 |
 |
 |
| |
Switch for Lipoatrophy to Abacavir or LPV/r+NVP
|
| |
| |
"Switching to a Thymidine Analog-Sparing or a Nucleoside-Sparing Regimen Improves Lipoatrophy: 24 Week Results of a Prospective Randomized Clinical Trial: AACTG A5110"
Rob Murphy reported these study results in an oral Late Breaker at CROI.
Murphy's CONCLUSIONS
--Switching d4T or ZDV to lopinavir/r + nevirapine, an NRTI-sparing regimen. is associated with significant improvement in SQ extremity fat at 24 weeks.
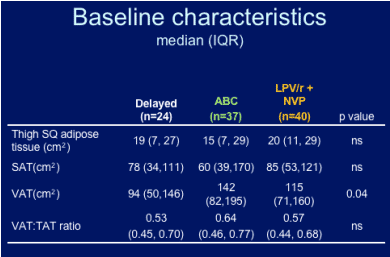
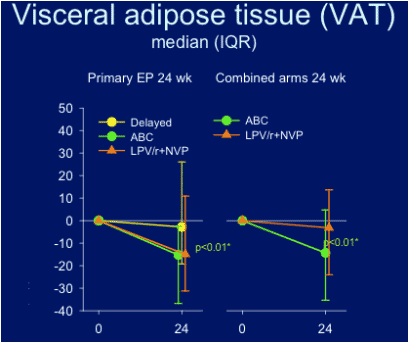
--Switching d4T or ZDV to abacavir, a non-thymidine analog, or lopinavir/r + nevirapine, an NRTI-sparing regimen, is associated with significant improvements in abdominal SAT, VAT and VAT:TAT; at week 24 there was not an improvement seen in thigh adipose fat tissue.
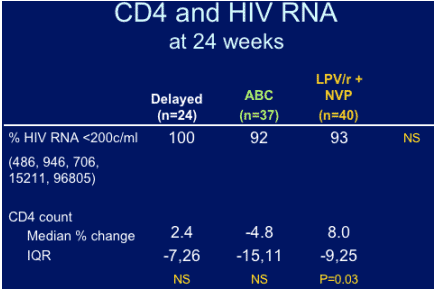
--Both interventions appear safe immunologically and virologically, the NRTI-sparing increased CD4 counts significantly
--Longer follow up is needed to better understand the long term implications of both interventions
--Central lipid, glucose, bone metabolism and quality of life studies and 48 week analyses are ongoing
Options for patients (patients) with peripheral lipoatrophy are limited. The MITOX and TARHEEL studies demonstrated that switching from a thymidine analog partially reversed lipoatrophy. ACTG 5125s showed that switching to a nucleoside reverse transcriptase inhibitor (NRTI) sparing regimen increases peripheral fat. We evaluated both interventional approaches and no treatment change.
The primary objective of this study were to investigate the effects of change in peripheral fat wasting by switching to a thymidine-sparing regimen and NRTI-sparing regimen. Patients had self-reported peripheral fat wasting including atrophy of the extremeties, since starting ARV therapy; fat wasting confirmed on a physical exam; they were on HAART including either AZT or d4T for >24 weeks; plasma HIV RNA was <500 copies/ml; CD4 count was >100; LABS: creatinine <3 x ULN. AST (SGOT) & ALT (SGPT) <5 x ULN, Lipase <1.5 x ULN.
The secondary study objectives were to investigate the effect of the treatment change on:
--subcutaneous and visceral fat tissue distribution in the abdomen
--safety & virologic activity
--glucose & lipid metabolism
--serum & urine markers of bone mineral metabolism
--quality of life
EXCULSION CRITERIA
Initiation of oral hypoglycemic agents,
Use of insulin, megestrol, rhGrowth hormone, supraphysiologic doses of corticosteroids, or hydroxyurea *
Initiation of androgen therapy*
Hyperthyroidism or hypothyroidism *
Systemic chemotherapy within 6 months
Pregnancy and breast-feeding
Drug abuse that could interfere with adherence to study
Serious illness
Current use of investigational agents
Documented or suspected acute hepatitis within 60 days of entry
Stable physiologic replacement was allowed
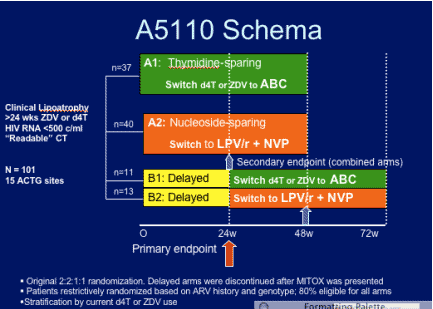
STUDY DESIGN
Patients at 15 ACTG sites receiving thymidine analogs stavudine (d4T) or zidovudine (ZDV) containing regimens with HIV RNA ≤ 500 copies/mL and clinical evidence of peripheral lipoatrophy were prospectively randomized to: switch thymidine analog to abacavir (ABC); discontinue all ART and switch to lopinavir/ritonavir (LPV/r) plus nevirapine (NVP), a regimen without NRTI; or delay switching for 24 weeks. Centrally analyzed single-slice CT of mid-thigh and abdominal fat, metabolic and virologic/ immunologic parameters were measured at baseline and week 24 as the primary endpoint. At week 24, the delayed switch group began their intervention. All patients were followed 48 weeks post-intervention.
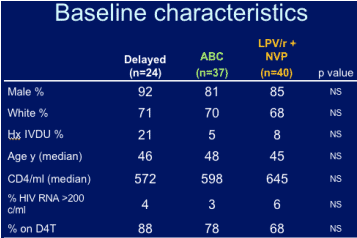
68% of patients on LPV/r+NVP were receiving d4T before the switch; 78% on abacavir were taking d4T before the switch; and 88% in the delayed switch arm were taking d4T before the switch.
| |
| |
| |
 |
|
| |
| |
PRIMARY STUDY ENDPOINT:
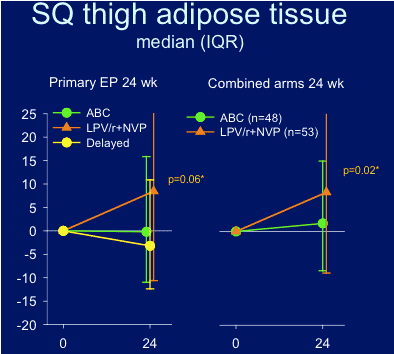
Change in thigh subcutaneous adipose tissue at 24 weeks for all 3 arms.
SECONDARY ENDPOINT:
--Change in thigh SQ adipose tissue after 24 weeks of intervention (combined arms)
--change in abdominal visceral adipose tissue (VAT), subcutaneous adipose tissue (SAT), glucose, lipid, bone, quality of life at 24 & 48 weeks
power: 50 patients per group, 80% to detect a 30% difference within arms.
BASELINE CHARACTERISTICS
| |
| |
| |
 |
|
| |
| |
 |
|
| |
| |
SQ thigh adipose tissue: did not improve at week 24 in abacavir arm, but did improve in LPV/r+NVP arm. This could be because more time is needed to see improvement or there could be variability in response between different body areas, or the thigh responds more slowly.
| |
| |
| |
 |
|
| |
| |
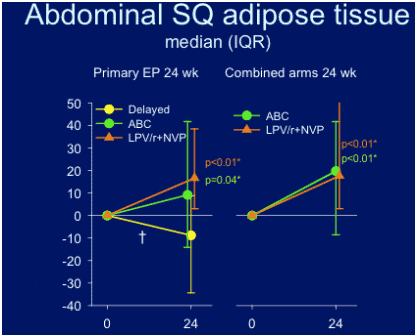
Abdominal SQ adipose tissue: improvement was seen for both groups, abacavir & LPV/r+NVP.
| |
| |
| |
 |
|
| |
| |
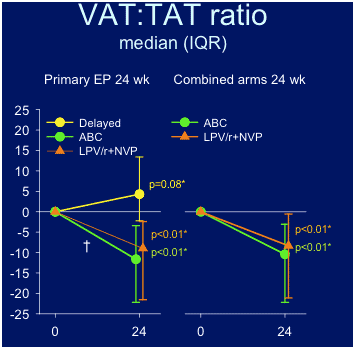
VAT:TAT ratio improved for both groups, abacavir & LPV/r+NVP.
| |
| |
| |
 |
|
| |
| |
 |
|
| |
| |
 |
|
| |
| |
 |
|
| |
| |
|
 |
 |
|
|