 |
 |
 |
| |
HBV Viral Load Risk Factor for Liver Cancer
|
| |
| |
Reported by Jules Levin
"ELEVATED SERUM LEVEL OF HEPATITIS B VIRUS DNA IS AN INDEPENDENT RISK FACTOR FOR HEPATOCELLULAR CARCINOMA: A LONG-TERM FOLLOW-UP STUDY IN TAIWAN"
C.J. Chen1, H.I. Yang1, J. Su2, C.L. Jen1, E. Kuo3, S.L. You1, U.H. Iloeje2
1 National Taiwan University, Taipei, Taiwan
2 Bristol-Myers Squibb Pharmaceutical Research Institute, Wallingford, USA
3 Bristol-Myers Squibb, Taipei, Taiwan
The long-term clinical goal of HBV treatment is to prevent or reverse cirrhosis, hepatic decompensation & liver cancer (improve event free survival). A number of studies at this 40th EASL meeting & published studies report that the greatest suppression of HBV DNA (viral suppression) is associated with better clinical outcomes. ALT normalization & HBeAg seroconversion are also associated with better clinical outcomes, but still HBV DNA viral suppression appears to be the best predictor of HBeAg seroconversion & ALT normalization. These markers of successful therapy all appear associated with improvement in liver histology, that isÑslowing or preventing disease progression, reducing risk for liver cancer, & improving survival; and event free survival in cirrhosis. Reducing viral load below 10,000 copies/ml has been shown in study to reduce risk for liver cancer. The study below was presented at the 40th EASL meeting & found that viral load <1000 copies/ml had the best effect in reducing risk for liver cancer. Yesterday I sent you a report from the 40th EASL of a study finding that a higher rate of histological improvement in liver disease in HBV was associated with HBV DNA <1000 copies/ml compared to HBV DNA >1000 copies/ml and ALT normalization <1.0x the upper limit of normal.
HBV DNA Decline & ALT Normalization Associated with Histologic Improvement
http://www.natap.org/2005/EASL/easl_21.htm
In the study below, HBeAg status was found to be associated with HBV DNA serum level (viral load): 28% of patients with HBV DNA <300 copies/ml were HBeAg-negative; 12% of patients with 300 to 1000 copies/ml were HBeAg-negative; 24% of patients with HBV DNA 1000 to 10,000 c/ml were HBeAg-negative; 19% with 10,000 to 100,000 c/ml were HBeAg-negative. After this level the of viral load the association was more stark: 9.4% of patients with 100,000 to 1 million c/ml were HBeAg-negative. 3% of patients with HBV DNA of 1 to 10 million were HBeAg-negative.
In a presentation at EASL this quote from Ana Lok was presented (New England journal of Medicine, 2002;346:1682-83): "Approximately 15-40% of infected patients will develop cirrhosis, liver failure, or liver cancer".
AUTHOR CONCLUSIONS: The incidence of HCC increases with increasing HBV DNA across a biological gradient of HBV DNA levels when compared with the group with undetectable HBV DNA. Elevated serum HBV DNA is a strong risk predictor for HCC, independent of HBeAg status, chronic HCV infection & elevated serum ALT level. Persistent elevation of serum HBV DNA level was associated with the greatest risk of HCC. Persistence was evaluated with baseline & followup testing. The majority of HCC cases were seen in people with HBV DNA >10,000 copies/ml. The absolute risk over the study period in this group was 7.5% (114/1515). In subjects with >100,000 copies/ml 10.7% developed HCC, and for subjects with HBV DNA >1 million 11.3% developed HCC.
The adjusted relative risk (RR adj) for developing liver cancer was;
1.0 (reference for patients with <300 (LOQ);
300 to 1000 copies/ml, 1.1 RR adj;
1.0-9.9x 104 (10,000 copies/ml), 2.4 RR adj;
1.0-9.9x 105 (100,000 copies/ml), 8.0 RR adj;
>=1.0x 106 (1 million copies/ml), 12.3 RR adj.
(adjusted for gender, age, habits of cigarette smoking, & alcohol consumption)
COX PROPORTIONAL HAZARDS HCC RISK PREDICTION MODEL
MULTIVARIATE -Adjusted Relative Risk (95% CI)
HBV DNA Level & risk for HCC (liver cancer)
Compared to patients with <10,000 copies/ml HBV DNA, patients with 10,000 to 100,000 HBV DNA had 2.5 (1.5 to 4.3) increased risk & patients with >100,000 copies/ml had 6.6 increased risk (4.1 to 10.1).
CJ Chen reported this study information in an oral session Friday afternoon at EASl April 2005.
The relationship between elevated HBV DNA serum levels and HCC (hepatocellular carcinoma).
Chronic HBV infection has been established as one of the most important risk factors for HCC.
Reduction in circulating HBV DNA level is a marker of efficacy for anti-viral treatment of chronic HBV infection.
However, there is little data on the prospective dose-response relationship between HBV DNA serum levels & HCC.
STUDY OBJECTIVES
To determine the independent effects of serum HBV DNA on the development of HCC adjusting for HBeAg status & serum ALT level.
To determine whether increasing levels of HBV DNA are associated with increasing risk of HCC across a biological gradient.
To determine if persistent elevation of HBV DNA is associated with the highest risk of developing HCC.
Methods
A cohort of 3,851 subjects seropositive for HBV surface antigen (HBsAg) was recruited from seven townships in Taiwan between 1991 and 1992. Serum samples obtained at enrollment and follow-up examinations were tested for HBsAg, HBeAg, HBV DNA by PCR, and serum ALT. The diagnosis of HCC was ascertained through data linkage with computerized profiles of the National Cancer Registry and Death Certification System in Taiwan. Multivariable adjusted relative risks (RRadj) were derived using Cox proportional hazard models.
15% were HBeAg+, 84% HBeAg-.
32% were age 30-39, 27% 40-48, 29% 50-69, 10% 60-65.
During 43,993 person-years of follow-up, 176 patients were newly diagnosed with HCC. After adjustment for gender, age, habits of cigarette smoking, and alcohol consumption, antibodies against hepatitis C virus, and HBeAg status, the risk of developing HCC was strongly associated with HBV DNA level in a dose-response relationship (P < 0.001). The adjusted relative risk was 6.6 (95%CI: 3.8-11.6) for those who had an elevated HBV DNA level (>10,000 copies/ml) compared with those who had an undetectable HBV DNA level (<300 copies/ml). For patients with >1 million the risk was 11.6. The risk was greater for patients with 10,000 to 100,000 copies/ml compared to <10,000. And the risk was greater for patients with >100,000 compared to those with 10,000 to 100,000.
COX Proportional Hazards HCC Risk Prediction Model
HBV DNA Level - Multivariate Adjusted Relstive Risk (95% CI)
<10,000 - 1.0
10,000 to 100,000 - 2.5 (p=0.01)
>100,000 6.4 (p=0.001)
After removing HCV coinfected patients from analysis the risks remain similar.
In people with ALT < 1 _ ULN, HBV DNA ³10,000 copies/ml had a higher risk of HCC compared to those with undetectable HBV DNA, adjusted relative risk 10.2 (95%CI: 5.7-18.4).
SUBGROUP ANALYSES: PERSISTENT HBC DNA ELEVATION & HCC RISK
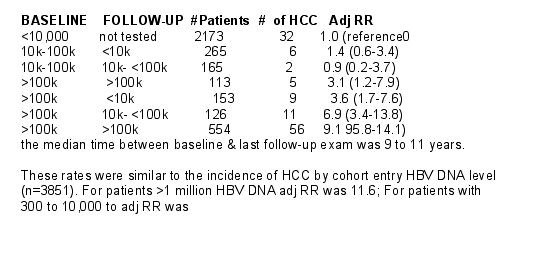
1,678 men & women had a serum HBV DNA level of >10,000 copies/ml at cohort entry. The serum samples collected at the last follow-up health examination or at the follow-up examination preceding the HCC diagnosis (for cases) were tested for serum HBV DNA level. 1,376 of 1,678 (82%) had serum samples collected at follow-up health exams available for HBV DNA testing. Samples from subjects with entry HBV DNA <10,000 were not re-tested; this wasthe referencegroup.
In people with high DNA at baseline, the HCC risk was higher with persistent elevation, compared to those clearing DNA on repeat sampling adjusted relative risk 6.4 (95%CI: 4.4-9.5).
|
|
| |
| |
| |
|
 |
 |
|
|