 |
 |
 |
| |
Pretreatment Questions and First-Line Options
|
| |
| |
Written for NATAP. A Report from the 10th European AIDS Conference (EACS), November 17-20, 2005, Dublin
Mark Mascolini
Everyone knows guideline writers favor efavirenz or lopinavir/ritonavir in first-line combos. Is one better than the other? The EACS meeting offered the first randomized trial—with 52 weeks of follow-up—to proffer an answer. Other intriguing trials of people starting therapy compared efavirenz with indinavir/ritonavir and sized up lopinavir in people with a desperately low CD4 count. Two teams looked at portentous pretreatment questions involving transmitted resistant virus and readiness to start therapy.
Infection with drug-resistant virus
Getting infected with HIV is bad news; getting infected with antiretroviral-resistant HIV is worse. Just after the EACS meeting, British researchers reported that 14% of people diagnosed with HIV in the UK between February 1996 and May 2003—but still untreated—had drug-resistant virus [1]. In the last 2 years of the survey, the resistance rate stood at 19%.
But in other places rates of resistant virus transmission seem stable, or even on their way down. The Montreal area is one region with a waning transmission rate, reported McGill University's Jean-Pierre Routy [2]. His EACS study of 230 people with primary HIV infection also sorted out predictors of getting resistant HIV from a sex or needle partner.
The Montreal cohort was 93% male. While 60% picked up HIV during gay sex, 48% got the virus while shooting drugs, and 28% during straight sex. Routy spotted resistant virus in 19 people (8.3%). But transmission route did not affect the chance of getting resistant virus; nor did gender, age, ethnicity, education level, or annual income. This result matches CDC findings in the United States, where a six-state analysis showed that primary resistant virus favored neither gays nor straights, whites nor blacks, women nor men, youngsters nor oldsters [3]. Everyone shared an equivalent risk.
In Routy's Montreal group, multivariate analysis did pry loose two variables that upped the odds of infection with resistant virus:
- Infection after the year 2000 lowered the risk of resistance 69% (odds ratio 0.31, 95% confidence interval 0.11 to 0.89)
- A higher viral load halved the risk of resistance (odds ratio 0.54, 95% confidence interval 0.33 to 0.87).
Routy measured a median viral load of 4.12 log copies/mL in people infected with resistant virus compared with 4.75 in those with nonresistant virus (P = 0.003). Untreated people with resistant HIV may have lower viral loads because resistant virus tends to replicate worse than nonresistant HIV. Perhaps reflecting the lower loads with resistant virus, the median CD4 count also proved higher in the resistant group (610 cells/mL) than in the nonresistant group (410 cells/mL), a difference that approached statistical significance (P = 0.09).
Does delaying treatment affect clinical outcome?
Most clinicians recommend antiretrovirals when CD4 counts start falling through the 200s. But if a person continues to refuse treatment or seems unlikely to adhere to a regimen, will starting therapy anyway pay off?
That's the question posed by Rita Murri of Rome's Catholic University and colleagues in the ICoNA cohort [4]. To answer it they looked at 5243 untreated cohort members, asking their doctors why they delayed therapy. Then, among people who eventually did start treatment, they tracked CD4 and viral load responses according to reasons for the initial delay.
Clinicians reported six reasons for delaying treatment in the following proportions of the cohort:
- No guideline indication for treatment: 37.7%
- Recent HIV diagnosis: 36.2%
- First clinic visit for HIV: 11.8%
- Person rejected therapy: 10.6%
- Poor adherence suspected: 3.2%
- Clinical contraindication to therapy: 0.5%
People who rejected therapy had the lowest median CD4 count, 263 cells/mL (range 1 to 792 cells/mL), and the suspected poor adherers had the highest proportion of injecting drug users, 80.5%.
Among 2243 people who did start therapy, two groups had a significantly higher risk of virologic failure (defined as a viral load above 400 copies/mL) in an analysis adjusted for demographics, pretreatment CD4 count and viral load, the year treatment began, and the regimen used: Compared with people who didn't start because they were making their first office visit, those who initially rejected therapy (P = 0.04) and those suspected of poor adherence (P = 0.008) ran a higher risk of failure.
In a similar analysis the group that initially turned down treatment also had a slimmer chance of gaining at least 200 cells/mL with therapy, a difference that edged up to statistical significance (P = 0.06). But people suspected of poor adherence did not have a bigger risk of a poor CD4 response when compared with other groups.
A final analysis adjusted for the same variables determined that people who initially rebuffed therapy and those suspected of poor adherence had a higher risk of clinical progression after starting antiretrovirals when compared with the group who had not started treatment because they were making their first clinic visit (P = 0.04 and P = 0.02 respectively).
These findings will not surprise clinicians whose files are stuffed with poor treatment responses by people averse to antiretrovirals. Delaying treatment forever is not an option with the reluctant patient. But Murri's results emphasize the importance of doing everything possible to prepare such people for their antiretroviral odyssey and to support them closely once it begins.
What's better: efavirenz or lopinavir?
Current guidelines anoint efavirenz and lopinavir/ritonavir as preferred cornerstones of first-line therapy. But is one drug a stronger antiviral for people just starting treatment?
Results from the first randomized trial comparing these oft-prescribed drugs showed no difference in virologic response after 52 weeks [5]. Alas, this study is not the cleanest comparison possible because people taking efavirenz got 3TC/tenofovir while those taking lopinavir/ritonavir got AZT/3TC.
Two already published nonrandomized comparisons of efavirenz and lopinavir/ritonavir yielded differing results. Roberto Manfredi at the University of Bologna "Alma Mater Studiorum" compared RNA and CD4 changes in 51 people whose first regimen included lopinavir/ritonavir and 46 who started with efavirenz [6]. The two groups had comparable viral loads, but people starting lopinavir/ritonavir had significantly lower CD4 counts, and more of them had AIDS. After 10 to 33 month of follow-up, viral control proved equivalent with the two drugs, but the lopinavir/ritonavir group gained significantly more CD4 cells—and gained them faster—than the efavirenz group.
Carlo Torti at the University of Brescia confirmed a better CD4 response to lopinavir/ritonavir than to efavirenz in a nonrandomized comparison of 124 people starting those protease inhibitors (PIs) and 348 starting the nonnucleoside [7]. But after 48 weeks of treatment the virologic response proved significantly better with efavirenz in an intention-to-treat analysis.
Of course nonrandomized studies like these can never establish the superiority of one regimen over another. Too many factors that may affect results may also influence a physician's decision to prescribe one drug rather than another. So Torti and colleagues pitted efavirenz against lopinavir in a randomized trial that enrolled 174 people starting their first antiretrovirals [5].
Despite randomization, the lopinavir/ritonavir group had a significantly lower pretreatment CD4 count (172 cells/mL versus 213 cells/mL in the efavirenz group, P = 0.05). The PI group also included significantly fewer women than the efavirenz group (16% versus 31%, P = 0.04). But the groups didn't differ in starting viral load (about 4.8 log copies/mL) or other variables.
After 52 weeks of treatment an intention-to-treat analysis discerned no differences in virologic or CD4 response. Serious complications arose in six people in each treatment arm. Among those taking lopinavir/ritonavir with AZT/3TC, three people had diarrhea, two had anemia (an AZT side effect), and one had a new cancer diagnosis. The serious complications with efavirenz plus 3TC/tenofovir were two cases of depression, two of rash, and one each of fever and vomiting.
Abbott's Martin King offered a retrospective look at 654 people enrolled in four lopinavir/ritonavir trials to see if virologic response differed according to baseline CD4 count, starting with the abysmal bracket of fewer than 25 cells/mL [8]. Two trials assessed the twice-daily 400/100-mg dose of lopinavir/ritonavir, and two trials compared that dose with 800/200 mg once daily.
Using the noncompleter-equals-failure method to gauge sub-50-copy responses at 48 weeks, King determined that the proportion of sub-50 responders was equivalent for every baseline CD4 cluster assessed: less than 25, 25 to 49, 50 to 199, 200 to 349, 350 to 499, and 50 cells/mL or more. Baseline viral load did not affect the under-50 response rate in a similar analysis. Time to loss of virologic response also proved consistent regardless of pretreatment CD4 count or viral load.
Efavirenz vs boosted indinavir at low CD4s
Efavirenz got good marks in a randomized, open-label contest with indinavir/ritonavir for people starting their first regimen at a CD4 count under 100 cells/mL [9]. Boosted indinavir also did well in these people, but a high dose and a switch option in the trial design may have clouded results.
Jose Miro from the University of Barcelona and colleagues across Spain randomized 34 people to start standard-dose efavirenz plus two nucleosides and 31 to start indinavir/ritonavir plus two nukes. Some may consider the indinavir dose—800/200 mg twice daily—high by today's standards. But it probably looked reasonable when the trial began in November 2001.
The treatment groups had precisely the same median age (43 years) and median CD4 count (40 cells/mL) when the study started. Median pretreatment viral loads were high and equivalent in the two arms (5.6 log copies/mL with efavirenz and 5.4 log copies/mL with indinavir/ritonavir).
In a missing-data-equal-failure analysis, significantly more people taking efavirenz had a week-24 viral load below 200 copies/mL, one of the trial's primary endpoints (68% versus 42% with indinavir/ritonavir, P = 0.05). The PI arm suffered in this analysis because 10 people (32%) stopped indinavir/ritonavir with side effects, while only 4 (12%) gave up on efavirenz for this reason. The high PI dose may have raised the risk of side effects.
Four people taking efavirenz (12%) switched to another drug—3 to nevirapine and 1 to lopinavir/ritonavir. But 10 people starting with indinavir/ritonavir (32%) swapped the PIs for another antiretroviral—9 switching to lopinavir/ritonavir and 1 to efavirenz. Those tradeoffs may have influenced a curiously transient CD4 plateau observed in on-treatment analyses. In the table below note that CD4 gains in the PI arm stalled between months 12 and 18—just as the largest proportion of people switched to other drugs or dropped out. The 24-month analysis—focused on only the 10 people who tolerated indinavir/ritonavir best—shows an apparent surge in CD4 response. Even so, at no point did CD4 gains differ significantly between treatment arms.
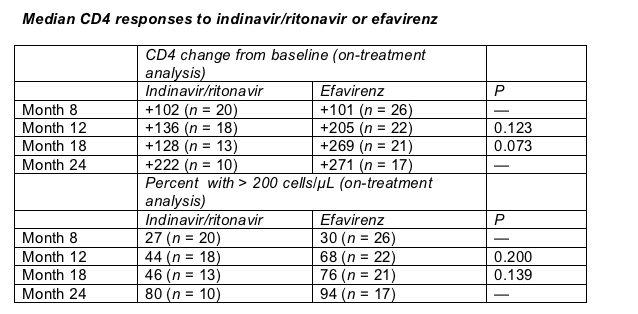
In a discussion after Miro's talk, EACS attendees stressed that the better viral load results recorded with efavirenz may apply only to indinavir/ritonavir, and maybe only to this lofty dose of indinavir/ritonavir.
What are the risks of once-daily PIs?
The profusion of once-daily PI options begs a question that researchers at the University of Liverpool and London's Chelsea and Westminster Hospital addressed in a retrospective survey of PI trough levels [10]: How often do troughs dip below the danger point with once-daily PIs?
Liverpool's Sara Gibbons toted troughs in people taking ritonavir-boosted once-daily doses of atazanavir (300 mg), fosamprenavir (1400 mg), lopinavir (800 mg), or saquinavir (1500 to 2000 mg) and in people taking boosted twice-daily doses of fosamprenavir (700 mg), lopinavir (400 mg), or saquinavir (1000 mg).
Gibbons found that only 6% of people taking atazanavir/ritonavir missed the target trough, compared with 30% to 50% of people taking once-daily fosamprenavir, lopinavir, or saquinavir (Table). Troughs proved significantly lower with once-daily dosing than with twice-daily dosing.
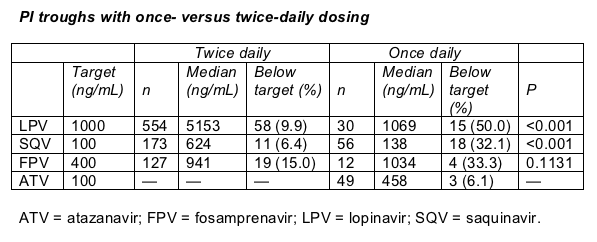
The lower once-daily troughs may reflect ritonavir's fading leverage over 24 hours, Gibbons and colleagues proposed. Such low PI levels become particularly risky if a person misses a dose. Missing a dose of a twice-daily regimen, in contrast, may leave a person some trough-level wiggle room.
Gibbons suggested these results mean "once-daily dosing may be more suitable for adherent patients with drug-sensitive virus."
Mark Mascolini writes about HIV infection (mailmark@ptd.net).
References
1. UK Group on Transmitted HIV Drug Resistance, David Dunn. Time trends in primary resistance to HIV drugs in the United Kingdom: multicentre observational study. BMJ Published online November 18, 2005. http://bmj.bmjjournals.com/cgi/rapidpdf/bmj.38665.534595.55v1
2. Routy JP, d'Edwardes MD, Rouleau D, et al. Factors predicting primary drug resistance and its impact on time to initiation of treatment in recently infected patients. 10th European AIDS Conference. November 17-20, 2005. Dublin. Abstract PE3.4/1.
3. Bennett D, McCormick L, Kline R, et al. U.S. surveillance of HIV drug resistance at diagnosis using HIV diagnostic sera. 12th Conference on Retroviruses and Opportunistic Infections. February 22-25, 2005. Boston. Abstract 674.
4. Murri R, Cozzi Lepri A, Vigevano G, et al. Reasons for delaying the starting of antiretroviral therapy show an association with clinical outcomes. 10th European AIDS Conference. November 17-20, 2005. Dublin. Abstract PS1/2.
5. Torti C, Quiros-Roldan E, Cologni G, et al. Tenofovir + lamivudine + efavirenz versus zidovudine + lamivudine + lopinavir/ritonavir in antiretroviral-naive HIV-infected patients: "ad interim" results of a prospective randomized study (SISTHER) of the Italian Master Cohort. 10th European AIDS Conference. November 17-20, 2005. Dublin. Abstract PE7.3/12.
6. Manfredi R, Calza L, Chiodo F. First-line efavirenz versus lopinavir-ritonavir-based highly active antiretroviral therapy for naive patients. AIDS 2004;18:2331-2333.
7. Torti C, Maggiolo F, Patroni A, et al. Exploratory analysis for the evaluation of lopinavir/ritonavir-versus efavirenz-based HAART regimens in antiretroviral-naive HIV-positive patients: results from the Italian MASTER Cohort. J Antimicrob Chemother 2005;56:190-195.
8. King M, Johnson M, Youle M, et al. Antiviral activity of lopinavir/ritonavir-based regimens in subjects with CD4 cell counts below 25 cells/mm3. 10th European AIDS Conference. November 17-20, 2005. Dublin. Abstract PE7.3/4.
9. Miro JM, Pick J, Plana M, et al. Immunological reconstitution in severely immunosuppressed antiretroviral-naive patients (<100 CD4+ cells/mm3) using a non-nucleoside reverse transcriptase inhibitor- or a boosted protease inhibitor-based antiretroviral therapy regimen: 96-week results (the Advanz trial). 10th European AIDS Conference. November 17-20, 2005. Dublin. Abstract PS1/4.
10. Gibbons S, Dickinson L, Boffito M, et al. Is once-daily dosing of PIs associated with decreased tolerance for missed or late doses? 10th European AIDS Conference. November 17-20, 2005. Dublin. Abstract PE4.1/8.
|
|
| |
| |
|
 |
 |
|
|