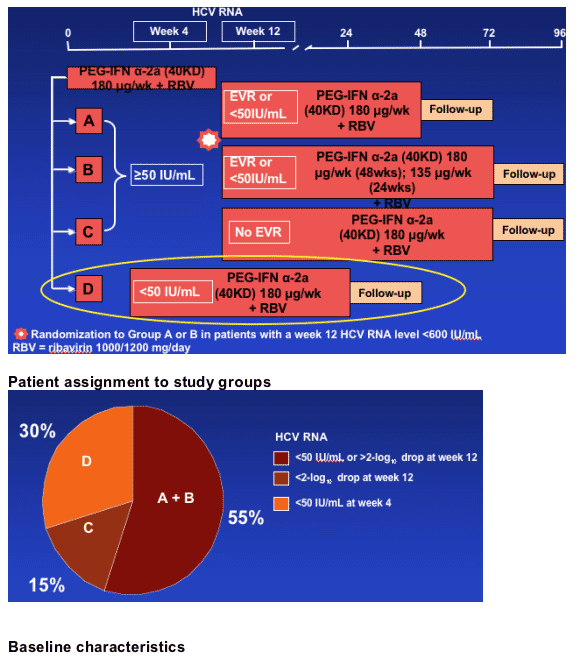
Study design
Patientsa received peginterferon alfa-2a (40KD) 180 _g/week plus ribavirin
1000/1200 mg/day
aTarget enrollment = 444 patients, currently recruited = 442 patients
Patients with undetectable HCV RNA at week 4 were assigned to treatment for 24 weeks (D group)
Patients with detectable HCV RNA at week 4 continued treatment and were re-tested at week 12:
--Patients with HCV RNA <600 IU/mL (or >2-log10 drop) were randomized to 48 or 72 weeks' treatment (A,B groups)
--Patients with HCV RNA >600 IU/mL (and <2-log10 drop) were treated for 72 weeks so long as HCV RNA was negative at week 24 (C groups)
|
|
| |
| |
|
 |
 |
|
|