 |
 |
 |
| |
HBV DNA (viral load) Predicts Liver Cirrhosis
|
| |
| |
Reported by Jules Levin
These two studies are among several presented at the 40th annual EASL meeting (April 2005, Paris) finding that HBV DNA levels (viral load) are associated with disease progression, that reducing viral load (virus replication) can reduce the risk for disease progression to cirrhosis, liver cancer & death. The incidence of liver cirrhosis in HBV increases as HBV viral load increases. The data from these studies suggest reducing viral load as low as possible to <300 copies/ml may be of benefit compared to a level of 10,000 copies/ml or perhaps to 1,000 copies/ml although this may be more debatable. Other studies at the meeting found that the incidence of liver cancer also increased as the HBV viral load increased. The strong suggestion from these studies is that reducing HBV viral load reduces risk for serious disease in HBV. Of note, in HCV disease viral load has not been found to be associated with disease progression: you could have liver cirrhosis & a low HCV viral load and conversely you could have a high HCV viral load (HCV RNA) & not have cirrhosis.
Some questions are: when to begin HBV therapy since therapy can be ongoing & not of time limited duration as in HCV; should peginterferon be the initial attempt with therapy since peginterferon therapy is time limited; should combination therapy be used: the potential benefits of combination therapy have not yet been well characterized; the only benefit so for identified with combination therapy is that it appears to reduce the development of drug resistance. Idenix Pharmaceuticals has reported data in the woodchuck model that there two HBV drugs (LdC & LdT) in combination reduce viral load additively, but clinical studies will have to confirm this finding. Of note, the authors of the first study showed data on incidence of cirrhosis in patients with undetectable HBV DNA & concluded that "even the absence of HBV DNA does not eliminate the risk of future cirrhosis. The second study below finds that these findings apply to both HBeAg negative & positive patients.
"SERUM HEPATITIS B VIRUS DNA LEVEL PREDICTS THE INCIDENCE OF LIVER CIRRHOSIS IN PERSONS CHRONICALLY INFECTED WITH HBV"
U.H. Iloeje1, H.I. Yang2, J. Su1, C.L. Jen2, E. Kuo3, S.L. You2, C.J. Chen2
1 Bristol-Myers Squibb Pharmaceutical Research Institute, Wallingford, CT, USA
2 National Taiwan University, Taipei, Taiwan
3 Bristol-Myers Squibb, Taipei, Taiwan
Chronic hepatitis B patients are at increased risk of developing liver cirrhosis. Cirrhosis develops as a result of hepatic inflammation and subsequent fibrosis. Ongoing viral replication is associated with hepatic inflammatory activity and suppression of viral replication in clinical trials is associated with improvements in liver histology. An association between elevated HBV DNA level and progression to cirrhosis has not been shown in a large epidemiological setting. This study was carried out to examine if increasing levels of HBV DNA is associated with increasing risk of cirrhosis.
A population based prospective cohort study of 3851 HBV infected subjects was established from seven townships in Taiwan between 1991 and 1992. Cohort entry serum samples were tested for HBV DNA by PCR, and the diagnosis of cirrhosis was by ultrasonography and ascertained by medical records review. Cirrhosis incidence rate per person year of follow-up (PYFU) for each strata of HBV DNA was calculated. The multivariable adjusted relative risk (RRadj) was derived from Cox's proportional hazard models.
Results
Of the 3851 subjects, 77 subjects were excluded (75 diagnosed with cirrhosis and 2 dying within 6 months of enrollment) leaving 3774 subjects. During 42,114.9 person-years of follow-up, 395 cirrhosis cases were newly diagnosed.
The incidence rate of cirrhosis ranged from 386.1/100,000 PYFU for those with HBV DNA <300 copies/ml to 2575.7/100,000 PYFU for those with HBV DNA ³106 copies/ml (test of trend: p < 0.0001) in a dose dependent manner.
With the undetectable group as reference, and adjusting for gender, age, habits of cigarette smoking, and alcohol consumption, and antibodies against hepatitis C virus, the risk of cirrhosis started increasing at HBV DNA level of 104 (10,000 copies/ml), RRadj 2.4 (95%CI: 1.6-3.5), and was 9.3 (95%CI: 6.5-13.1) for those who had serum HBV DNA level >106 copies/ml (1 million copies/ml).
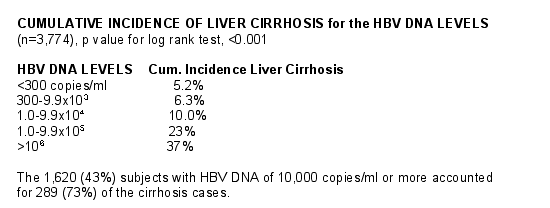
The 1,620 (43%) subjects with HBV DNA of 10,000 copies/ml or more accounted for 289 (73%) of the cirrhosis cases.
The incidence of cirrhosis even in the subjects with undetectable HBV DNA was 386.1 per 100,000 person-years, showing that even the absence of dectetable HBV DNA does not eliminate the risk of future cirrhosis.
Author Conclusions
HBV DNA level is a strong predictor of cirrhosis risk. The incidence of cirrhosis increases with HBV DNA level in a dose dependent manner. It is expected therefore that reducing the HBV DNA should conversely decrease cirrhosis risk in chronic hepatitis B patients.
2nd Study
"VIRAL LOAD IS A STRONG PREDICTOR OF LIVER CIRRHOSIS RISK IN PEOPLE CHRONICALLY INFECTED WITH HEPATITIS B VIRUS REGARDLESS OF HEPATITIS B e ANTIGEN STATUS"
"É.reducing the viral replication should result in lower cirrhosis risk in both HBeAg positive & negative subjects."
C.J. Chen1, H.I. Yang1, J. Su2, C.L. Jen1, E. Kuo3, S.L. You1, U.H. Iloeje2
1 National Taiwan University, Taipei, Taiwan
2 Bristol-Myers Squibb Pharmaceutical Research Institute, Wallingford, USA
3 Bristol-Myers Squibb, Taipei, Taiwan
HBeAg is considered a marker of active viral replication often associated with high levels of viremia. This study was carried out to examine the impact of HBV DNA level on the risk of disease progression to cirrhosis stratified by HBeAg status.
A population based prospective cohort of 3851 subjects chronically infected with HBV was established was from seven townships in Taiwan between 1991 and 1992. Subjects were prospectively followed by hepatologists by clinical examinations including ultrasonography through June 30th 2004. The diagnosis of cirrhosis was based on ultrasonographic findings. All cirrhosis cases diagnosed within 6 months of enrollment were excluded from analyses. Multivariable adjusted relative risks (RRadj) were derived using Cox proportional hazard models.
Results
Overall, 3774 subjects with 42,115 person years of follow up contributed data to this analysis. There were 395 cases of cirrhosis.
Of the 3774 participants, 3214 (85%) were seronegative for HBeAg, of which 1082 (34%) had serum HBV DNA level ³104 copies/ml; 560 (15%) were HBeAg positive of which 538 (96%) had serum HBV DNA ³104 copies/ml at enrollment. There was a dose dependent relationship between HBV DNA and cirrhosis risk within the HBeAg strata.
With the HBeAg negative undetectable DNA group as reference, the highest risk of progression was found in the HBeAg positive group with HBD DNA over 105 copies/ml (>100,000 copies/ml).
Subjects with HBV DNA level of 1.0-9.9x10 to the 5th and >1 million (10 to the 6th) had very similar incidence rates of cirrhosis regardless of HBeAg status.
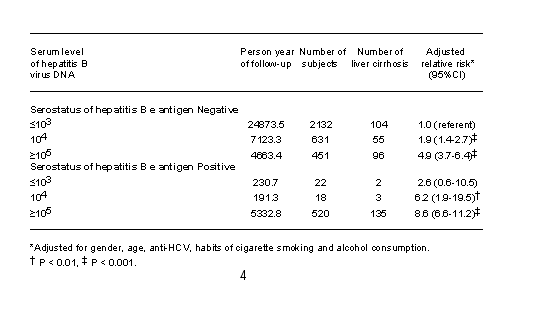
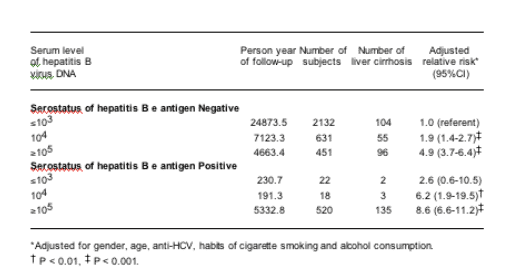
Author Conclusion: Elevated serum HBV DNA is a strong predictor of cirrhosis risk in HBV infected persons regardless of HBeAg status. Effective suppression of HBV DNA to very low levels especially in HBeAg negative persons could reduce progression of CHB to cirrhosis.
|
|
| |
| |
| |
|
 |
 |
|
|