 |
 |
 |
| |
Switch to Unboosted Reyataz
|
| |
| |
Reported by Jules Levin
http://www.natap.org
EACS
Dublin, Nov 18, 2005
"Efficacy and Safety of Atazanavir (ATV)–Based HAART in Patients Switched From a Stable Boosted or Unboosted Protease Inhibitor (PI) Treatment
The SWAN Study: Final Results (48 Weeks)"
J. Gatell, D. Salmon-Ceron, A. Lazzarin, E. Van Wijngaerden, F. Antunes, C. Leen, A. Horban, V. Wirtz, L. Odeshoo, M. Van den Dungen, C. Gruber, and E. Ledesma, for the SWAN Study Group
The authors concluded: 48 weeks after a switch to Reyataz-based HAART regimen viral suppression was maintained, lipids were improved, and gastrointestinal adverse events were less in the Reyataz group compared to the Comparator PI group, those who remained on an unmodified Comparetor PI-containing HAART regimen. More details follow.
SUMMARY
Jose Gatell presented the study results in an oral session at Dublin. This study switched patients with <50 copies/ml to unboosted Reyataz with interesting results. 90% of the patients in the study were switched to unboosted Reyataz and 10% of patients received boosted Reyataz because they were also taking tenofovir in their regimen. This was a non-inferiority study. Patients who switched maintained their viral suppression through the 48 weeks of the study. Virologic rebound >50 c/ml by RTV use in prior regimen at screening (ITT-randomized): patients who were on boosted PI, 8% (12/150) had viral rebound after switch to ATV vs 11% (8/76) on Comparator PI (ns); patients who were on unboosted PI, 5% (71/128) had viral rebound >50 c/ml after switch to ATV vs 22% (14/65) for Comparator PI (p<0.001). The study authors concluded the efficacy analysis on treated subjects supported the non-inferiority of the ATV regimen.
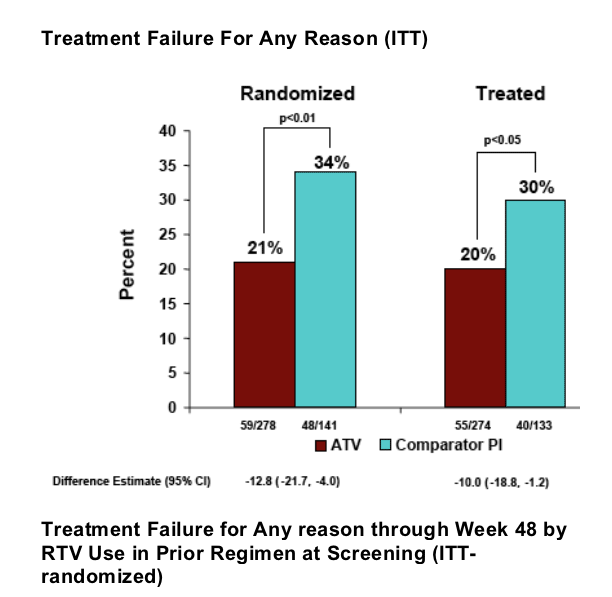
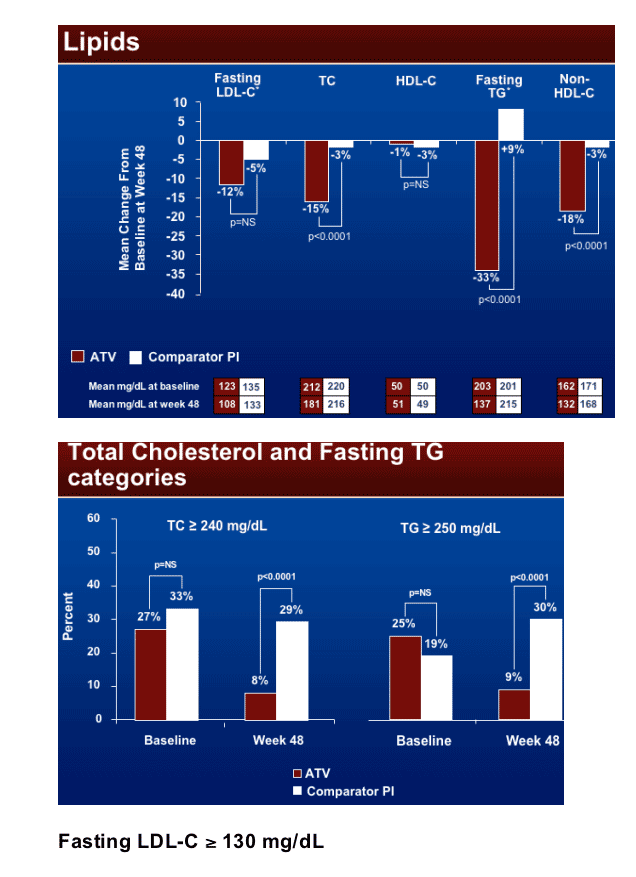
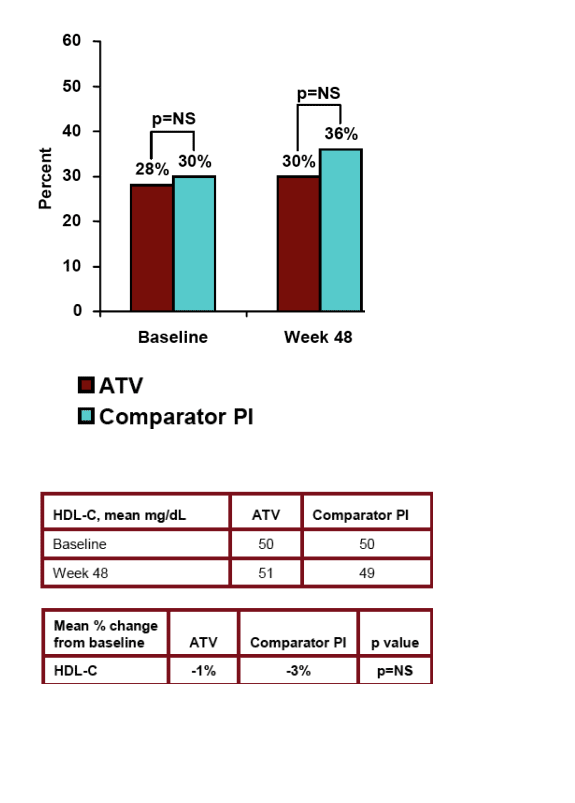
Treatment failure for any reason (ITT) was 21% (59/278) in the Reyataz group vs 34% (48/141) in the comparator PI group (p<0.01). Less diarrhea was experienced for patients who switched to ATV (2%) vs patients remaining on comparator PI (6%) (p<0.05). Lipid improvements were observed for patients who switched to Reyataz. Fasting LDL improved 12% for Reyataz vs 5% improvement in comparator arm (NS); total cholesterol improved by 15% in Reyataz group vs 3% in comparator PI arm (p<0.0001); HDL cholesterol declined by 1% in Reyataz group & 3% in comparator group (NS); fasting triglycerides improved by 33% in Reyataz group vs increased by 9% in comparator PI group (p<0.0001); non-HDL cholesterol improved by 18% in Reyataz group vs 3% in comparator PI group (p<0.0001).
The percent of patients with fasting triglycerides ≥250 mg/dl for the ATV group at baseline was 25% and after 48 weeks 9%, compared to 19% at baseline for Comparator PI group and 30% after 48 weeks. The mean fasting triglycerides level declined for the ATV group from 203 at baseline to 137 after 48 weeks, and for the Comparator PI group mean triglycerides was 201 at baseline and 215 after 48 weeks.
The percent of patients with total cholesterol >240 mg/dL improved for patients who switch to ATV but not for those who remained on Comparator PI: 27% of patients on ATV at baseline & 8% after 48 weeks had >240 mg/dL total cholesterol, compared to 33% of patients remaining on comparator Pi who had >240 total cholesterol at baseline & 29% after 48 weeks. Mean total cholesterol for the Reyataz group was 212 at baseline & 181 after 48 weeks, while for the Comparator PI group it was 220 at baseline & 218 after 48 weeks.
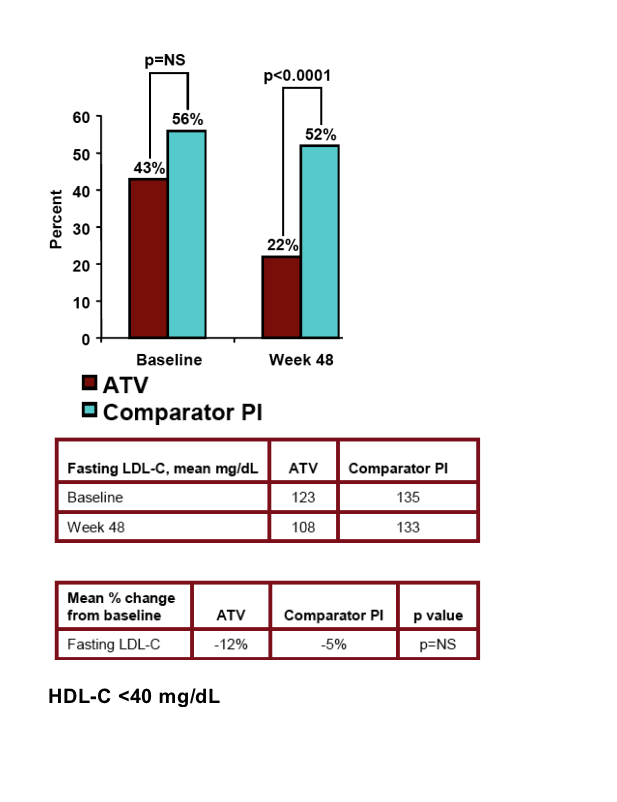
Similarly for fasting LDL-cholesterol, 43% of patients who switched to Reyataz had fasting LDL-C at baseline and 22% after 48 weeks had >240 mg/dl. For patients who remained on Comparator PI, 56% had fasting LDL-C at baseline but 52% still had fasting LDL-C after 48 weeks. Mean fasting LDL-C for patients who switched to Reyataz was 123 at baseline & 108 after 48 weeks. For patients remaining on Comparator PI it was 135 at baseline & 133 after 48 weeks.
Fasting LDL-C/HDL-C Ratio ≥3
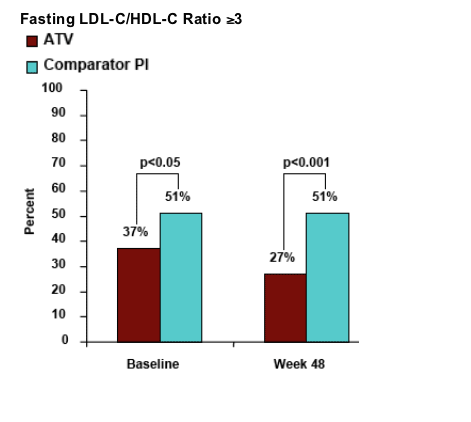
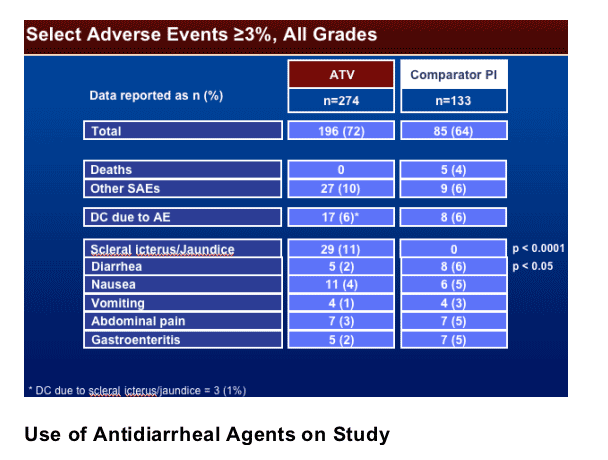
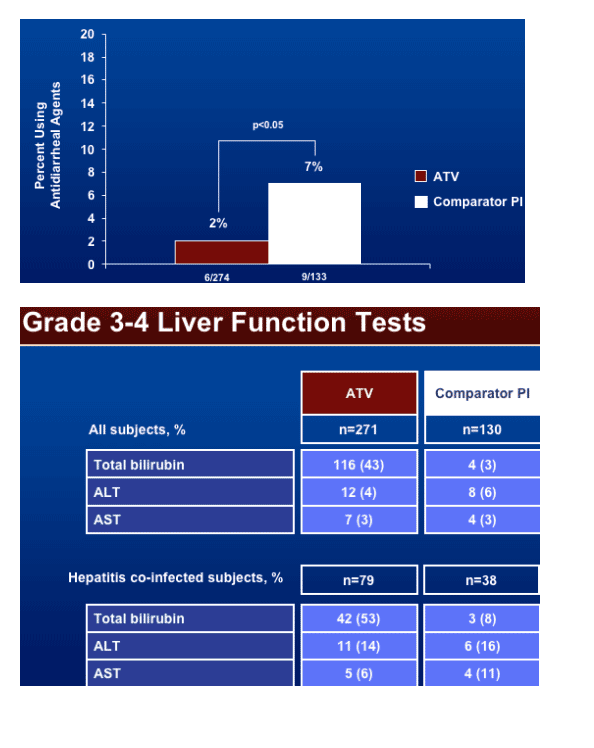
Use of antidiarrheal agents was 2% in Reyataz group vs 7% in comparator PI group (p<0.05). Grade 3-4 liver function tests values were similar for both study arms: ALT 4% in Reyataz vs 6% in comparator; among hepatitis coinfected patients: 14% in Reyataz group vs 16% in comparator PI group.
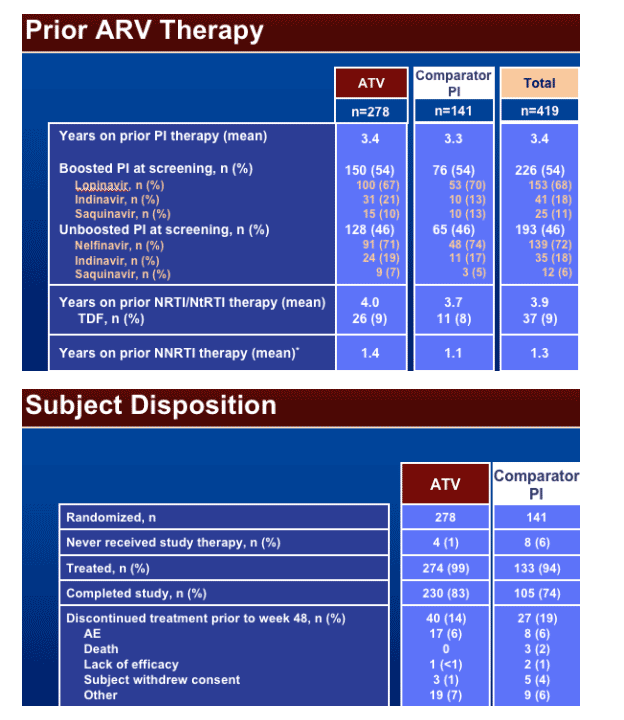
The mean time on therapy was 44 weeks for both regimens. Discontinuation of study therapy was 14% for ATV vs 19% for Comparator PI.
The switch to ATV (vs Comparator PI) was not associated with an increase in the discontinuation rates due to AEs (6% in both regimens), the incidence of SAEs (10% vs 6%), or deaths (0% vs 4%).
On-treatment emergent AEs of any grade related to gastrointestinal toxicity (diarrhea, nausea, vomiting, or abdominal pain) were higher on the Comparator PI regimen than in the subjects switching to ATV (13% vs 8%).
Three percent (9/271) of the subjects on the ATV regimen experiencing grade 3-4 bilirubin elevation also experienced grade 3-4 AST/ALT elevations.
Hyperbilirubinemia was reported predominantly on the ATV regimens, with 5 (2%) subjects with grade 3-4 jaundice and/or scleral icterus and 3 discontinuations (1%) due to jaundice and/or scleral icterus.
The use of lipid-lowering agents was observed to be higher on the Comparator PI regimen (19/133 [14%] vs 22/274 [8%], p=0.055).
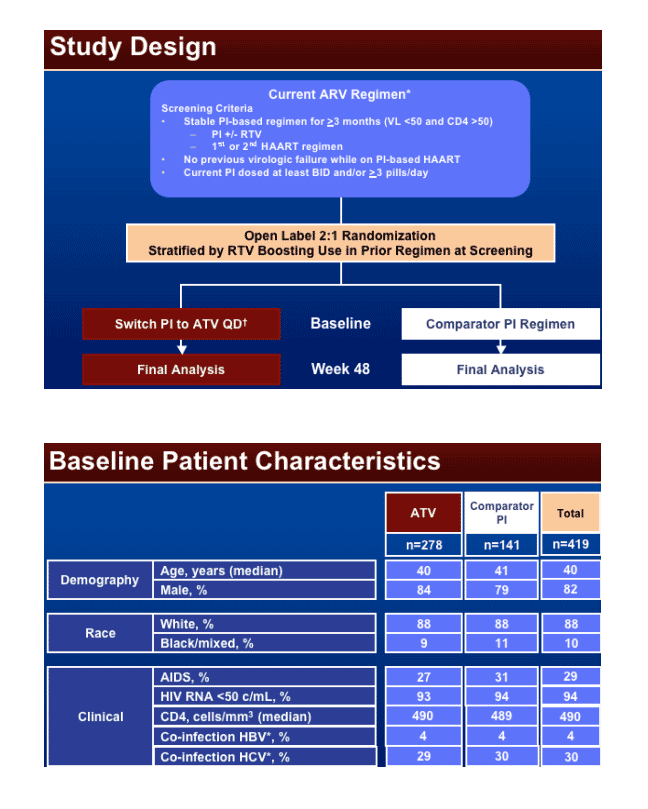
Atazanavir (ATV) is a once daily protease inhibitor studied in naïve & treatment experienced patients. The current trial was designed to evaluate the efficacy and safety of a switch to ATV (or ATV/ritonavir [RTV] if coadministered with tenofovir [TDF]) in HIV-infected patients experiencing virologic suppression but seeking regimen simplification. The primary study objective was to compare the proportion of subjects with virologic rebound ≥50 copies/ml through week 48 between subjects who switched to an ATV-containing HAART regimen and those who remained on an unmodified Comparator PI-Containing HAART regimen. The secondary objectives were to assess the following for each regimen: time to virologic rebound; magnitude of change from baseline in CD4 count through week 48; frequency and severity of clinical and laboratory adverse events (AEs), and discontinuations for AEs; change from baseline in lipids through week 48. Preliminary 24-week data from this 48-week trial have been previously presented and have suggested that a switch to ATV was generally well-tolerated and resulted in lower rates of virologic rebound ≥50 copies/ml.
Comparator protease inhibitors is taken with or without RTV at a dose of 200 mg or less + backbone. Admistration of Reyataz: ATV 400 mg QD (once daily + unchanged backbone. When TDF was part of the backbone, ATV/RTV 300/100 mg QD was used.
Statistical Design
– Powered to evaluate the equivalence (non-inferiority, _ 12%) of the primary endpoint for subjects switched to an ATV-containing regimen or those remaining on continued unmodified therapy.
A planned sample size of 372 subjects randomized in a 2:1 ratio provided at least 90% power to demonstrate the non-inferiority of the ATV regimen to the Comparator PI regimen, as assessed by the proportion of subjects with virologic rebound ≥50 copies/ml through week 48.
Statistical Analysis
Efficacy analysis were based on randomized subjects, and were stratified by RTV use in prior regimen at screening (boosted, unboosted PI):
--the primary endpoint was the proportion of subjects with virologic rebound (confirmed on-study HIV RNA ≥50 copies/ml or last on-study HIV RNA ≥50 copies/ml followed by discontinuation) through week 48. Treatment comparisons used the difference (ATV-Comparator PI) in proportions with 95% CIs and p values based on normal approximations.
--post-hoc analysis included the proportion of subjects with treatment failure for any reason (virologic rebound, never initiating study therapy or discontinuing of study drug) through week 48, and time to treatment failure.
Lipid analysis summarized values and percent changes from baseline at week 48 for treated subjects. Treatment comparisons used the difference in mean percent change (ATV-Comparator PI) with 95% CIs and p values based on t tests. Percent changes from baseline were computed on the log scale and back transformed. Data were excluded after the start of lipid-lowering therapy.
Safety analysis included data available through time of analysis for treated subjects. Serious AEs (SAEs) and deaths were included without regard to treatment status at time of onset for enrolled subjects.
RESULTS
Virologic Rebound ≥ 50 c/mL (ITT)
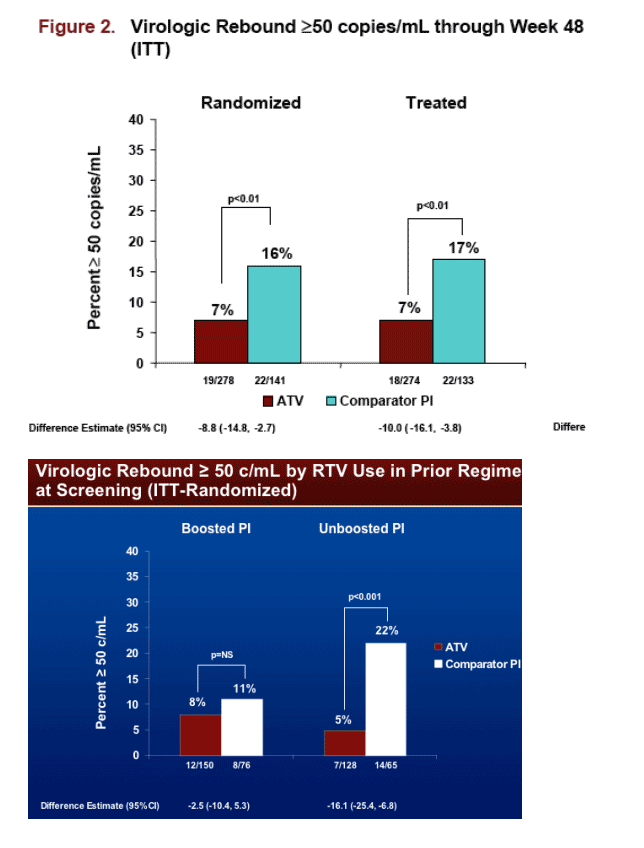
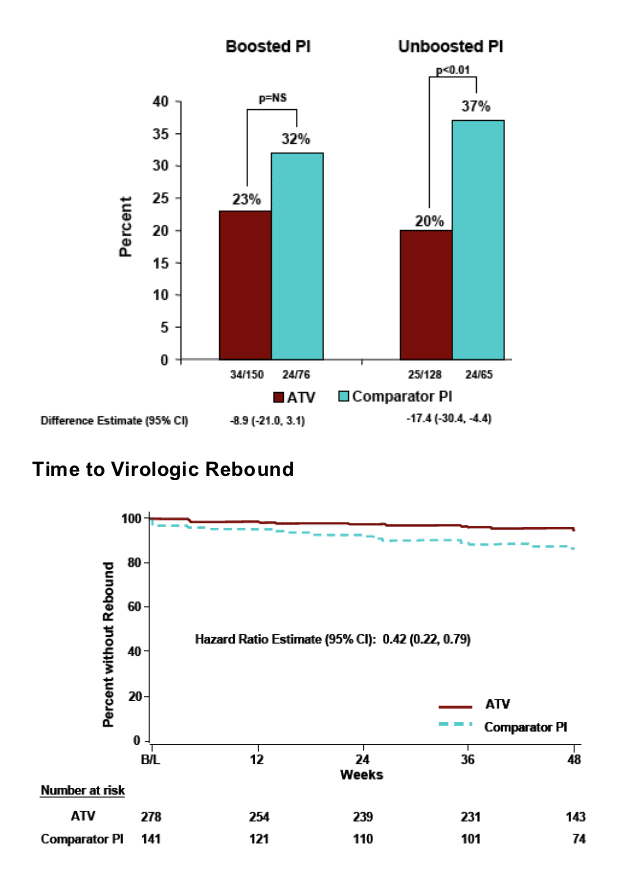
Hazard ratio estimates (95% CI) for time to virologic rebound (ATV:Comparator PI) in the boosted PI & unboosted PI strata were 0.71 (0.29, 1.78) and 0.25 (0.10, 0.63), respectively.
Hazard ratio estimates (95% CI) for time to treatment failure (ATV:Comparator PI) in the boosted PI and unboosted PI strata were 0.68 (0.40, 1.15) and 0.51 (0.29, 0.89), respectively.
Changes in CD4 counts were similar between treatment regimens with week 48 mean changes of +47 cells on the ATV regimen and +48 cells on the Comparator PI regimen.
|
|
| |
| |
|
 |
 |
|
|